固定されたツイート

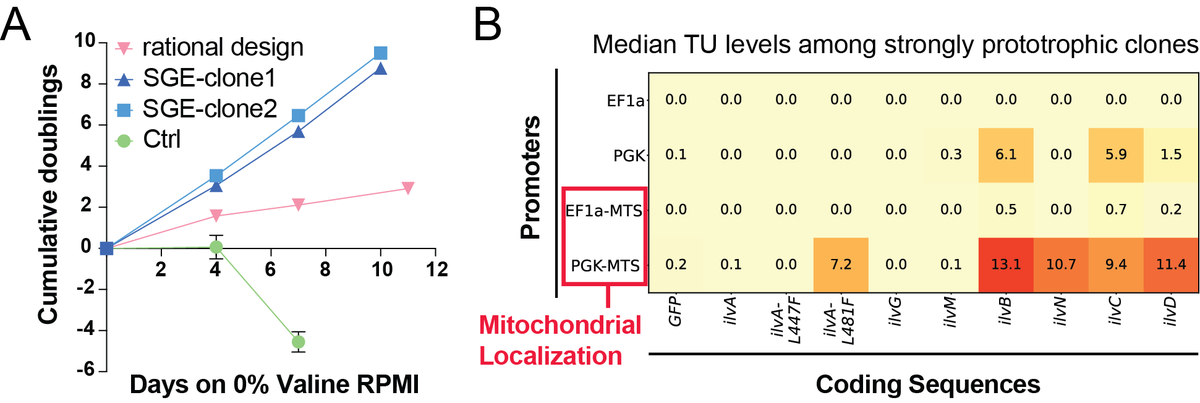
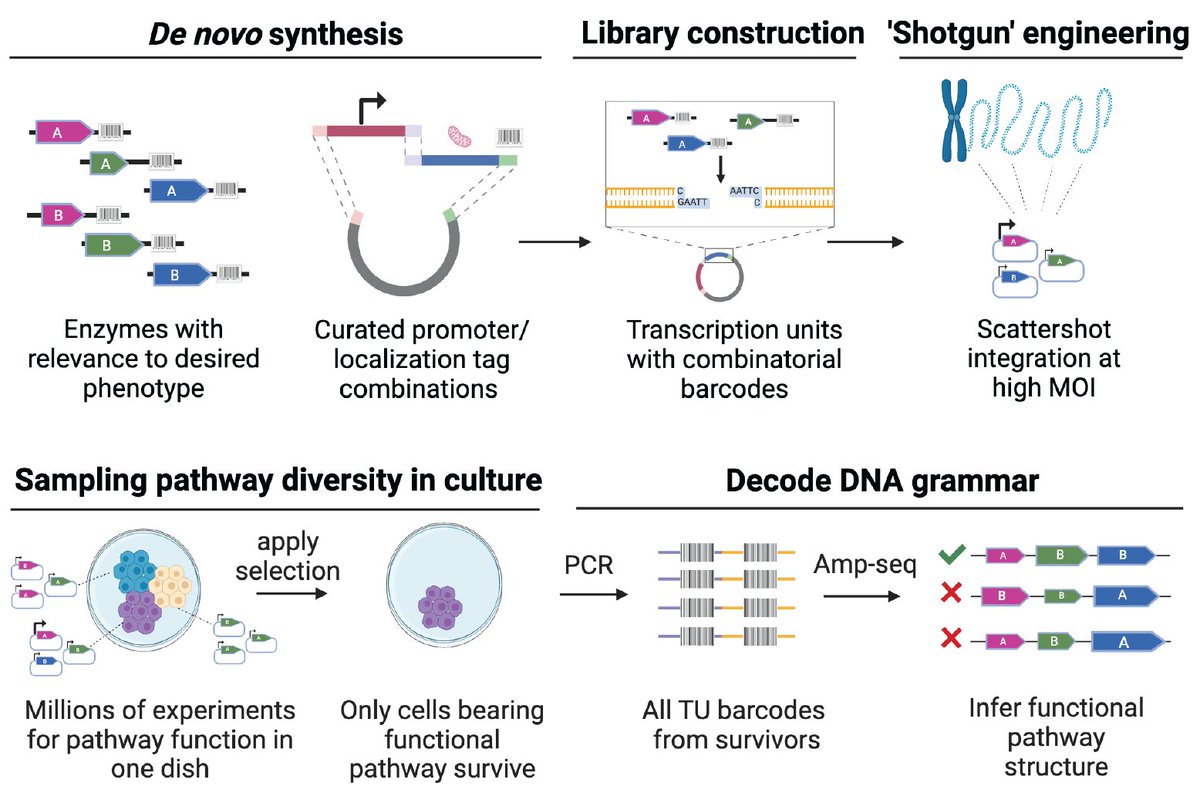
Now out in @ScienceMagazine we present 'Genome-shuffle-seq': a method to shuffle mammalian genomes and characterize the impact of structural variants (SVs) with single-cell resolution in one experiment.
science.org/doi/10.1126/sc…
English