Angus Liu
116 posts

Angus Liu
@Angus_Fierce
Biopharma reporter @FiercePharma & @FierceBiotech. Medill MSJ 16
انضم Kasım 2013
619 يتبع762 المتابعون

The FDA's press release says zongertinib's application was FILED on Jan. 13, 2026. But Boehringer told us that they actually completed the package under real-time oncology review on Nov. 14, 2025, and that it was ACCEPTED on Jan. 13.
Stephen V Liu, MD@StephenVLiu
A new first line standard of care for HER2 mutant NSCLC? Zongertinib receives accelerated approval as initial therapy based on Beamion LUNG-1 trial with 1L RR 76%. Part of the new FDA voucher program, approval granted 44 days after filing! fda.gov/news-events/pr…
English

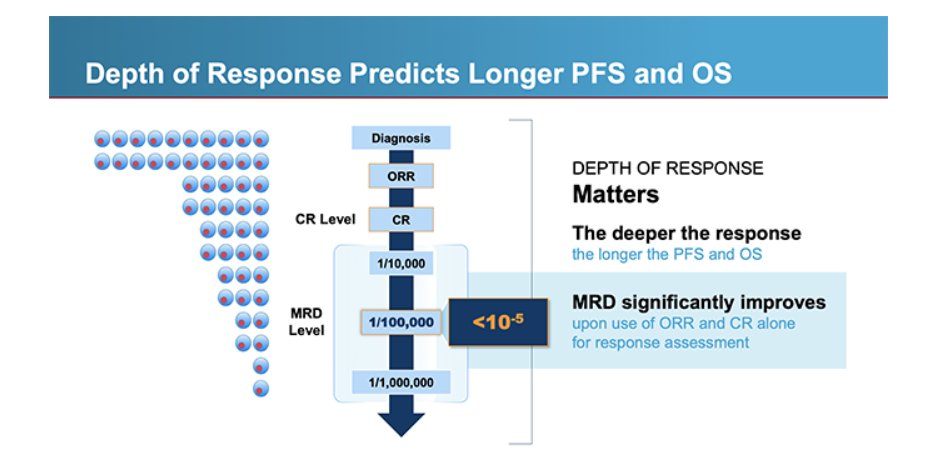
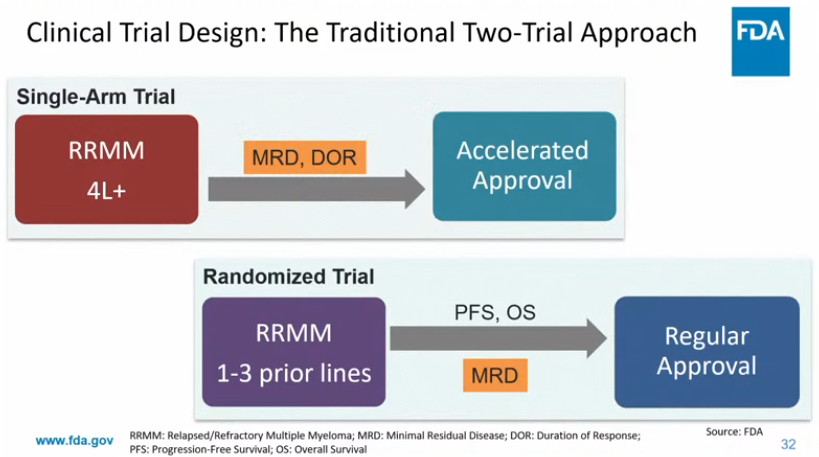
@JacobPlieth @PearlF I think there's a difference between "can" and "should/must." FDA is not "requiring" MRD. The language is "MRD can be used as an endpoint to support accelerated approval in MM"
English

@PearlF @Angus_Fierce Requiring MRD (and not just CR rate) would set a higher bar for an accelerated approval in multiple myeloma, no?
English

FDA issued a draft guidance on the potential use of minimal residual disease (MRD) & complete response (CR) to support accelerated approval of drugs to treat multiple myeloma #mmsm. It was on FDA's agenda in '25 but had been delayed by new FDA/HHS leaders said @FierceBiotech. A link to the story is in the reply

English

@JacobPlieth @PearlF @AuclairDan I think the focus is on earlier line given that FDA is already fully comfortable with late-line approvals based on ORR/CR. Even though earlier-line doesn't get singled out, the guidance makes it a possibility. But I agree, I'd be more comfortable if there were specific language
English

@PearlF @Angus_Fierce @AuclairDan I'm not sure... the only clarity from the ODAC seemed to be this: possibility of a single trial to back AA **in a relatively early setting** But the FDA guidance doesn't seem to spell this out

English

@DisneyAnimation @Disney @AMCHelps
@AMCTheatres I went to see Zootopia 2 in
@RealD3D at AMC Courthouse Plaza on 11/27 1110am based on AMC's ad promising a character keychain. But it was not available. CS refused a satisfactory resolution. False advertising is unacceptable @FTC

English

FDA unveils ‘plausible mechanism pathway,’ a new approach that could trigger a seismic shift in how bespoke gene editing therapies are developed and approved. fiercebiotech.com/biotech/fdas-n…
English

Was just wondering what Vivek's been up to.
The Hill@thehill
Vivek Ramaswamy set to launch Ohio governor bid on Feb. 24 trib.al/8DJ4C6p
English
Angus Liu أُعيد تغريده

Akeso, Summit's ivonescimab crushes Merck's Keytruda in China study, signaling potential new standard of care in lung cancer #WCLC24 fiercepharma.com/pharma/akeso-s…
English
Angus Liu أُعيد تغريده

Before approving Amgen's breakthrough lung cancer med Imdelltra, FDA flagged 'a large number' of missing adverse events from a pivotal trial. fiercepharma.com/pharma/approve…
English
Angus Liu أُعيد تغريده

Eli Lilly invests additional $5.3B for production of diabetes, obesity drugs in Indiana fiercepharma.com/manufacturing/…
English
Angus Liu أُعيد تغريده

WuXi Biologics has quietly withdrawn from the BIO International Convention 2024 amid worsening geopolitical tensions fiercepharma.com/pharma/wuxi-bi…
English

More background on the FDA's first-of-its-kind, confirmatory trial-related CRL for Regeneron. fiercebiotech.com/biotech/what-d…
English

Just imagine this: Every case of an important update being buried in an SEC filing is fed to an AI somewhere fiercebiotech.com/biotech/modern…
English
Angus Liu أُعيد تغريده

Special report: CAR-T hype faces infrastructure reality check fiercepharma.com/pharma/mid-dec…
English
Angus Liu أُعيد تغريده

Novo Nordisk answers Eli Lilly's challenge in obesity treatment with its own head-to-head trial for a Wegovy combination against Zepbound. Details in story. fiercepharma.com/pharma/novo-no…
English
Angus Liu أُعيد تغريده

The FDA has approved Merck's Keytruda as part of a neoadjuvant-plus-adjuvant therapy for resectable non-small cell lung cancer. The label already includes overall survival data. fiercepharma.com/pharma/mercks-…
English
Angus Liu أُعيد تغريده

In the Breakthroughs category, we’ll recognize organizations, clinicians, researchers and others whose work has pushed the boundaries of knowledge and improved the lives of countless individuals. fiercepharma.com/fierce-50/fier… #Fierce50 #WeAreFierce
English

Merck and Moderna's phase 3 plan for their mRNA cancer vaccine is here. $MRK $MRNA fiercebiotech.com/biotech/merck-…
English

This week's The Top Line podcast from @FiercePharma includes a recap of the star-studded ADA meeting omny.fm/shows/the-top-…
English