Vitaccess
2.1K posts


Vitaccess
@vitaccess
Helping biopharma generate real-world data and insights Vitaccess combines technology and patient-centered #realworldevidence #heor #consulting #data #analytics
Oxford, England انضم Haziran 2016
612 يتبع468 المتابعون

The myasthenia gravis (MG) treatment landscape is evolving at a rapid pace. In our latest blog, Vitaccess Founder and CEO, Mark Larkin, discusses how recent progress is reshaping both clinical and value frameworks in MG care.
Read the blog here: bit.ly/4aCidyx

English

💙 Today, on Rare Disease Day, we stand with the 300 million people worldwide living with a rare disease and their families.💜
bit.ly/3QljMWk
English

Next Friday, 28th February, we will be celebrating Rare Disease Day.
Rare Disease Day is a global movement to raise awareness and generate change in rare diseases, coordinated by @EURORDIS and 65+ national alliance patient organization partners.
hubs.ly/Q030ZY520

English

To move beyond symptom management to disease modification, remission, and cure, a deeper understanding of immunological diseases is necessary.
Real-world research provides opportunities to gather a wealth of information. Get in touch to find out more. hubs.ly/Q030ZTMK0

English

The idea behind the Delphi panel is to gain consensus through systematic and structured feedback from a group of subject experts. Learn some tips for conducting Delphi panels in our infographic.
Get in touch with us today at hubs.ly/Q030ZrfB0.

English

How much does a real-world study cost? Read our blog to understand the costs behind real-world research and learn why spending money on a real-world study could be a worthwhile investment. hubs.ly/Q030ZW-00

English

Chronic autoimmune and immune-mediated diseases take a toll, not only on patients, but on their families and caregivers too.
Read this case study describing research we conducted to generate patient- and caregiver-reported data in myasthenia gravis.
bit.ly/4fHGC4N

English

What type of data can you collect in a real-world immunology study?
We've got immunology on the mind at Vitaccess this month. Contact us at bit.ly/4gYPu6S to speak to a member of our team and find out how we can support your evidence generation needs.

English

Have a patient-centered research need, but not clear on the optimal study design?
Get in touch to chat through your objectives with our experts and we can produce a high-level summary of the possible study design. hubs.ly/Q030ZHr80
English

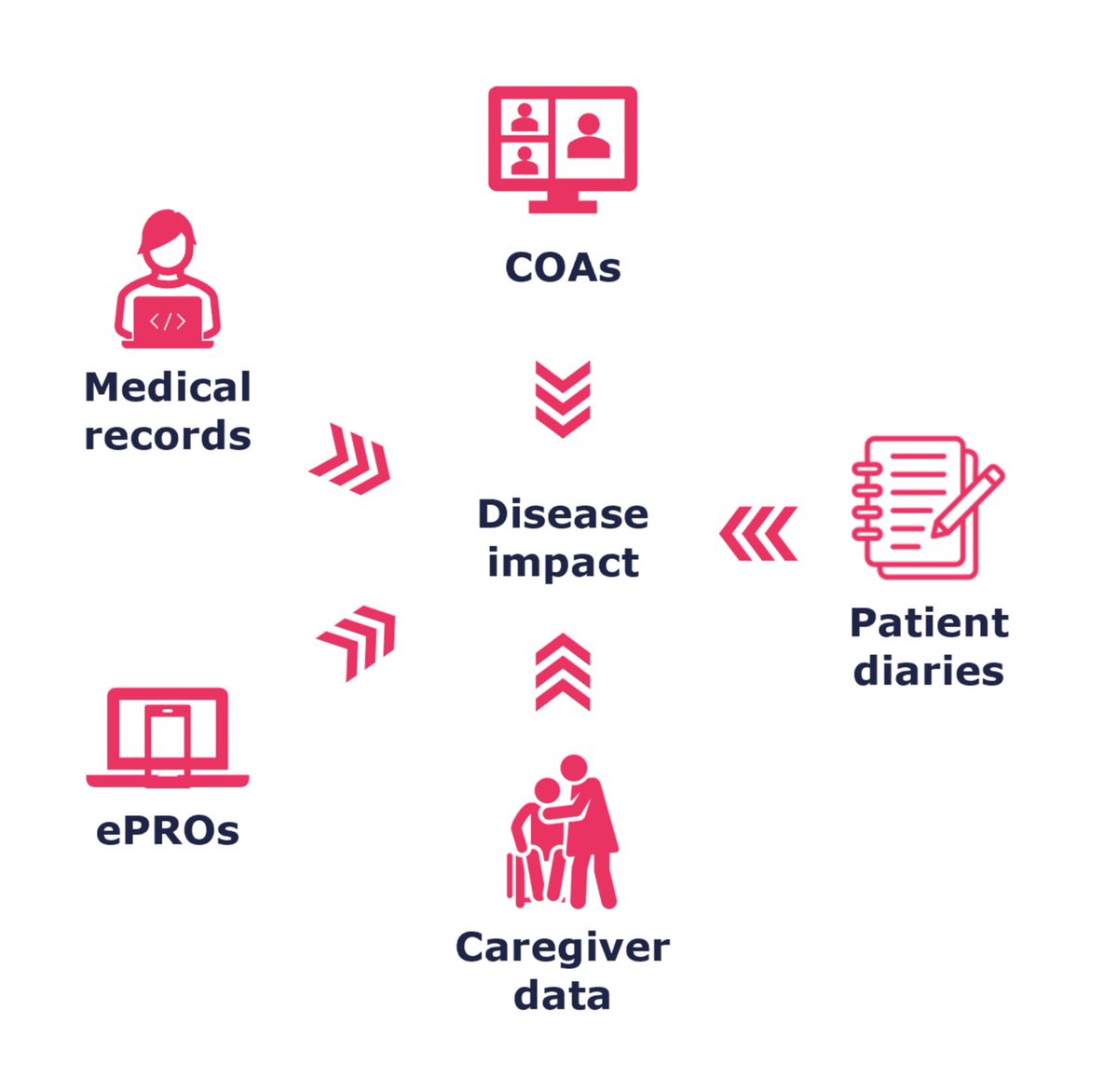
For a comprehensive understanding of complex immune-related diseases, it is important to capture data from a range of sources.
Work with our experts to identify and select the data sources and outcomes most relevant to your research questions.
bit.ly/3W6JwsL
English

The two sides of having a sibling with a medical condition.
To learn about our research in the fields of family, caregiver, and sibling impact, get in touch at info@vitaccess.com.

English

We offer flexible research opportunities with existing and upcoming global registries. Get in touch to discuss our syndicated solution. hubs.ly/Q02_0SvX0

English

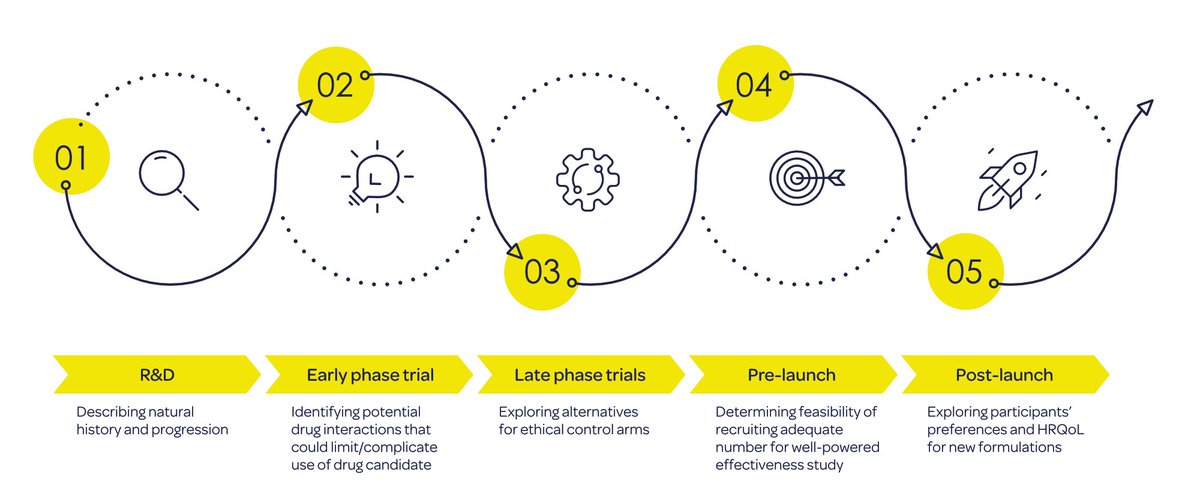
'Real from start to finish: making the most of real-world data from disease registries across the product lifecycle'. In this blog, we shared some insights from our experiences with building disease registries. bit.ly/3BkzzRn #registries #RWD
English

What do you know about our patient-centered outcomes service offering? We are experts in understanding, measuring, and articulating patient experience in the real world. Find out about our end-to-end service offering with case studies on our website. bit.ly/3BbSFcu

English

Whether your research requires site-based recruitment or high accessibility with community recruitment via patient advocacy groups and social media, we can reach patients around the globe. Speak to us to learn more. bit.ly/4gqdBLM
#RWE #realworldstudies

English

If you missed #ISPOREurope last week, all of the publications we presented can be found on our website: bit.ly/3XRYaUR
English

And that's a wrap for #ISPOREurope 2024! You can read all of our publications from the conference here: bit.ly/3BDCvYW Get in touch if you weren't able to catch up in person to discuss how we do patient-centered research differently.

English


