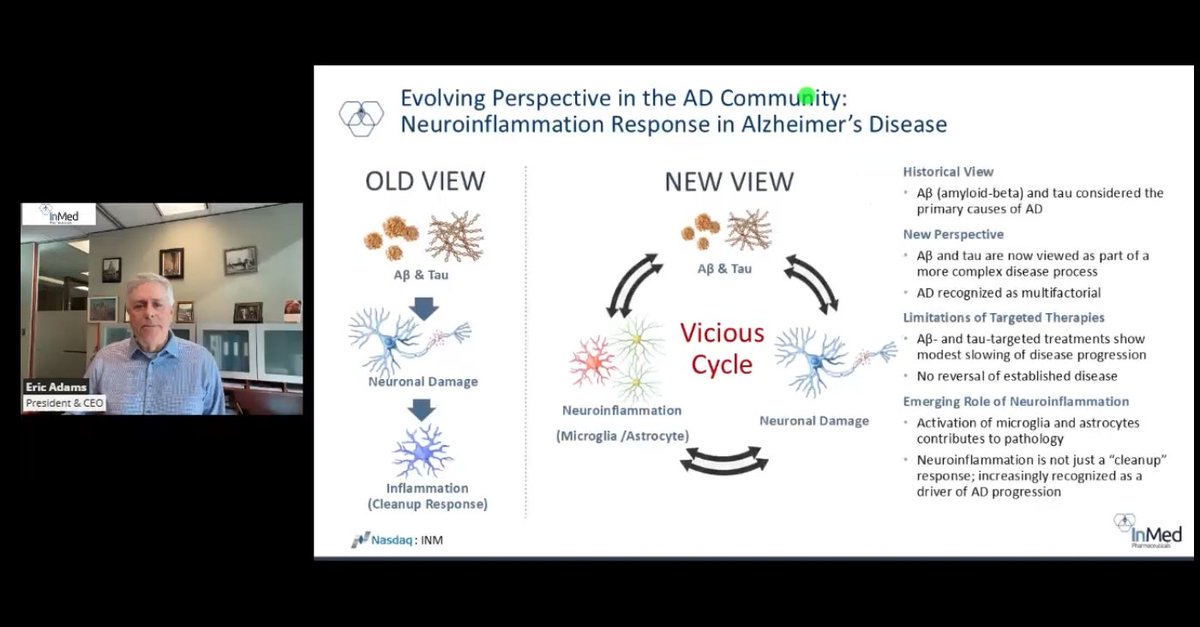
InMed Pharmaceuticals Announces Positive Data from Human Brain Organoid Neuroinflammation Models Supporting the INM-901 Alzheimer’s Disease Program. See full press release: ow.ly/J5tx50Yxo1u

English
InMed Pharmaceuticals
1.2K posts

@inmedpharma
Developing small molecule compounds for the treatment of #Alzheimers, Age-related #Macular Degeneration #AMD, & #EB. Our subsidiary @BayMedica NASDAQ: $INM