



Léo PioLopez
20 posts

@LPiolopez
Complex systems, embodied cognition, AI, networks








Trending in #Biophysics: ooir.org/index.php?fiel… 1) Neural cellular automata 2) Expanding Histone Universe 3) Biological complexity beyond networks 4) Mitochondrial position in pancreatic beta cell (@BiophysJ) 5) Promoter-proximal pausing & transcription control (@NatureSMB)


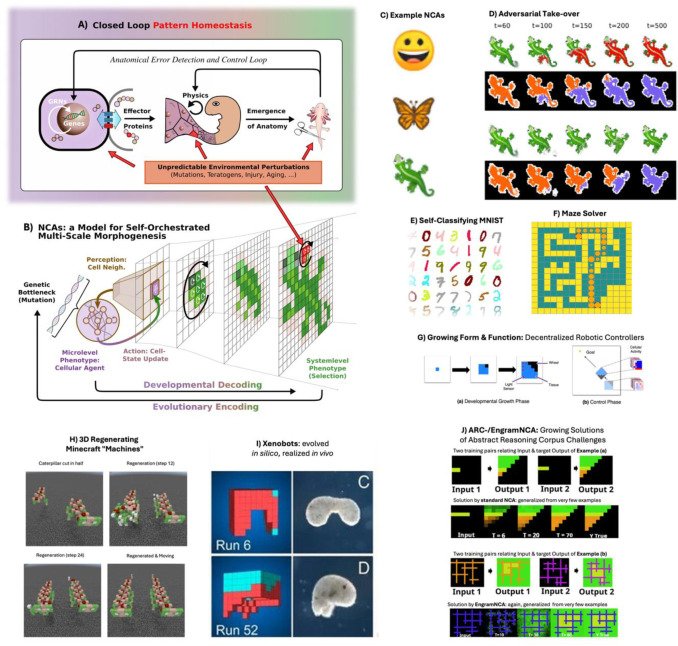
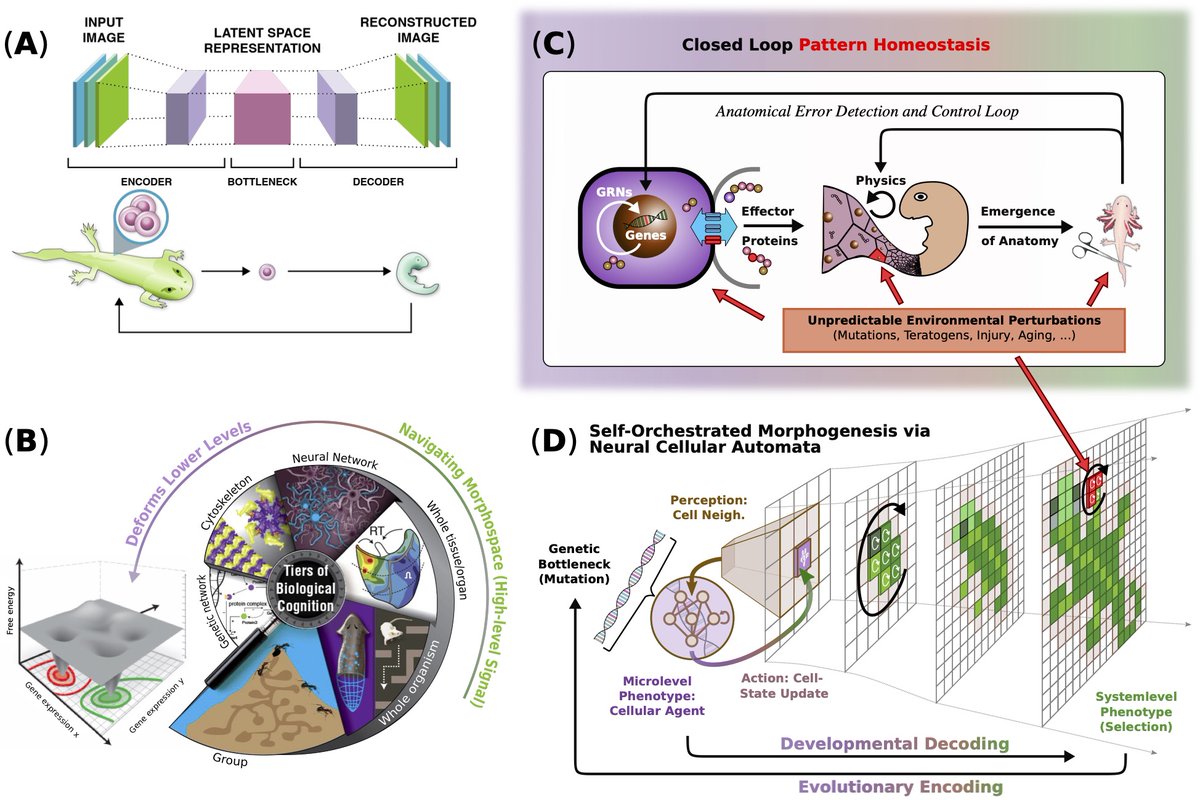
Final version is out: authors.elsevier.com/c/1mEoa5bD-sxf… "Neural cellular automata: Applications to biology and beyond classical AI" @LPiolopez Benedikt Hartl "Neural Cellular Automata (NCA) represent a powerful framework for modeling biological self-organization, extending classical rule-based systems with trainable, differentiable (or evolvable) update rules that capture the adaptive self-regulatory dynamics of living matter. By embedding Artificial Neural Networks (ANNs) as local decision-making centers and interaction rules between localized agents, NCA can simulate processes across molecular, cellular, tissue, and system-level scales, offering a multiscale competency architecture perspective on evolution, development, regeneration, aging, morphogenesis, and robotic control. These models not only reproduce canonical, biologically inspired target patterns but also generalize to novel conditions, demonstrating robustness to perturbations and the capacity for open-ended adaptation and reasoning through embodiment. Given their immense success in recent developments, we here review current literature of NCAs that are relevant primarily for biological or bioengineering applications. Moreover, we emphasize that beyond biology, NCAs display robust and generalizing goal-directed dynamics without centralized control, e.g., in controlling or regenerating composite robotic morphologies or even on cutting-edge reasoning tasks such as ARC-AGI-1. In addition, the same principles of iterative state-refinement is reminiscent to modern generative Artificial Intelligence (AI), such as probabilistic diffusion models. Their governing self-regulatory behavior is constraint to fully localized interactions, yet their collective behavior scales into coordinated system-level outcomes. We thus argue that NCAs constitute a unifying computationally lean paradigm that not only bridges fundamental insights from multiscale biology with modern generative AI, but have the potential to design truly bio-inspired collective intelligence capable of hierarchical reasoning and control."


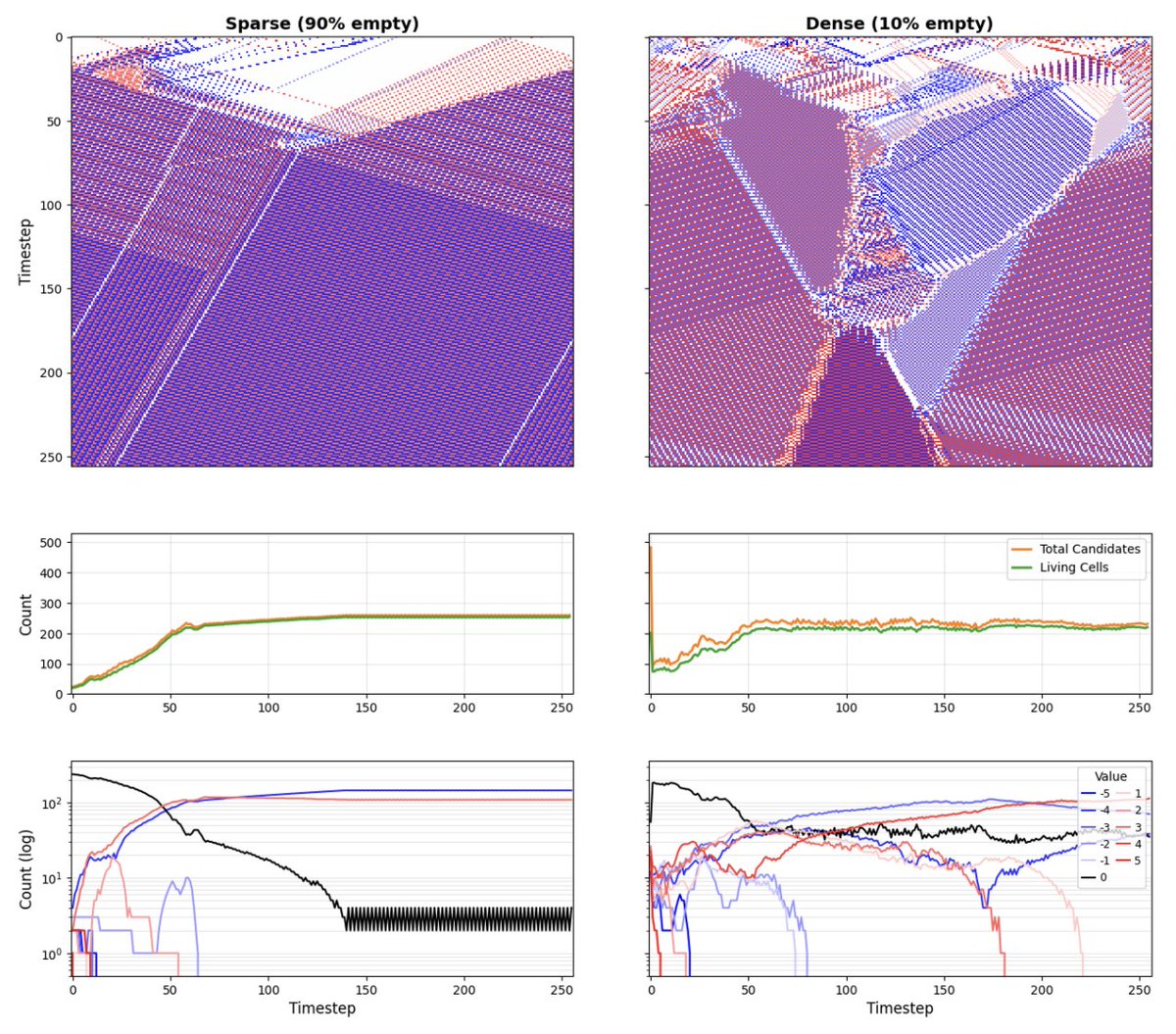
Final version is out: aging as the result of loss of goal-directedness advanced.onlinelibrary.wiley.com/doi/epdf/10.10… @BeneHartl @LPiolopez "Although substantial advancements are made in manipulating lifespan in model organisms, the fundamental mechanisms driving aging remain elusive. No comprehensive computational platform is capable of making predictions on aging in multicellular systems. Focus is placed on the processes that build and maintain complex target morphologies, and develop an insilico model of multiscale homeostatic morphogenesis using Neural Cellular Automata (NCAs) trained by neuroevolution. In the context of this model: 1) Aging emerges after developmental goals are completed, even without noise or programmed degeneration; 2) Cellular misdifferentiation, reduced competency, communication failures, and genetic damage all accelerate aging but are not its primary cause; 3) Aging correlates with increased active information storage and transfer entropy, while spatial entropy distinguishes two dynamics, structural loss and morphological noise accumulation; 4) Despite organ loss, spatial information persists in tissue, implementing a memory of lost structures, which can be reactivated for organ restoration through targeted regenerative information; and 5) rejuvenation is found to be most efficient when regenerative information includes differential patterns of affected cells and their neighboring tissue, highlighting strategies for rejuvenation. This model suggests a novel perspective on aging caused by loss of goal-directedness, with potentially significant implications for longevity research and regenerative medicine."

















1/n Very excited to put this paper out in the world! Does reduction always lead to a gain in information? Turns out: no. @ThosVarley and I show macroscales can convert information (making info more synergistic), and reduction can lead to a loss of info arxiv.org/abs/2104.13368

