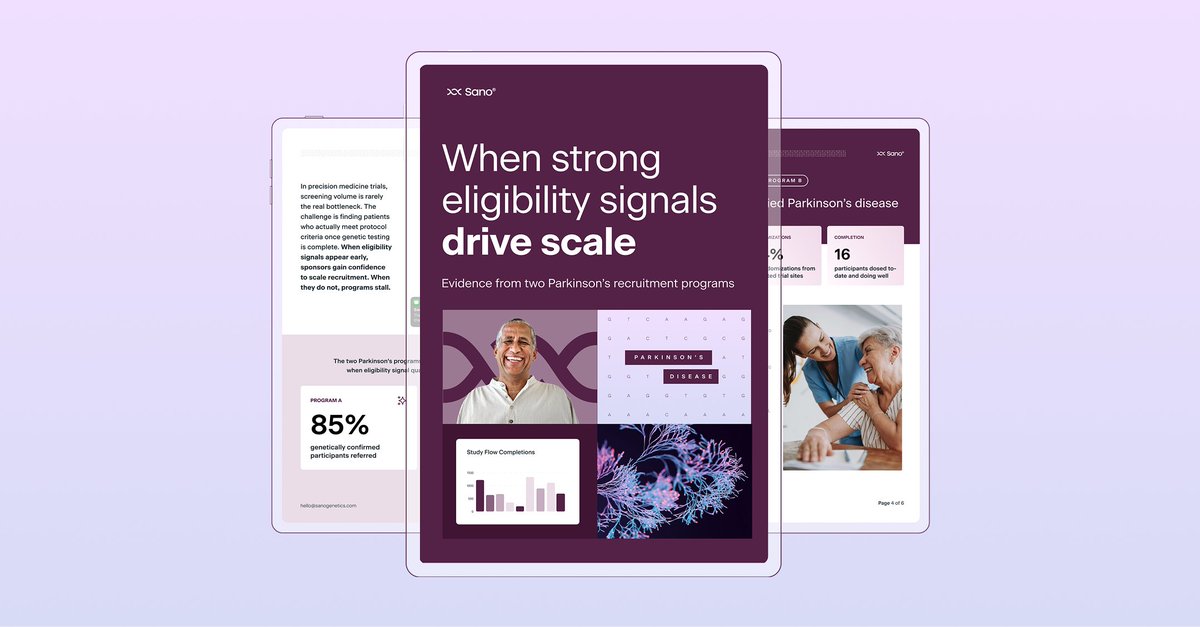
🧬 In precision trials that require genetic testing as part of the pre-screening process, the highest drop-off doesn’t happen where you might think.
Kit return rates are often strong once a participant orders a kit. The real bottleneck is earlier, between eligibility and action.
That moment determines whether interest turns into participation or disappears.
We looked at what actually drives that decision across several rare disease and precision medicine studies that we have supported. Effort, clarity, relevance, trust, and timing all shape whether participants move forward.
Small improvements at this stage can translate into faster enrollment, lower costs, and more complete datasets.
📖 Read the full breakdown here: eu1.hubs.ly/H0t1P0r0
#GeneticTesting #PrecisionMedicine #GeneTherapy #PatientEngagement

English