ClearPath me-retweet
ClearPath
568 posts

ClearPath
@ClearPath100
Particular focus on bio, fintech, deeptech and broader vertical AI themes.
Bergabung Mart 2025
375 Mengikuti314 Pengikut

@Ichaka_001 Massively fat tails, everything that was working has huge expects baked in, out of their control
English

$SPX $QQQ This is the fourth consecutive weekly decline for both S&P 500 and Nasdaq.
The confluence that is driving today’s move:
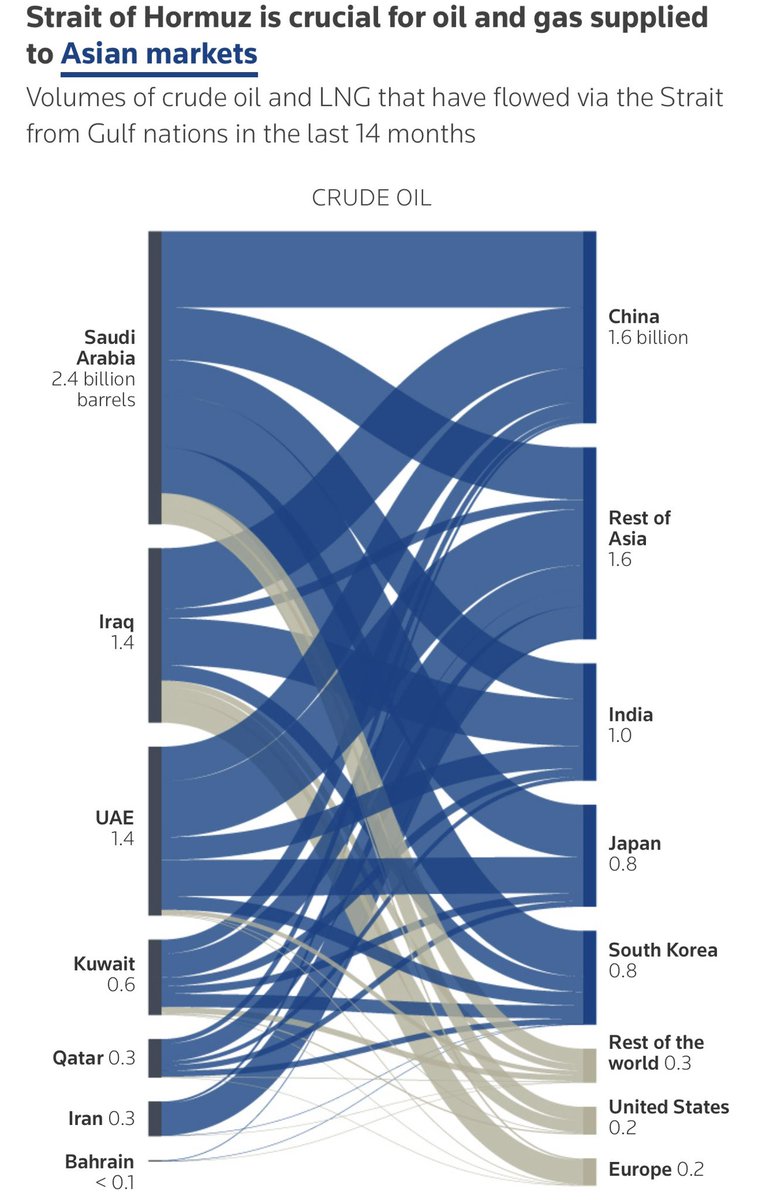
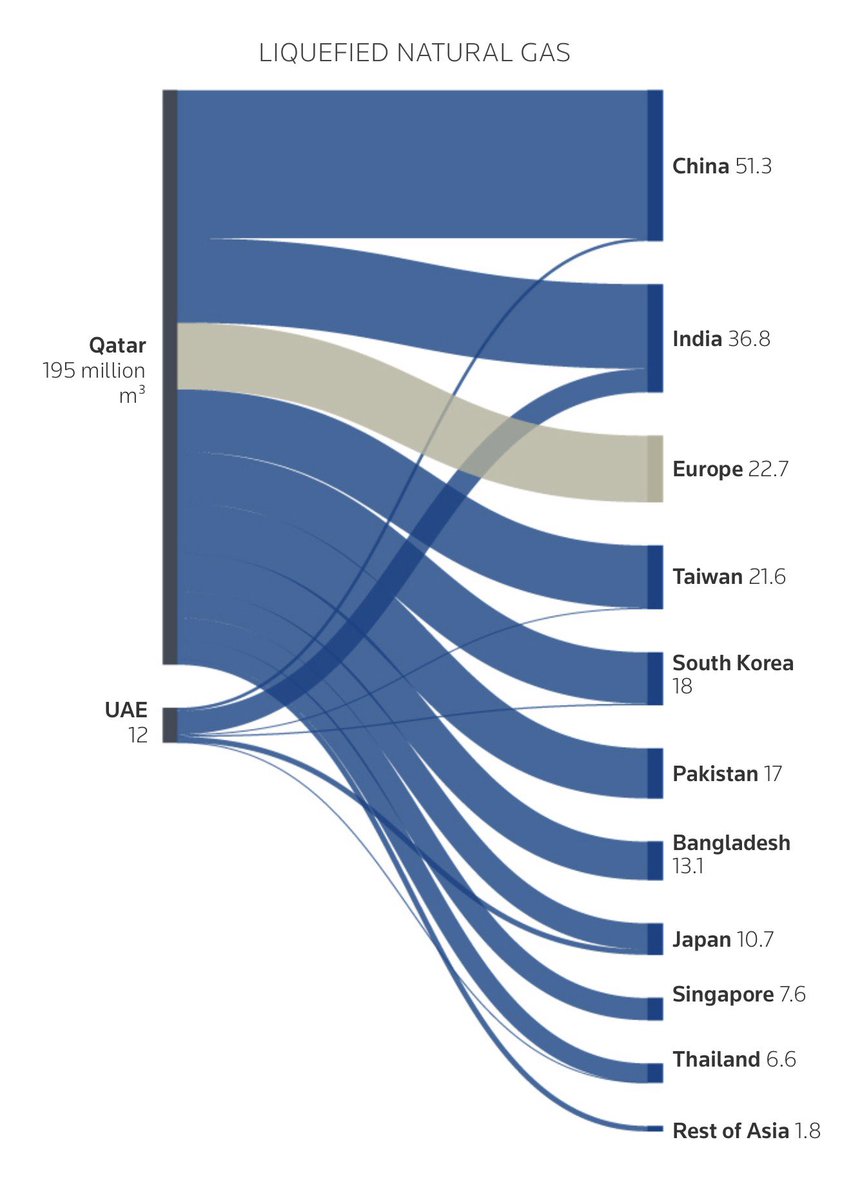
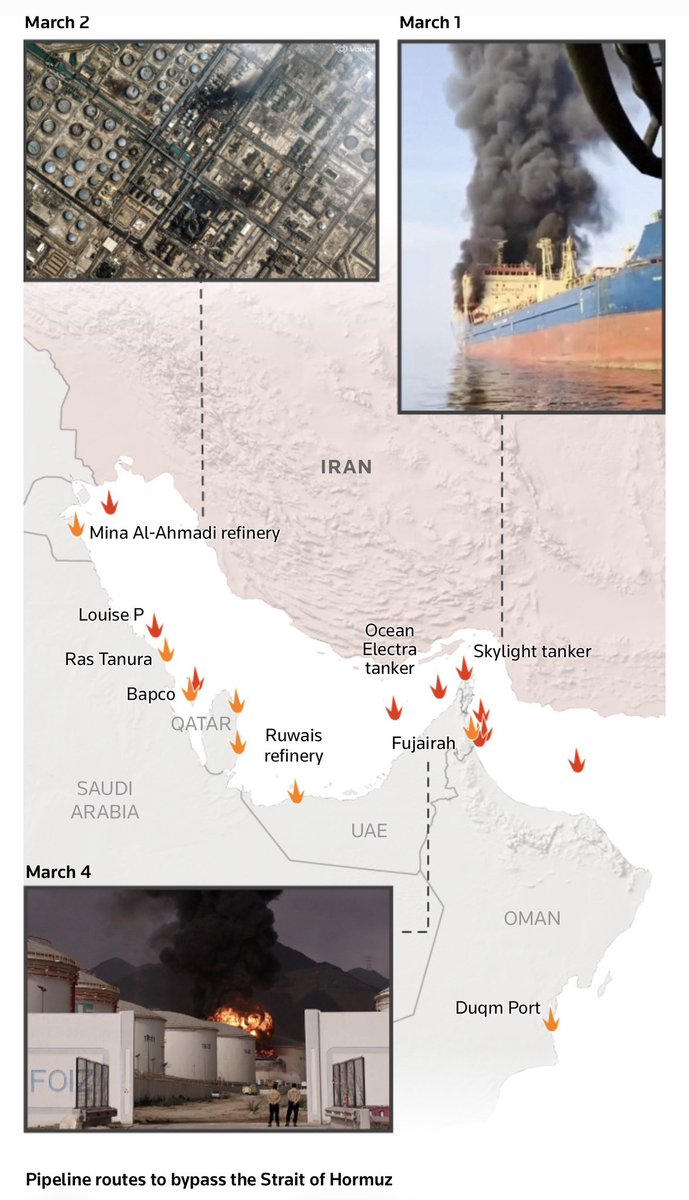
- Iraq force majeure on foreign oilfields. Strait of Hormuz risk is still very much unresolved. Brent at $108.
- Back-to-back hot PPI prints. Fed on hold and longer than expected.
- OIS markets are now pricing a 50% probability of a hike by October.
- S&P 500 has sold through its 200-day MA. JPM recently flagged 6,000–6,200 as the next meaningful support zone.
Dip-buying flows, a consistent feature of this cycle, are notably absent this time too. Quad witching has also amplified this move.
The market is now really repricing for an energy shock, sticky inflation, and the absence of a Fed backstop simultaneously.
English

$SIVE The strategic LiDAR customer has sharply raised its revenue outlook: from a cumulative $28–53M for 2026–2030 to $53–138M over the product lifecycle. SOP in Q4 2026 is confirmed, with use cases now specified as automotive and industrial/robotics.
Moody@MoodyWriter13
$SIVE thesis: proprietary InP laser tech validated by Ayar Labs & $POET; per an analyst call they’re in talks with larger players. Photonics could be acquired by a major player (e.g., $LITE) or spun/IPO’d as a value unlock. mmWave is attractive too, albeit with a smaller TAM.
English
ClearPath me-retweet

$NVO $LLY
Roche sees a big shift coming in obesity.
The company believes the market will split into segments, opening the door for new players to challenge Novo and Lilly
There won’t be a one-size-fits-all solution, as different patients will need different drugs, pricing models, and access pathways.
Roche is building a broad portfolio through acquisitions and partnerships and aims to become a top three player in the space.
Their view is that we are still “just scratching the surface” of demand.
Link to article: wsj.com/health/pharma/…
#stocks #Investing
English
ClearPath me-retweet

$qure what this debate around AMT-130 is really all about (blog): rnaitherapeutics.blogspot.com/2026/03/approv…
English

$XBI $QURE The Q2 '26 Type B meeting will define the US development path. The requirement for sham neurosurgery in a uniformly fatal disease with no approved alternatives introduces ethical and practical complexity that may support negotiation toward an adaptive or hybrid-control design, potentially compressing any Phase III timeline materially from the 3–4 year base case.
Separately, conditional approval pathways in Europe - where regulatory frameworks accommodate strong benefit-risk profiles from earlier-stage data in serious unmet-need indications - represent a credible route to commercial revenue + real-world evidence from ex-US commercial use would also substantially de-risk subsequent US re-engagement.
In short, $QURE has now has retraced to pre-data levels, effectively assigning zero value to the Phase I/II dataset.
(3/3)
English

$XBI $QURE At $15/share, $QURE is valued at roughly $400M above its cash position.
For reference, consensus peak-sales estimates for an approved disease-modifying Huntington's therapy are in the $1–$3 billion range, addressing approximately 30'000 eligible patients in the US alone with no competing approved treatment.
Even heavily risk-adjusted, the implied pipeline value appears somewhat disconnected from the quality of the underlying data.
(2/3)
English

$XBI $QURE Despite the last two weeks' turn of events, it is important to remember that AMT-130 remains the only investigational therapy to have demonstrated statistically significant disease modification in Huntington's disease.
The Phase I/II dataset - 75% slowing on cUHDRS at 36 months (p=0.003), significant TFC preservation, sustained NfL suppression below baseline, clean safety has not been challenged on its merits.
The FDA's position concerns the adequacy of the external control methodology, not the clinical signal. This is an important distinction.
(1/3)
English
ClearPath me-retweet

$XBI $EVMN Evommune came public backed by top-tier healthcare investors, including RA Capital, EQT Life Sciences, Pivotal bioVenture Partners, Andera Partners and SymBiosis.
Since IPO, the company raised $172.5 million and then added a further $125 million private placement after positive Phase 2a data, extending their runway into 2028 so balance sheet is strong.
This is critical for both execution and but for strategic value. In immunology/derm, differentiated Phase 2 assets with clean early data, multiple follow-on readouts, and no financing overhang are exactly the kinds of stories larger biopharma tracks closely.
I would not make M&A the primary underwriting basis here, but $EVMN increasingly looks like the type of funded, sponsor-backed inflammatory disease platform that could attract acquisition interest if 2026 data continue to validate the biology.
(3/3)
English

$XBI $EVMN What stands out is the shape of the setup. EVO301 hit its primary endpoint in Phase 2a in AD, while management also highlighted tolerability and PK/target-engagement data supportive of quarterly dosing.
At the same time, EVO756 remains on track for additional Phase 2 readouts in CSU and AD in 2Q26 and 2H26. Importantly, Evommune is NOT a one-asset, one-event biotech, but rather a multi-readout inflammation platform with several shots on goal over the next 6-12 months.
(2/3)
English

$XBI $EVMN is undoubtedly one of the more interesting inflammatory-disease biotech setups in small/mid-cap biotech today.
Evommune is only months post-IPO, yet has already delivered positive Phase 2a proof-of-concept data for EVO301 in moderate-to-severe atopic dermatitis, strengthened the balance sheet with a $125m private placement, and guided runway into 2028.
Fresh clinical validation + balance sheet strength is a powerful combination in the current market.
(1/3)
English
ClearPath me-retweet
ClearPath me-retweet

@RNAiAnalyst Agree, also sitting close to cash at current market cap
English

@bioinvesting5 @MaartenDik $QURE's post Prasad de-risking is indeed now at least partially priced in.
English

@MaartenDik I’d only buy $QURE here if you realize that it can easily go back to $7 on you. The FDA fight is just starting.
English

$QURE RBC: “However, Vinay Prasad is now leaving the FDA. On one side, this is a positive for QURE in our view as it is not inconceivable that the FDA reverts to its prior stance (accelerated approval based on comparison to natural history). However, we also note that the FDA Commissioner has also criticized QURE and while Dr. Prasad is leaving, Dr. Makary is not.”
I’d say not YET.
RBC thinks there’s 50% chance that the drug ultimately gets approved.
Phoenix Biotech@BiotechAnalysst
RBC $QURE Upgrading to OP PT $35 Open Up A More Balanced Discussion On HD
English