Stay ahead of FDA shifts that could impact your strategy. vist.ly/4yi8m
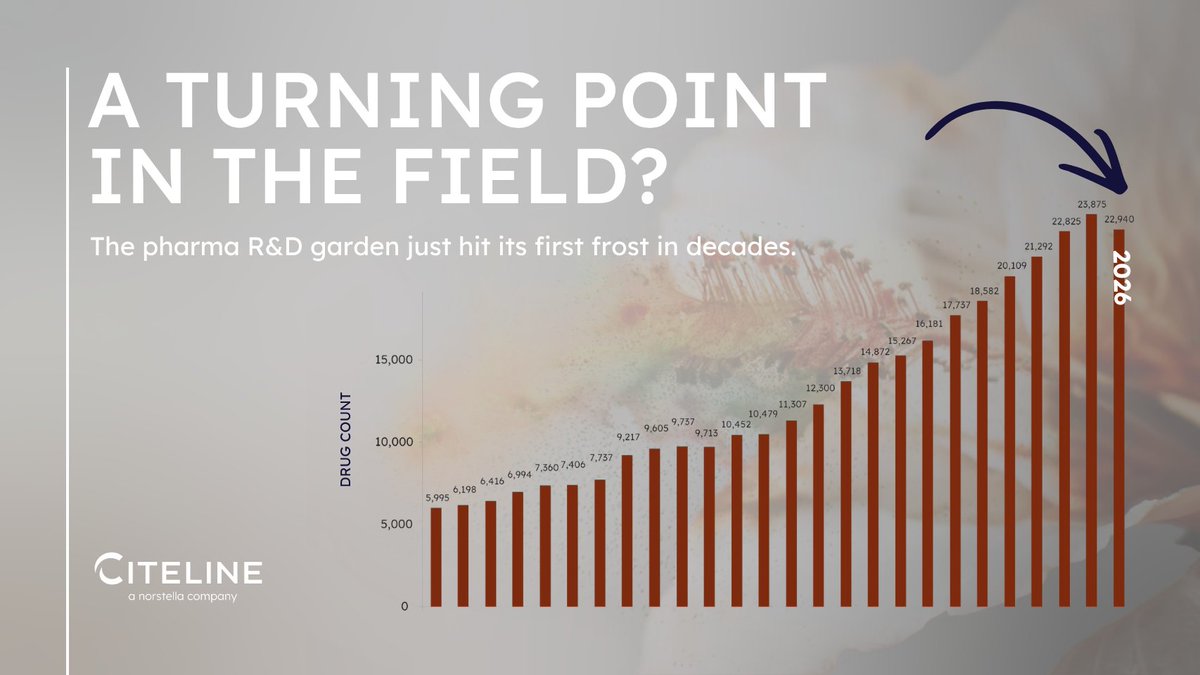
The landscape of drug approvals is evolving under new leadership and changing regulations.
Save time and focus on what matters—accelerating life-saving therapies. Download our latest eBook now!

English