qmsWrapper
3.9K posts

qmsWrapper
@qmsWrapper
qmsWrapper is QMS software that helps all companies increase productivity with ISO 9001: 2015 or QMS for MedDev creators. [email protected]
Canada انضم Şubat 2016
1.3K يتبع706 المتابعون

Every QMS looks good on paper.
Until someone actually has to use it.
Recognized as a High Performer in G2 Spring 2026 (Medical QMS, Europe).
We don’t just manage documents.
We connect decisions.
→ See how it works: store.qmswrapper.com
#MedTech #QMS #ISO13485




English

In many MedTech companies, change control starts weeks after the first signal appears.
By then the most important part of the story is already missing.
Auditors don't just ask for the change request.
They ask when the decision actually began.
↓
shorturl.at/0FQza

English

The audit question that breaks most QMS tools:
“Show me how this issue connects to risk, design, and verification.”
The data exists. The connections don’t.
Why semantic AI search is becoming the missing glue in MedTech QMS ↓
shorturl.at/IuQT9

English

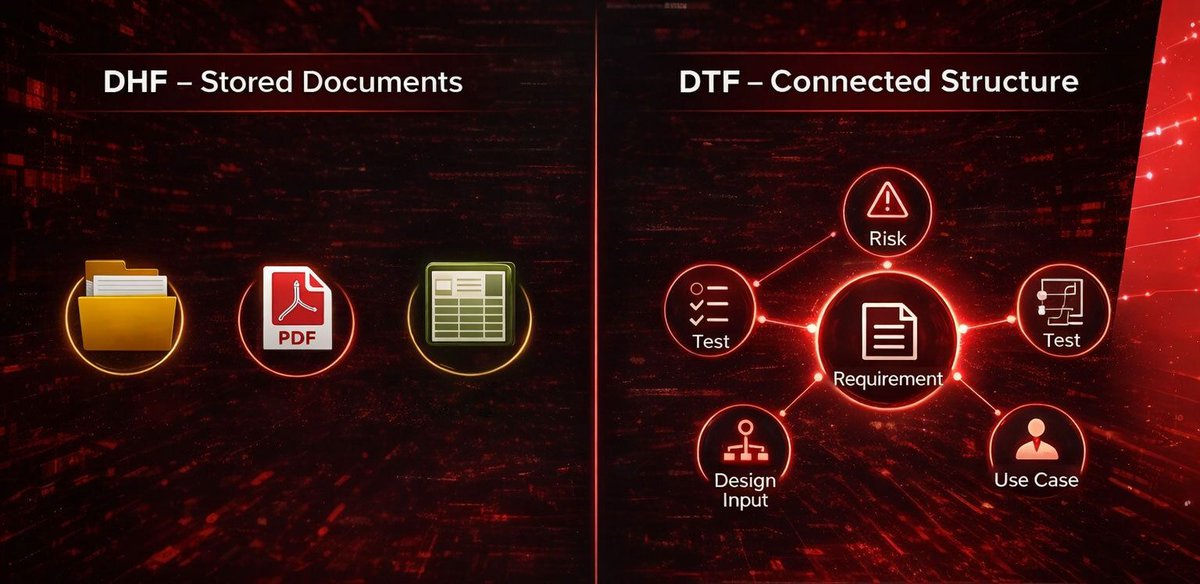
Traceability doesn’t fail during audits.
It fails during change.
DHF = stored documents.
DTF = connected structure.
If one requirement update breaks your chain, this is worth 3 minutes.
shorturl.at/b7Ivk
English

Can you instantly show:
→ impacted risks
→ linked tests
→ updated records
If not, your traceability is static — not structural.
New article: shorturl.at/b7Ivk
Why Traceability Breaks the Moment a Requirement Changes
#MedTech #ISO13485 #DesignControls #QMS

English

Action without structure creates audit pressure.
But structure usually breaks even earlier.
When something happens,
most systems force you to decide immediately:
CAPA?
Risk?
Change?
What if you didn’t have to decide yet?
#QES #EventDrivenQMS #ISO13485 #MedTech

English

Most audit gaps don’t start in CAPA.
They start in chat messages that never became records.
We wrote about the “shadow QMS” every MedTech team unknowingly runs —
and how to close that gap without creating more bureaucracy.
5 min read.
shorturl.at/PDYAG

English

#QMSR key changes aren’t about more procedures.
They’re about connected evidence.
FDA inspections now follow signals across risk, design, CAPA, and records — not static documents.
What this means in practice 👇
qmswrapper.com/qmsr-key-chang…
#FDACompliance #ISO13485 #MedTechQuality

English

Checklist shows gaps.
Systems prevent them.
That’s the difference between approvals as tasks and approvals as part of your QMS.
See how approval workflows work in qmsWrapper.
👉 See how it works meet.brevo.com/qmswrapper-dem…

English

If you can’t answer these in 2 minutes, your system can’t either.
Can you enforce reviewer order?
Can you retrieve approval history fast?
Do outdated versions get blocked?
Quick approval self-check ↓
qmswrapper.com/approval-workf…
#ApprovalWorkflows #ISO13485 #QMS

English

Approval isn’t a final step.
It’s evidence of how a decision was made.
Auditors don’t just look for signatures — they look for structure behind them.
We outlined what auditors expect from approval workflows and where systems most often fall short.
👉 qmswrapper.com/approval-workf…

English

Approval workflows aren’t just signatures.
Auditors review how decisions were made — roles, routing, versions, traceability.
We summarized what auditors expect under ISO 13485 & FDA.
👉 Read the article qmswrapper.com/approval-workf…
#ApprovalWorkflows #ISO13485 #QMS #MedTech

English

Checklists show gaps.
Systems prevent them.
Why approval workflows fail as tasks — and how qmsWrapper embeds them into the QMS.
👉 Book a meeting to see how approval workflows are built into qmsWrapper.
meet.brevo.com/qmswrapper-dem…
#ApprovalWorkflows #QMS
English

We mapped the most common approval gaps auditors flag —
not as theory, but as patterns we see repeatedly during audits.
Sharing the overview here ↓
qmswrapper.com/approval-workf…
English

Email-only approvals.
Missing timestamps.
“final_v2_update”.
Auditors don’t call these mistakes.
They call them findings.
Most approval findings aren’t about missing signatures — they’re about approvals that can’t be explained.
#ISO13485 #QMS #AuditReadiness #MedTech

English

