The Science of Parkinson's
24.7K posts


The Science of Parkinson's
@ScienceofPD
Personal account of the Director of Research at @CureParkinsonsT - All views my own - Kiwi - Kia kaha - 侘寂
London, England Entrou em Ocak 2016
249 Seguindo11K Seguidores

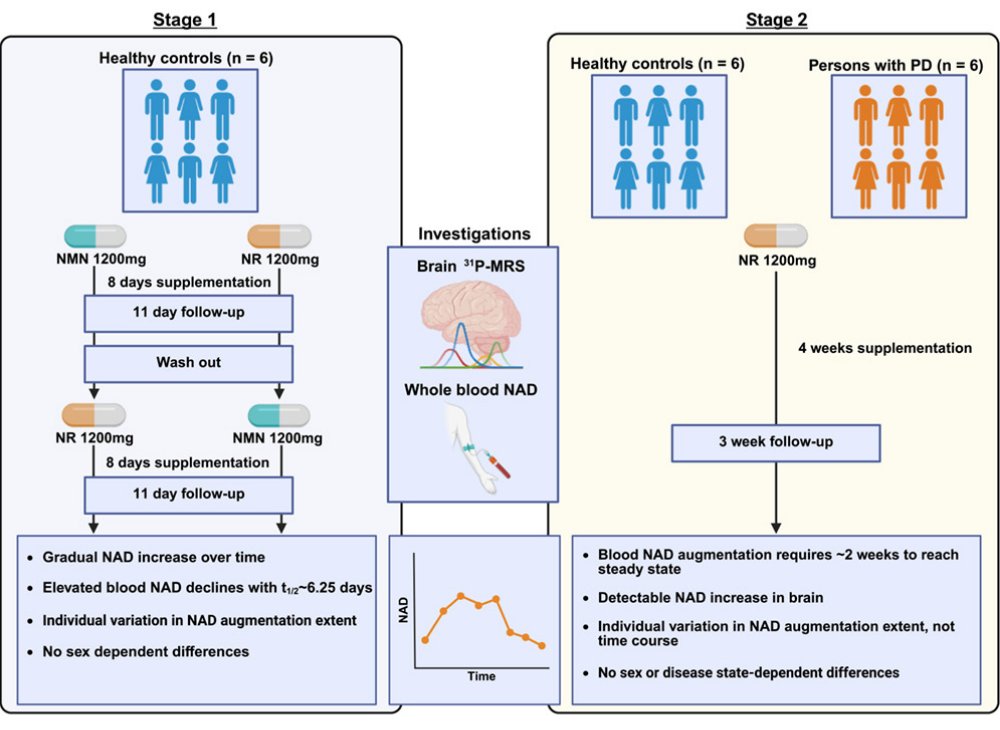
Researchers report the results of a phase I pharmacokinetic trial assessing systemic & cerebral responses in healthy individuals (n = 6) & people with #Parkinsons (n = 6) receiving 1,200 mg/day nicotinamide riboside or nicotinamide mononucleotide
cell.com/iscience/fullt…
English

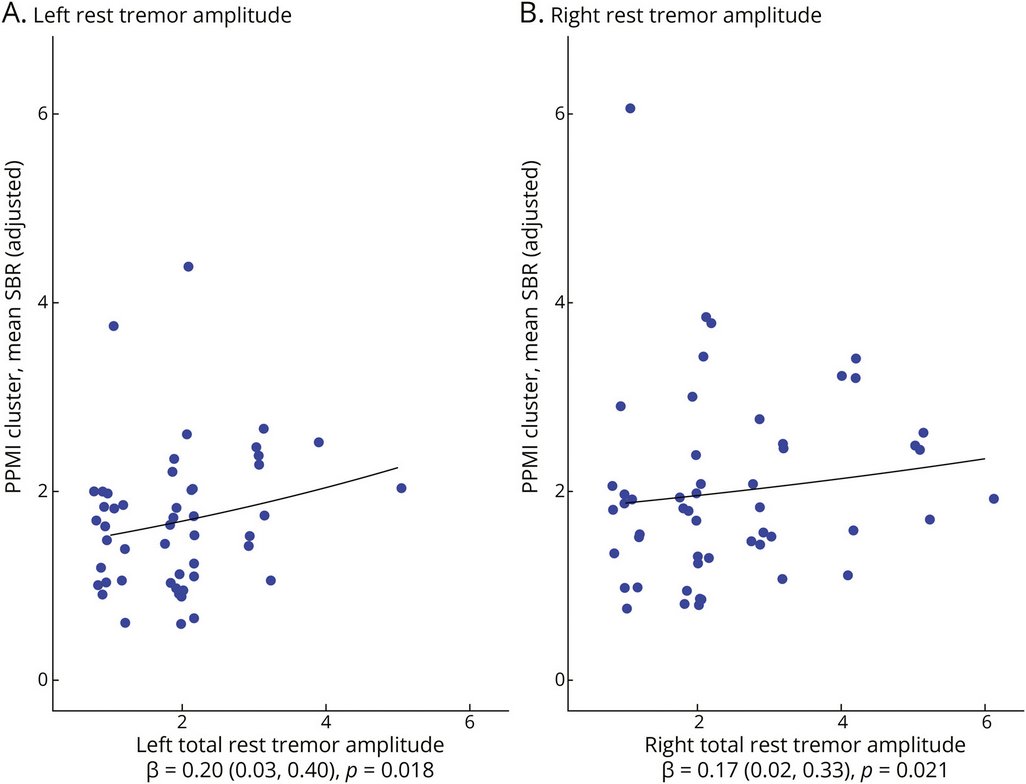
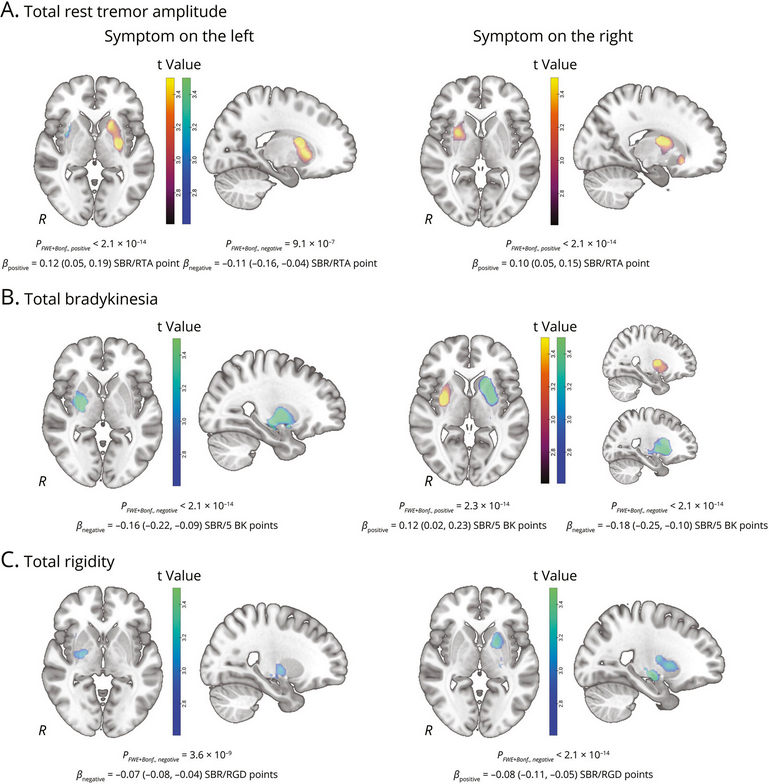
New research confirms the positive association between rest tremor amplitude & ipsilateral striatal DAT binding in #Parkinsons patients, but non-PD groups were diagnostically heterogeneous, limiting conclusions about disease specificity
neurology.org/doi/10.1212/WN…
English

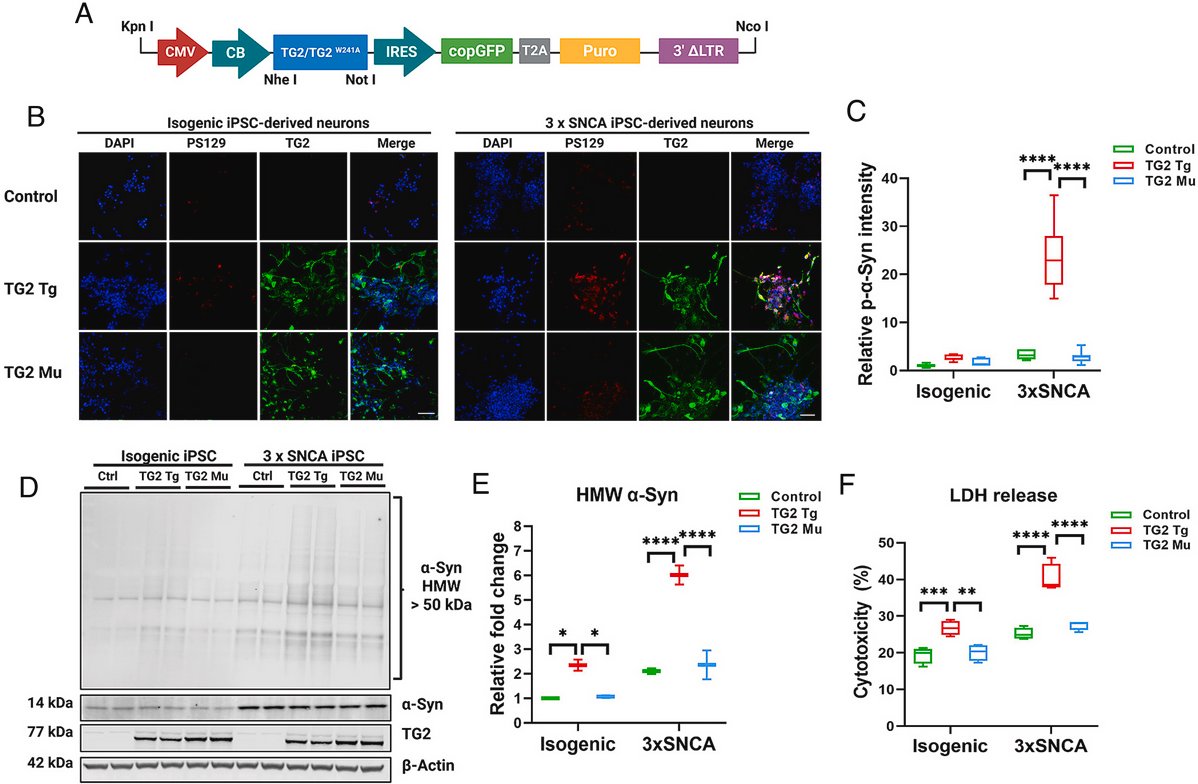
Using transgenic mice & #Parkinsons patient IPS cells, researchers report on a single point mutation in Transglutaminase 2 that abolishes only its cross-linking activity, rendering it incapable of causing α-Synuclein aggregation & neurodegeneration
pnas.org/doi/10.1073/pn…
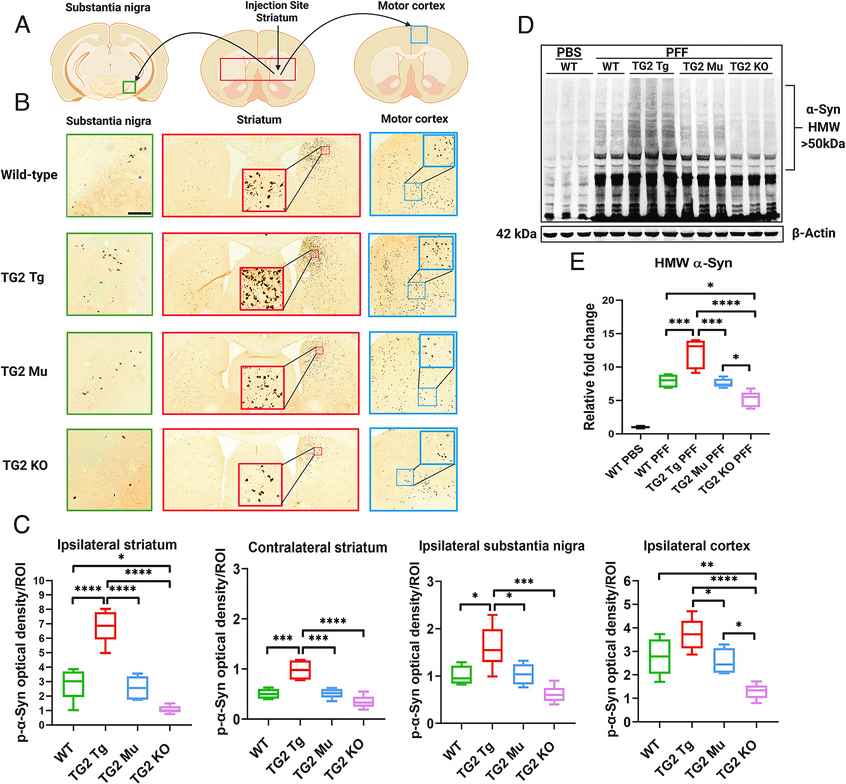
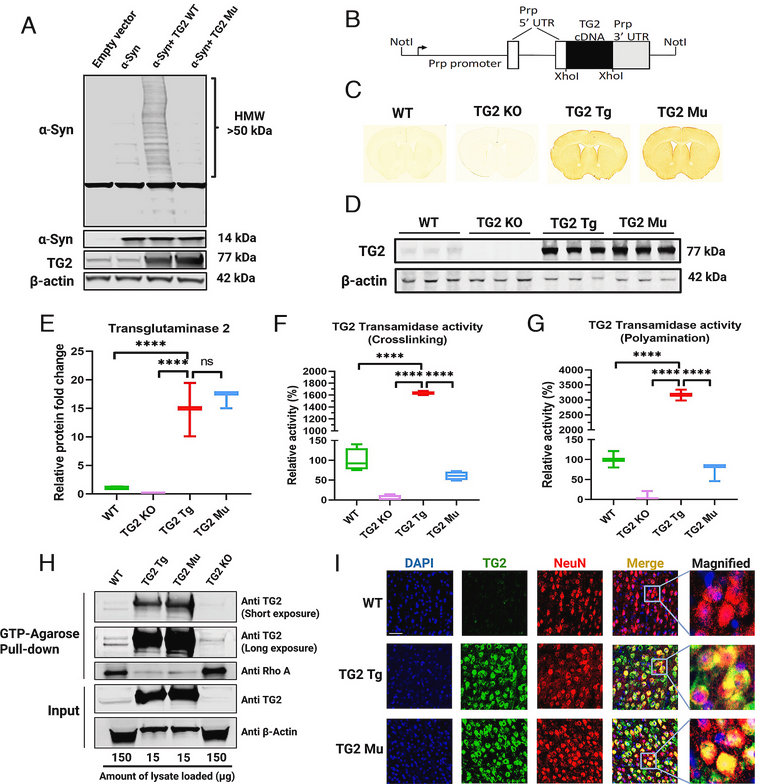
English

Mitochondria in a capsule?
Researchers demonstrate mitochondrial transplantation (via erythrocyte membrane encapsulation), rescuing mitochondrial defects in patient-derived cells & #Parkinsons mouse model
cell.com/cell/abstract/…
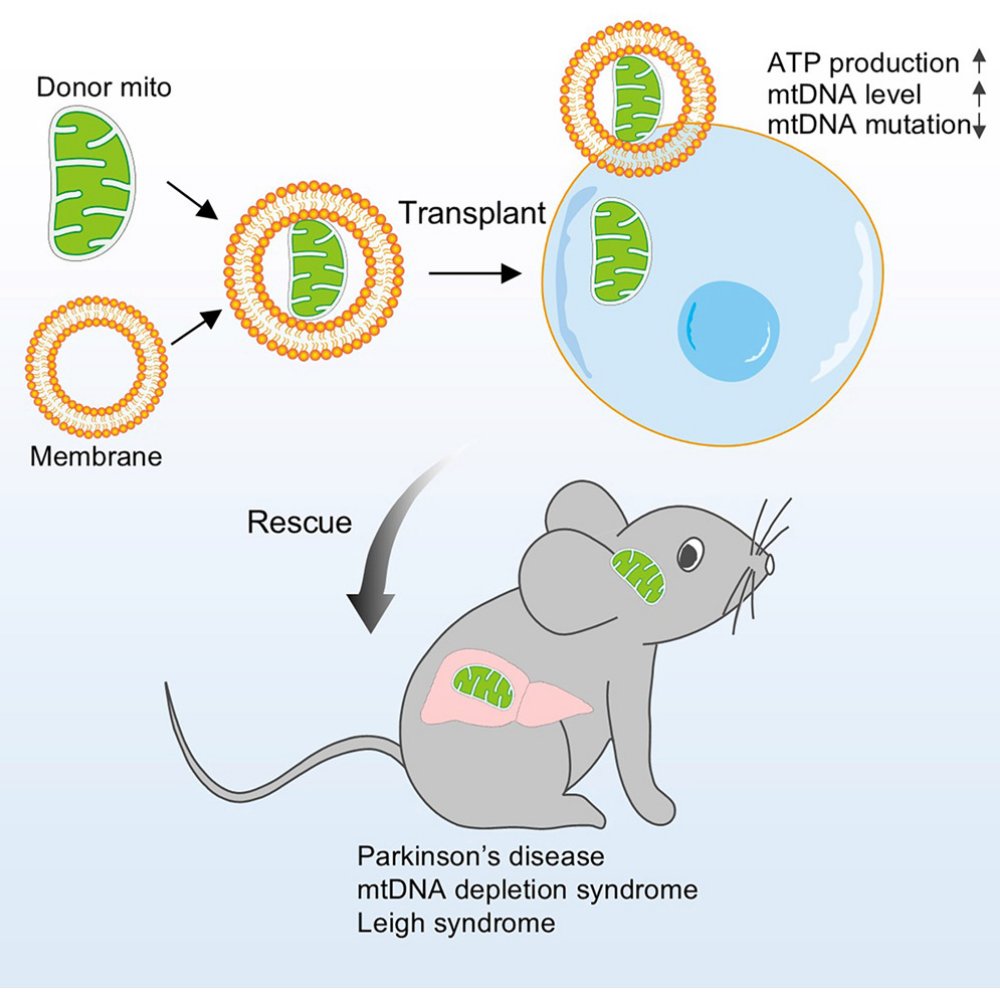
Català


I also liked the @InnoMedica poster presenting their NEON clinical trial results for Talineuren in people with #Parkinsons
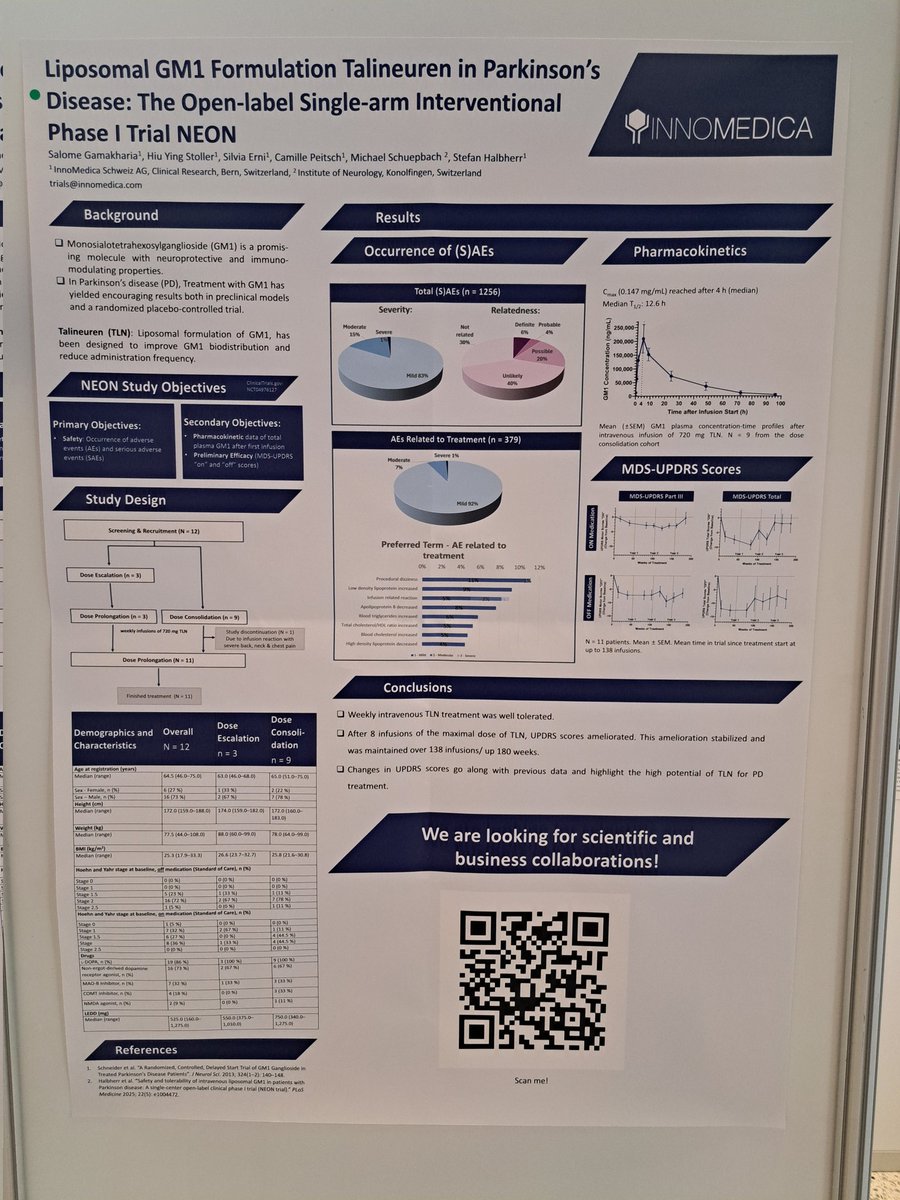
English

Best poster of the #adpd2026 meeting so far - presenting a global alliance for #Parkinsons [clinical trial] platforms
(I may be a little biased - @CureParkinsonsT is involved)

English
The Science of Parkinson's retweetou

Neurodegenerative diseases at the single-cell level, a powerful tool to differentiate and better understand jci.org/articles/view/… @jclinicalinvest
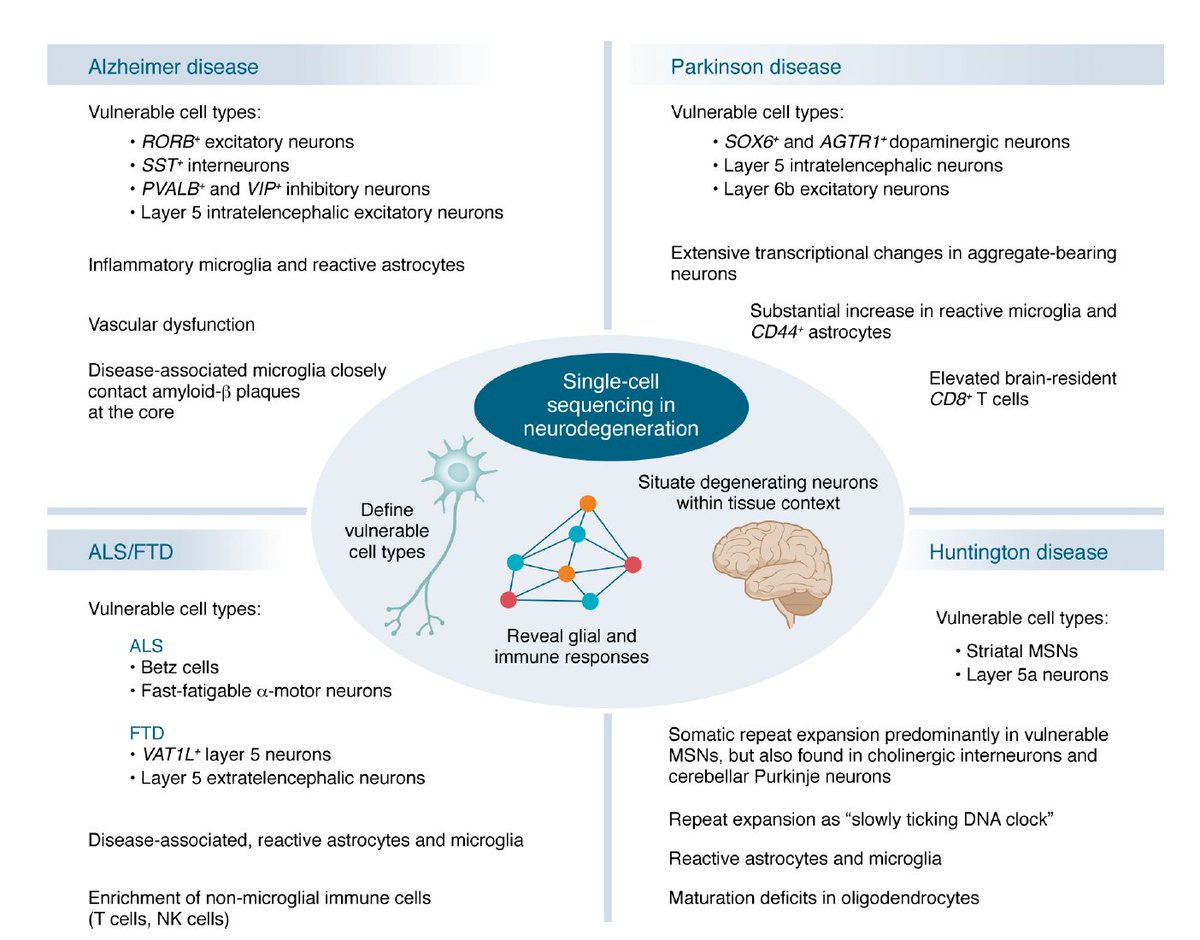
English

@BrundinPatrik Opps, thanks for the clarification Patrick!
English

@ScienceofPD I think it was F-DOPA PET.
Indeed, the pre- and post graft scans were close to identical with the signals (striatal to occipital ratios) being 0.703 and 0.720 before and 12 mo after grafting, respectively.
English

Aspen Neuroscience @AspenNeuro present 12-month data from their Phase 1/2a ASPIRO clinical trial (safety, tolerability, & efficacy data of Sasineprocel (ANPD001 - transplanted autologous iPS cell‑derived dopaminergic neuron precursors) in #Parkinsons
aspenneuroscience.com/aspen-neurosci…

English

Arvinas @ArvinasInc presented positive Phase 1 data for their LRRK2 PROTAC, ARV-102 at the #ADPD2026 meeting today; They see >50% LRRK2 degradation in the CSF of people with #Parkinsons treated for 28 days; Safe & well tolerated; Half-life=68 hours
ir.arvinas.com/news-releases/…

English
The Science of Parkinson's retweetou

#NEWS: Arvinas announces Positive Phase 1 data for its #PROTAC LRRK2 Degrader presented at the 2026 International Conference on Alzheimer’s and Parkinson’s Diseases and Related Neurological Disorders (AD/PD™ 2026).
Learn more here: bit.ly/4rJcqMH

English

Gain Therapeutics @GainThera presented really interesting data at the #ADPD2026 meeting today: Phase 1b trial results for their allosteric glucocerebrosidase (GCase) modulator GT-02287 in 19 people with #Parkinsons; Safe & well tolerated...
gaintherapeutics.com/newsroom/press…

English

The results of the Takeda Pharma Phase 2 trial of TAK-341 (monoclonal antibody for a-synuclein) in Multiple systems atrophy were presented today at the #ADPD2026 meeting - treatment was safe, but no difference between groups (programme terminated)

English

Encouraging data from Herantis Pharma @HerantisPharma today at the #adpd2026 meeting - Phase 1b data on their small CDNF-like peptide HER-096; Results met all primary & secondary endpoints; Safe & well tolerated; Good biomarker analyse; Phase 2 in 2026
herantis.com/news-events/re…

English

Encouraging data presented by Roche @roche today at the #adpd2026 meeting - Phase 1b safety, pharmacokinetics, & pharmacodynamics of their NLRP3 inhibitor selnoflast in people with #Parkinsons; Positive data (safe with peripheral & central effects); No mention of next steps

English

Data was presented at the #adpd2026 meeting. 8 treated patients – 4 on low dose & 4 on high dose; Parieto-occipital approach for delivery; Mean good ON time (+2.1 hours in low dose, +2.4 hours in high dose), mean MDS-UPDRS III OFF scores (-15.5 points in LD, -13.5 points in HD)
English
