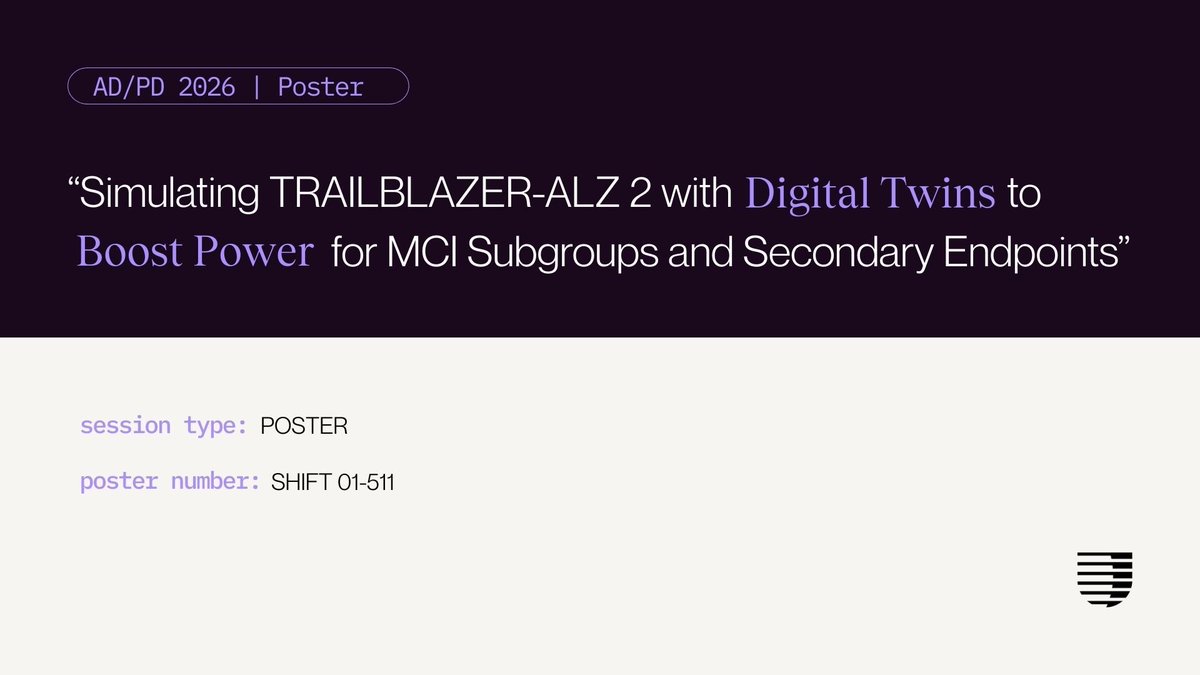
#ICYMI: 🎉 Unlearn is excited to partner with SOLA Biosciences on an early-phase ALS study of SOL-257, supported by our AI-generated #digitaltwins. We're grateful for the opportunity to help advance clinical research for people living with #ALS. Read the full announcement:
na2.hubs.ly/H04kxv_0

English