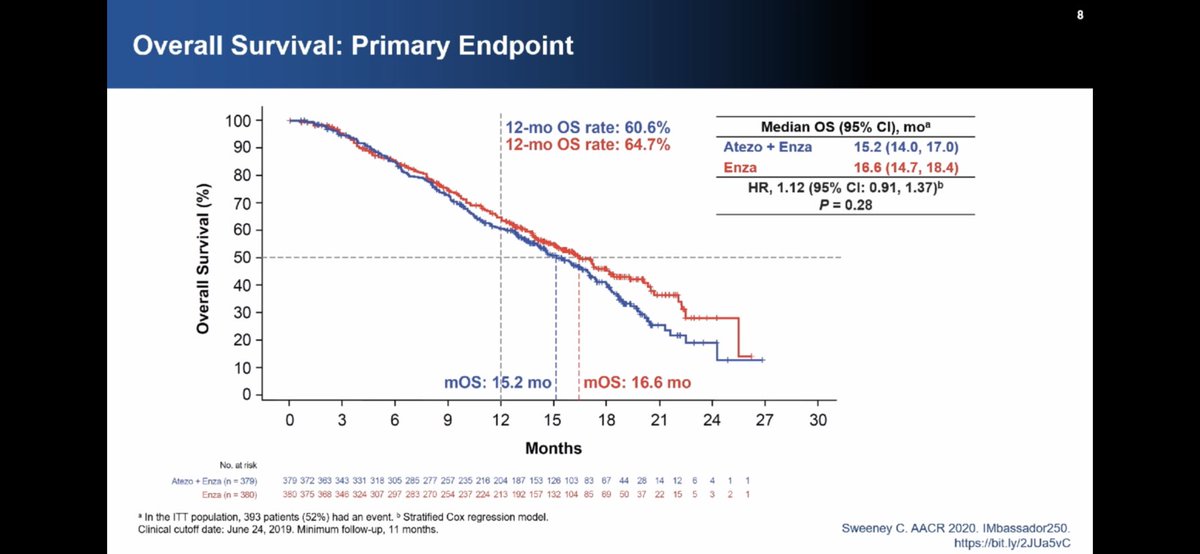
Naka-pin na Tweet

Merck's oral MET inhibitor, #Tepotinib, receives FDA BTD status for the treatment of metastatic #NSCLC patients with specific MET alterations. Although only 3-5% of patients have such alterations, such targeted approach could be both clinically and commercially successful.
English