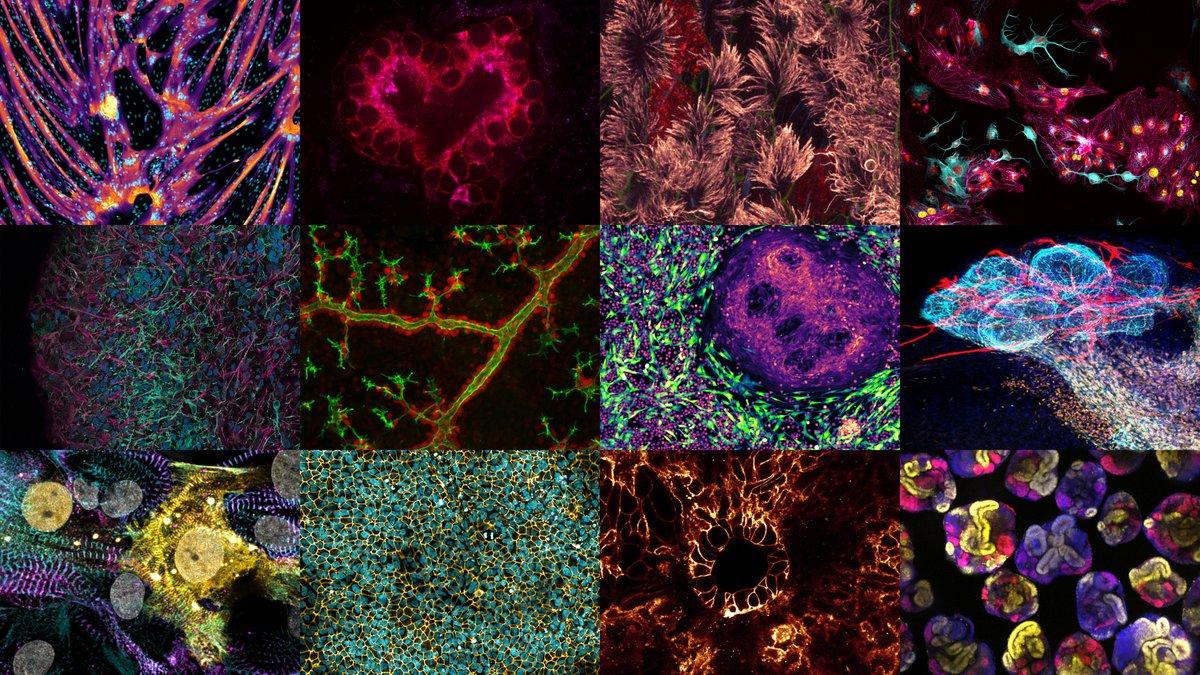
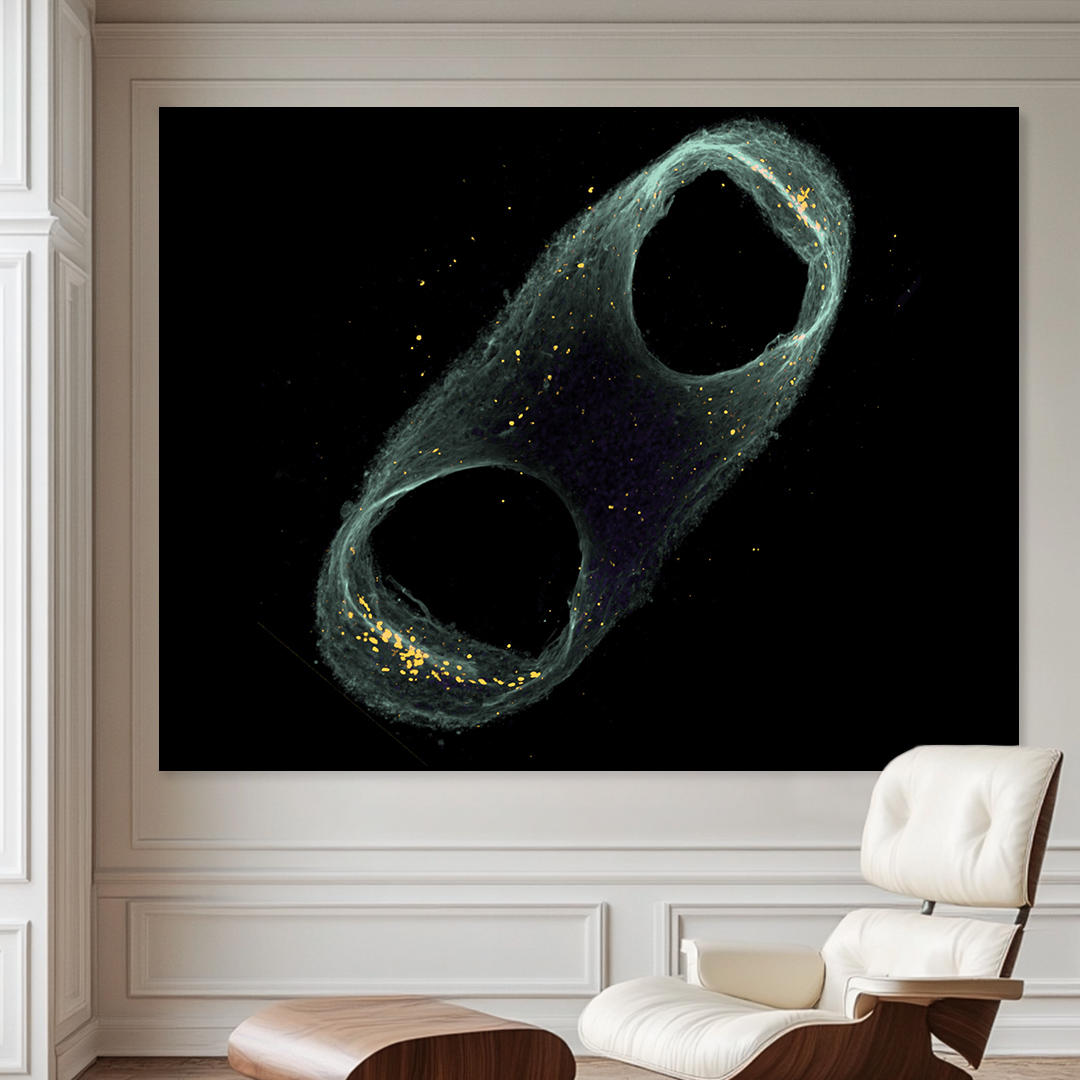
Our #ARTxSCIENCE Exhibition illuminates the night in the hall of the @DanskIndustri @DILifescience right on the Town Hall Square of Copenhagen.
During the day you may go and experience the exhibition where it greets you right at the entrance.
@UCPH_Research @novonordiskfond
English