analyze&realize GmbH
966 posts


analyze&realize GmbH
@analyze_realize
Your innovation experts in natural health products. https://t.co/3dvlCa1aOi https://t.co/7QFY5xVbC8
Berlin, Germany Beigetreten Şubat 2016
542 Folgt571 Follower

❗ The new Contaminants Regulation (EU) 2023/915 enters into force today. Here are the most important changes at a glance.
lnkd.in/gudJnmnV
#regulatoryaffairs #regulatorycompliance #contaminants #eu #riskmanagement
English

#Fermented beverages and foods are on the rise. Extension of shelf-life and development of useful metabolites - to name just a few of the benefits.
But what about the legal framework of food cultures? To find out more about the regulatory challenges: a-r.com/dissent-about-…
English

👉 𝗘𝘅𝗶𝘀𝘁𝗶𝗻𝗴 𝗨𝗟𝘀 𝗳𝗼𝗿 𝗳𝗼𝗹𝗶𝗰 𝗮𝗰𝗶𝗱 𝘁𝗼 𝗯𝗲 𝗿𝗲𝘁𝗮𝗶𝗻𝗲𝗱!
As reported in a previous newsletter article, EFSA is currently updating the safety thresholds for certain vitamins and minerals (). Now EFSA has pu…lnkd.in/ejp_9rSP buff.ly/3Hin6NK
English

Looking for a CRO with extensive experience in #naturalhealth products?
We provide support in the development of highly customized study designs covering a broad range of #clinicaltrials types and indications.
Contact us today to learn how we can support you!

English

💡 Enjoy the latest issue of a&r NEWS DIGEST with exciting articles on the Green Claim Directive, the dissent on the classification of food cultures, and active tracking in clinical trials. Don't miss our SPECIAL OFFER! #newsletter…lnkd.in/ewJ7hsM6 lnkd.in/e3SudxMN
English

The global consumer #healthcare market is extremely dynamic.
In this highly competitive market, a&r provides a unique combination of scientific & #regulatory expertise to support market assessments and the creation of innovative solutions.
Connect with us to meet at #Vitafoods!

English

a&r offers expert advice on regulatory and scientific issues, in particular for ingredients that may require pre-market authorization, such as:
- Novel Foods
- Food Improvement Agents,
Please get in touch arrange a meeting at #vitafoodseurope in Geneva.

English

Are you manufacturing food enzymes or use enzymes to manufacture your innovative food ingredients, additives or flavourings?
EFSA implemented numerous updates to the model used in the exposure assessment of food enzymes and initiated a public consultation…lnkd.in/erJ6SrkB
English

💡Did you know that 53% of all #greenclaims made are misleading, vague, or lacking evidence?
▶Now, the EU Commission's proposed Green Claims Directive aims to stop #greenwashing. Green claims will only be allowed if substantiated by scientific evidence.
lnkd.in/e_z9MDGZ
English

Italy is attempting to ban the production of #culturedmeat, citing concerns over consumer safety and the importance of preserving the agricultural heritage. However, it is unclear how the decision will be affected by #novelfood authorizations at EU level. a-r.com/italy-plans-th…
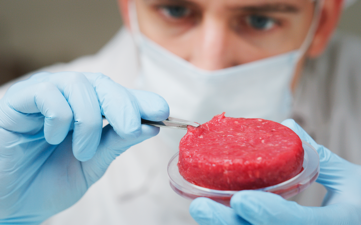
English

💡 Enjoy the latest issue of a&r NEWS DIGEST with exciting articles on the interpretation of #FSMPs, the #novelfood status of sodium hyaluronate, some botanical extracts under scrutiny, recent novel food and #healthclaim authorizat…lnkd.in/eiTy6Ra6 lnkd.in/eaFyF4aP
English

#EFSA is currently in the process of updating the UL for several #vitamins and #minerals. Read our comments in NutritionInsight on the ongoing safety assessments and the potential implications for manufacturers. buff.ly/3ZblqfW
English

💡 Enjoy the latest issue of a&r NEWS DIGEST with exciting articles on the extension of the MDR transition period, the future of CBD in Europe, eDiary in clinical trials and an EFSA upgrade or reduction of ULs. #clinicaltrials #new…lnkd.in/ewpZHeNx lnkd.in/etvYHhr4
English

Do not give up on your medical device, due to the extension of the transition periods there is still the opportunity to update the certificates in time. Let us help you navigate the new regulations. #MDR #IVDR #medicaldevices #regu…lnkd.in/eKGDnMuT lnkd.in/ewc5GKtn
English

Do you wonder what is currently going with #CBD in the EU or the UK?
Please read the comments of a&r’s #NovelFood expert Steffi Dudek in NutraIngredients.
buff.ly/3I9iM2Y
English

Last month EFSA updated the list of microorganisms with QPS status. Read our article explaining QPS approach and the difference to #Novelfood status:
buff.ly/3BRM1nG
English

Important news for the US:
As part of the recently updated "Health Products Compliance Guidance", the Federal Trade Commission (FTC) announced that #clinicaltrials for dietary #supplements are required to substantiate #healthclaims. buff.ly/3IbUwyv
English

The marketing of #CBD products as dietary #supplements is not admissible and the future regulatory classification of CBD is unclear, as the FDA announces that it will work with Congress on a new pathway.
buff.ly/3YEg5xh
English

Know today what tomorrow will bring:
Check out our Managing Director’s outlook and his expectations for the #consumerhealth industry in 2023.
buff.ly/3WMIXln
Do you share our view? What are your predictions for 2023?
English