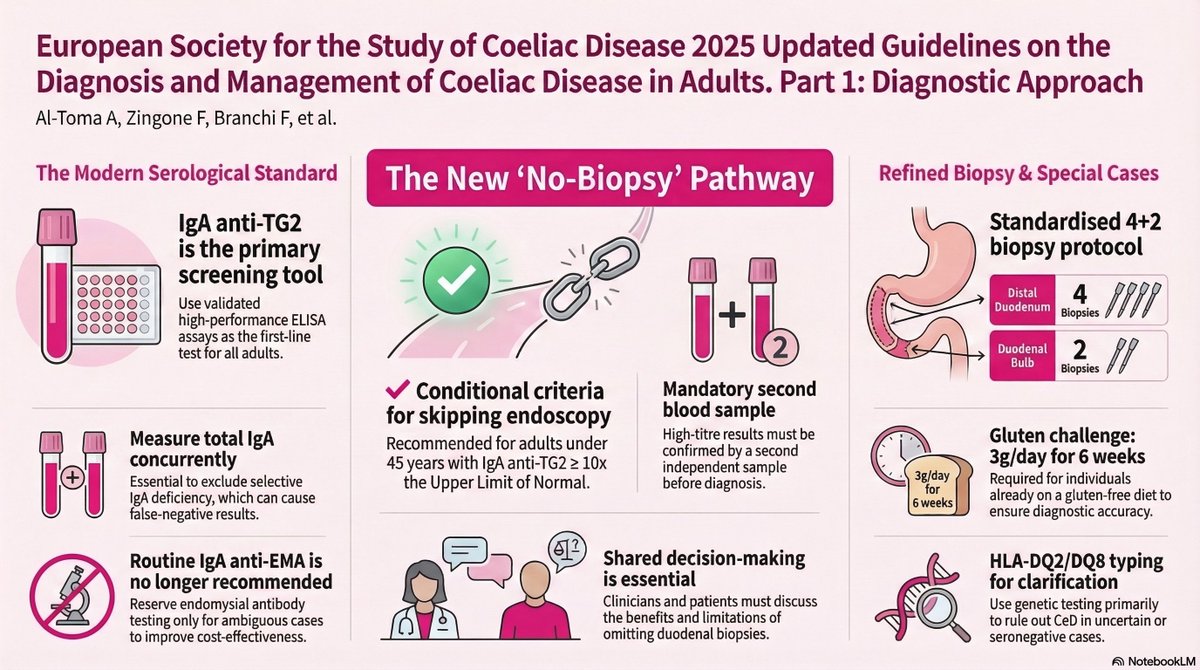
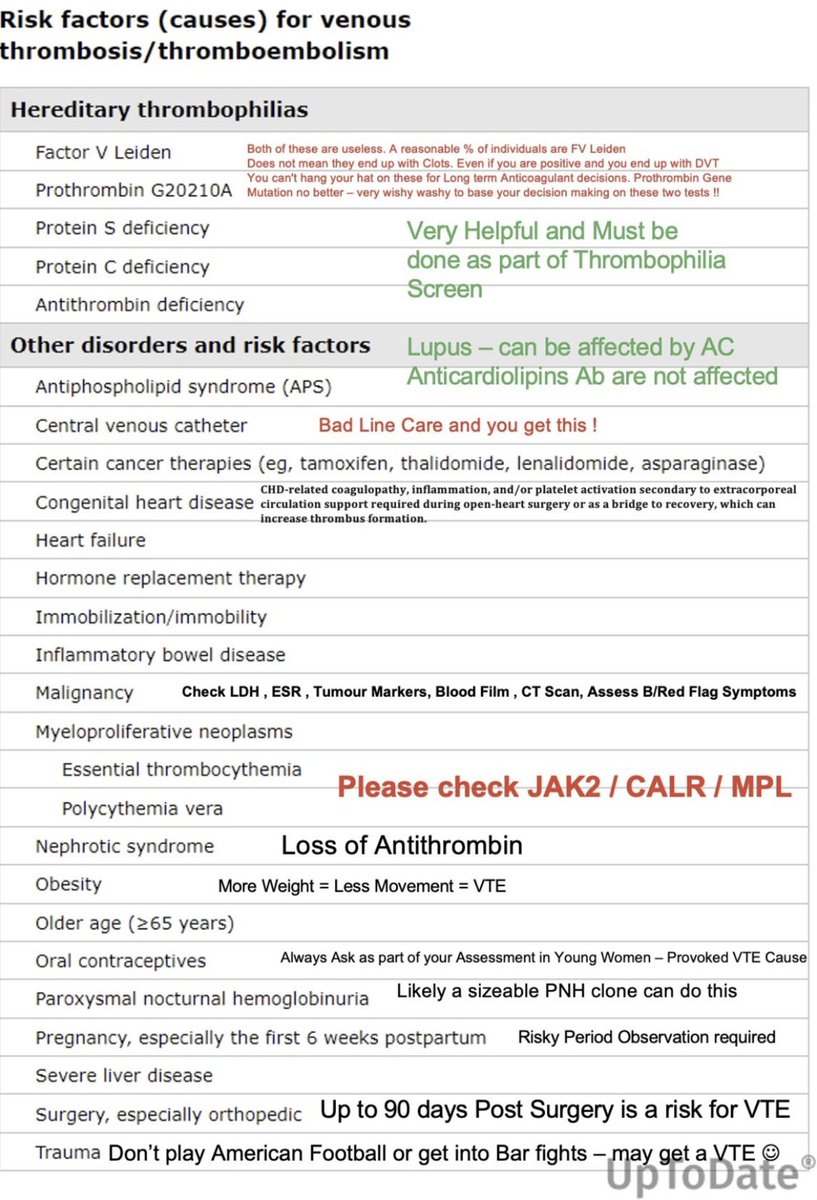
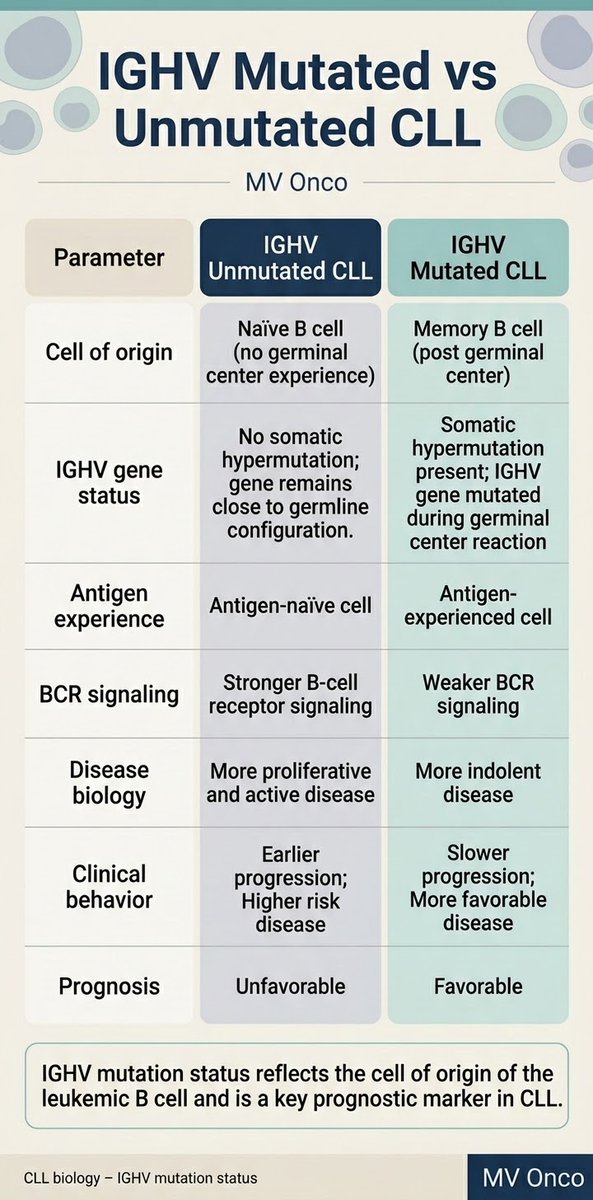
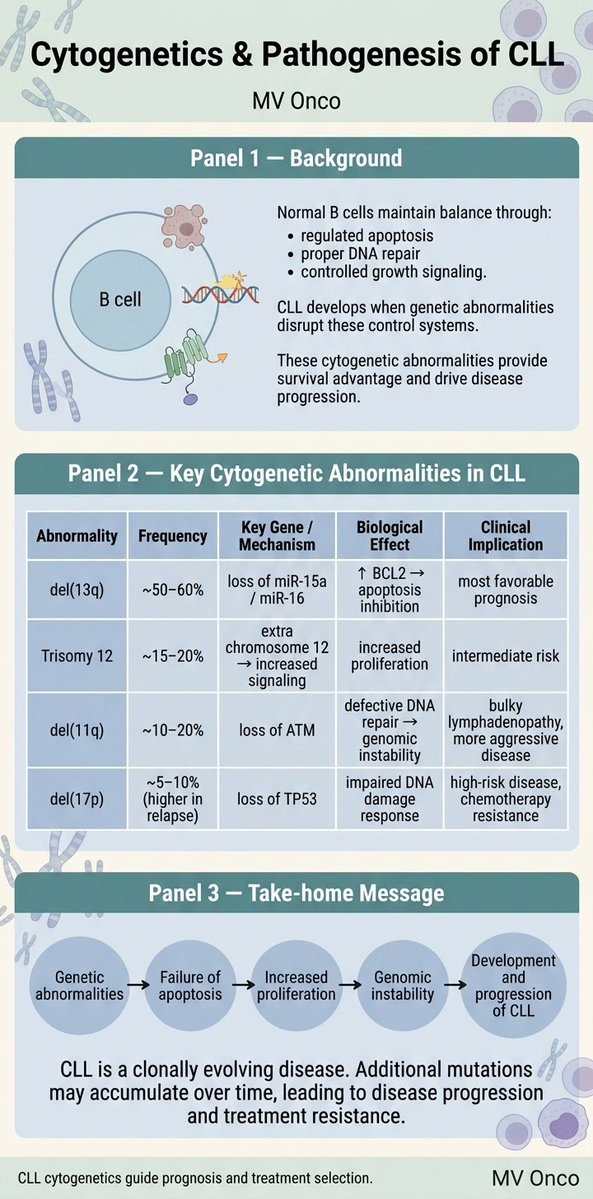
Ankur Singh, DNB (Path)
7.5K posts

@ankspath
Pathologist with interests in #molecularpath #GIpath and #oncopath #DNBPathology Here on Twitter for learning and sharing T/RT not medical advice



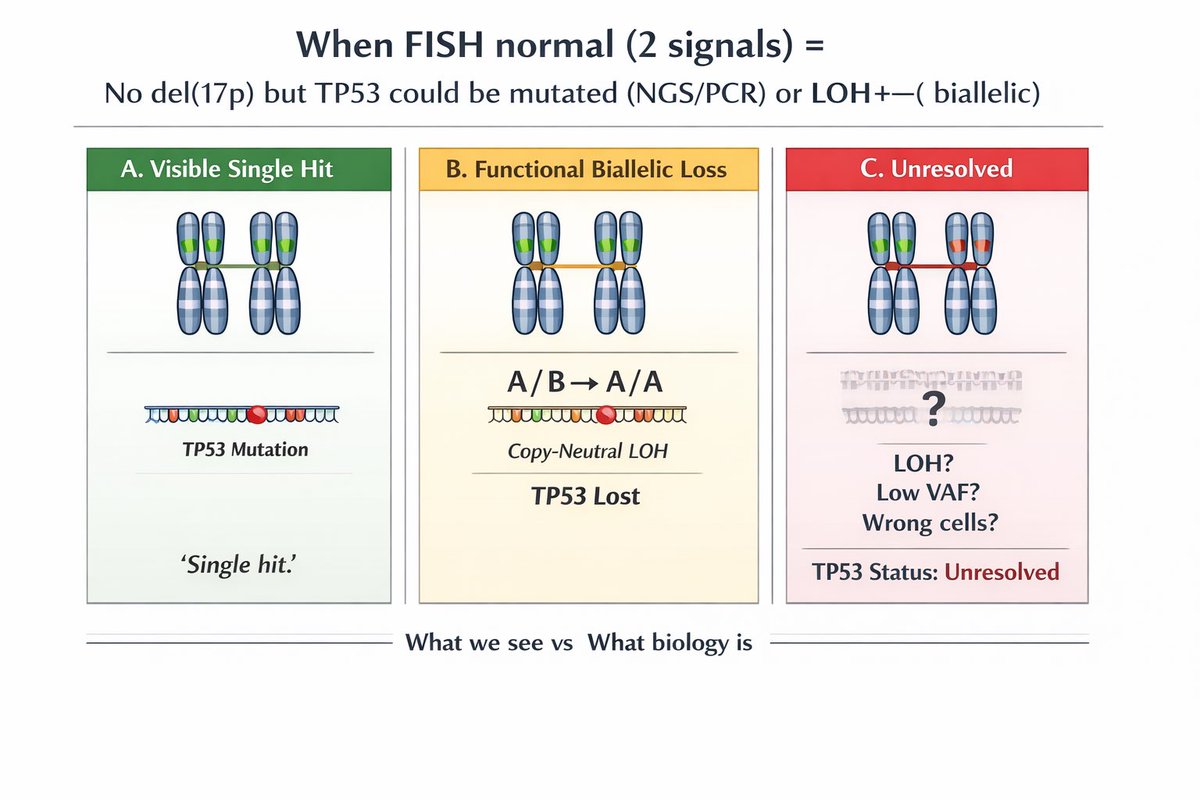
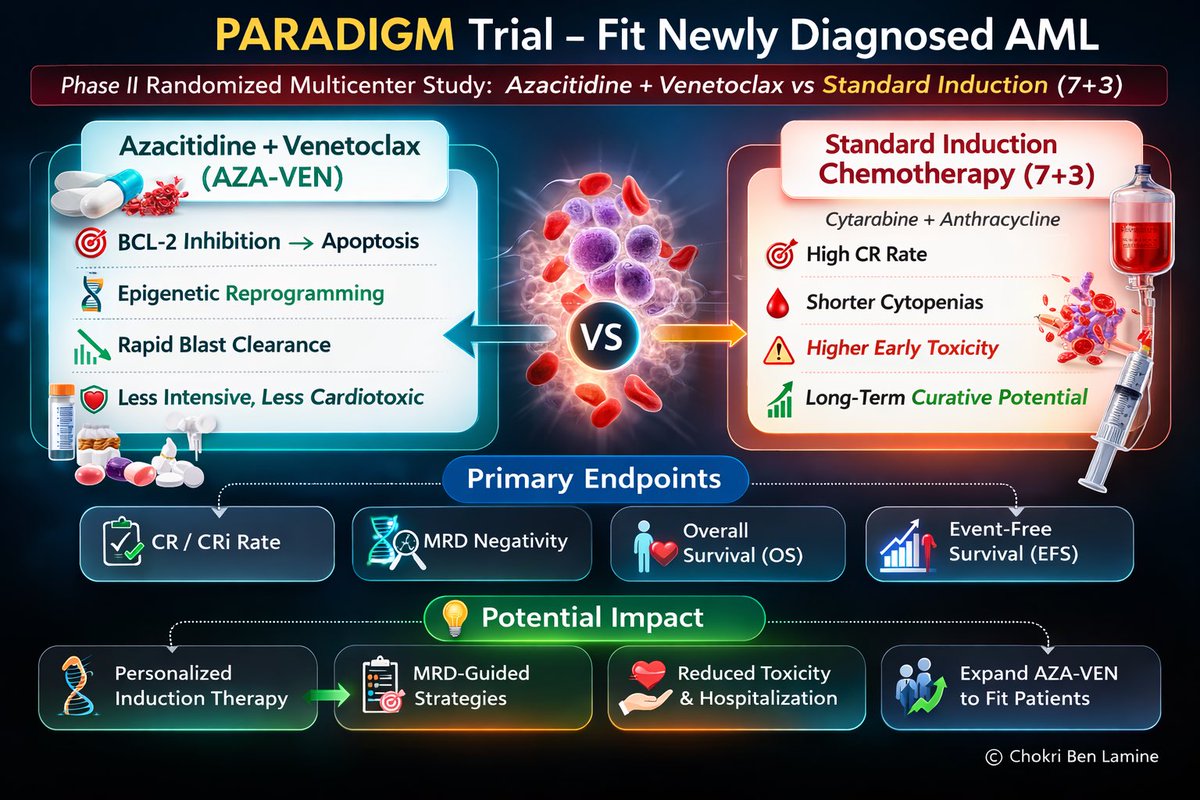
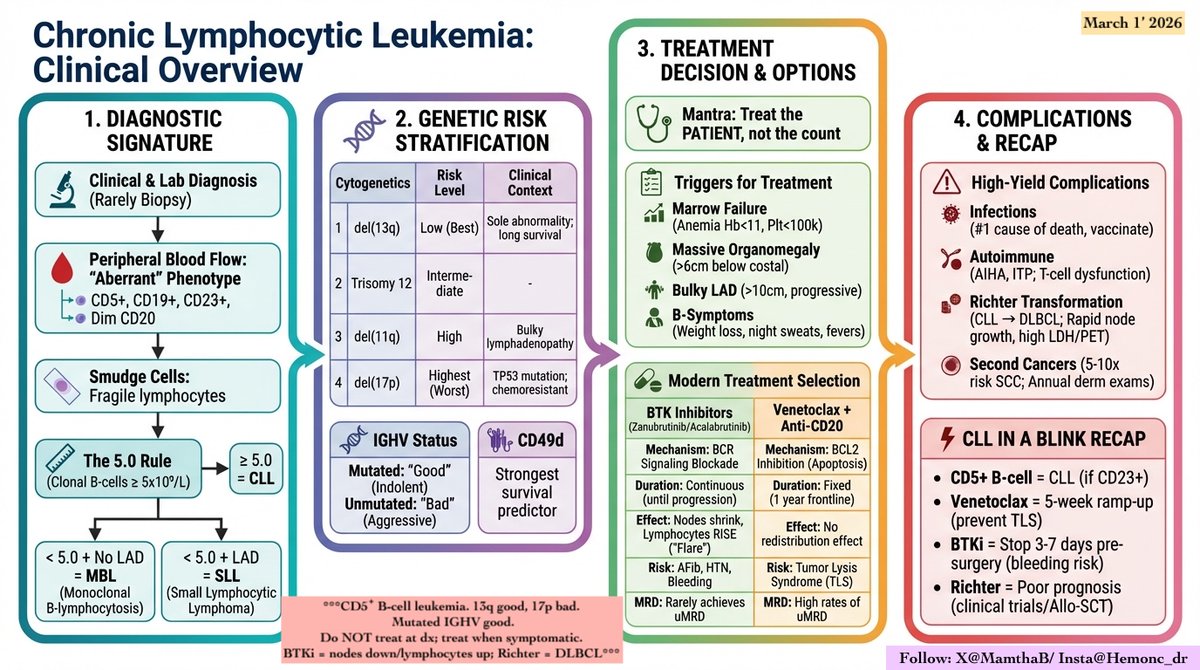
Biallelic TP53 loss is NOT included in IMS/IMWG 2024 risk stratification. Yet most would agree: this is ULTRA –High RISK myeloma. WHY ? Because we only include what we can measure. FISH detects del(17p) → only one hit NGS detects mutation → may suggest double hit but misses many VAF >60% is suggestive, not confirmatory CMA/SNP is key: • Detects copy-neutral LOH • Mutant allele duplicates, wild-type lost • True biallelic inactivation We measure deletions… BUT biology operates through loss of function. Bottom line: FISH + NGS + CMA are needed for full TP53 assessment Until then, we are under-calling ultra–high risk myeloma. #myeloma #MultipleMyeloma #Hemetwitter #smmm