Tim
733 posts




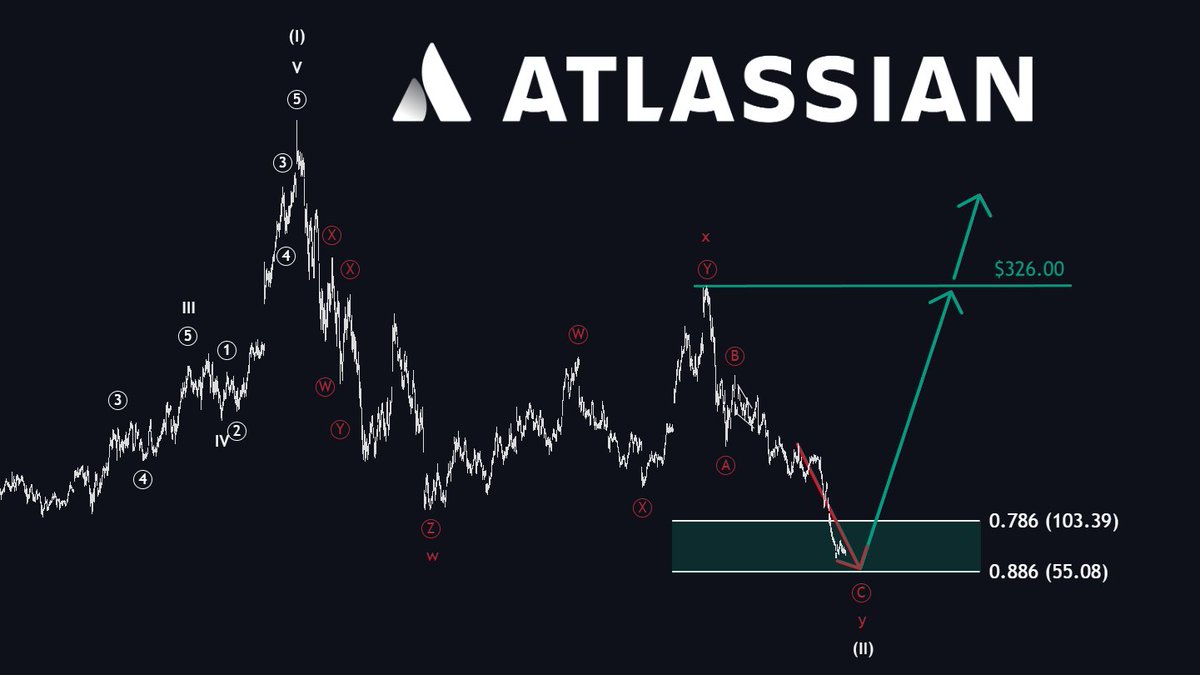
No major changes to the $ASTS chart as of yet. Holding firm for now as it attempts a higher low to initiate MW5 of PW5 to complete LT Wave 3. It does not want to lose $82, and beyond that, the range of $71-$76. This will open a whole new can of worms and facilitate early onset of LT Wave 4. With the recent outperformance of $PL after a stellar earnings and SpaceX IPO on the horizon, it is clear the space basket has continued interest. However, $RKLB appears heavier than $ASTS and $PL. I still contend that $RKLB wants to visit ~$50, roughly 25-30% lower from current levels. It is known that algorithms love to trade these names altogether. It brings into question the direction $ASTS wants to move going forward. Do these companies finally diverge in price action? We can only wait and see.


@TradeCycles What do you think about $RKLB and $KRKNF






Humacyte® has received a minimum $1.475M purchase commitment for our Symvess® acellular tissue–engineered vessel-tyod to support a clinical evaluation and outreach program in the Kingdom of Saudi Arabia. This program will help expand awareness of Symvess among healthcare providers across KSA as we continue discussions on establishing a joint venture and commercialization license in the region. This is another important step in our global expansion efforts and we look forward to supporting medical teams as they evaluate the potential of Symvess to improve patient care. For more details read the full press release here: 🔗 bit.ly/3PGVQiZ #Humacyte #Symvess #Biotechnology #GlobalHealth #TissueEngineering #RegenerativeMedicine #MedicalInnovation #ATEV #Innovation PLEASE SEE IMPORTANT SAFETY INFORMATION INCLUDING THE BOXED WARNING, AND FULL PRESCRIBING INFORMATION at bit.ly/4dl5tuL INDICATION SYMVESS is an acellular tissue engineered vessel indicated for use in adults as a vascular conduit for extremity arterial injury when urgent revascularization is needed to avoid imminent limb loss, and autologous vein graft is not feasible. GRAFT FAILURE: loss of SYMVESS integrity due to mid-graft rupture or anastomotic failure can result in life threatening hemorrhage. DO NOT use SYMVESS in patients who have a medical condition that would preclude long-term antiplatelet therapy after acute injury resolution. WARNINGS AND PRECAUTIONS: Vascular Graft Rupture, Anastomotic Failure, and Thrombosis have occurred in patients treated with SYMVESS. SYMVESS is manufactured using cells and reagents that may transmit infectious diseases or agents; none have been reported during clinical testing. ADVERSE REACTIONS: most common (≥3%): thrombosis, fever, pain, anastomotic stenosis, rupture or anastomotic failure, and infection. Trademarks are property of Humacyte Global, Inc © 2026 Humacyte Global Inc. All Rights Reserved.

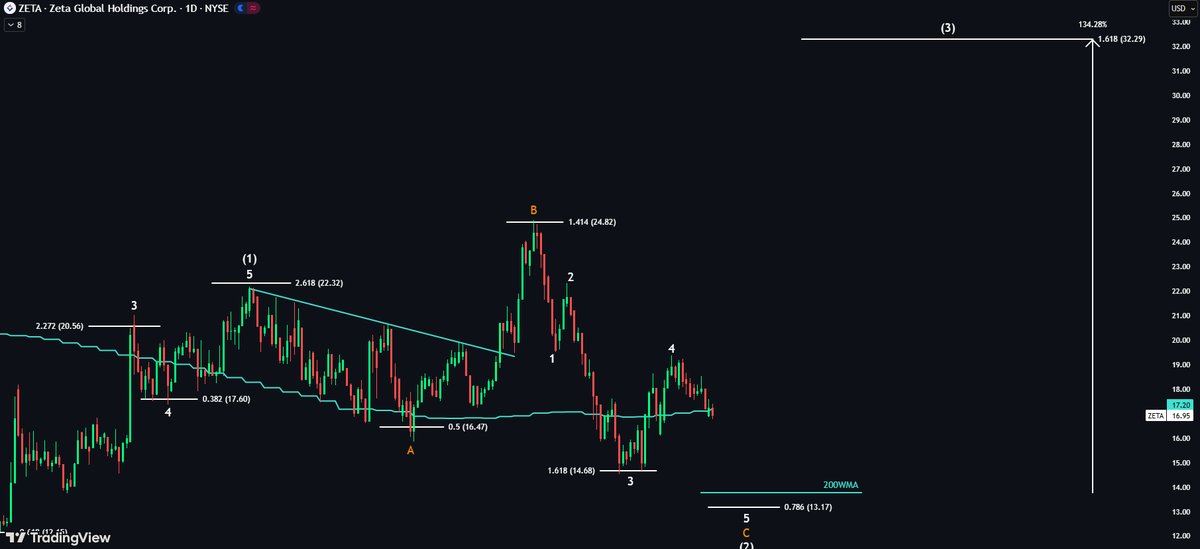
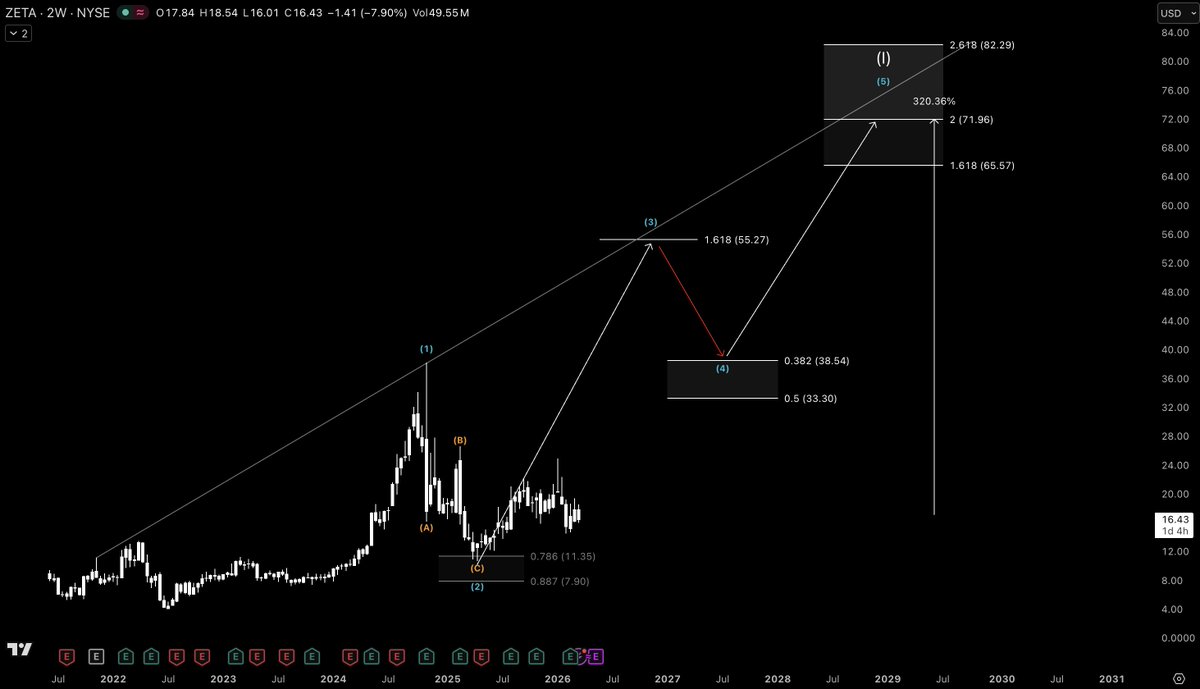
$ZETA — Pullback isn’t over ⚠️ Price is now testing the 50WMA. Losing this level signals bears are in control. I still expect a test of the 200WMA, that’s where I load the boat. Wave 3 target: +138% 🚀