AT
845 posts



Putting criticism about timing and data presentation aside, $ERAS' ERAS-0015 seems very similar to $RVMD's daraxonrasib imo. ERAS-0015 is more potent which explains the lower exposure required for efficacy (and associated with on target toxicity of course). It might have a slight advantage on GI side effects (to be seen) but other than that it is likely to have a similar therapeutic index. If we compare just the PDAC data sets, $ERAS reported 4 PRs in 21 patients (~19% ORR) in the US at relevant doses which is lower than dara's 35% ORR but that gap will likely close with more follow up as $ERAS has multiple near PRs at the first scan than can convert in the future (They need 3 such conversions to reach 33% ORR). The limited follow up probably has a confounding effect on safety as well (longer treatment typically leads to more side effects) which should take $ERAS to a similar dose interruption/reduction rate in the future. Gr5 pneumonitis case requires attention but it could be an isolated case. Is $ERAS cheap at $3B? Not sure but for pharmas who cannot afford or simply won't get $RVMD this is the closest fast follower program that could be P3 ready later this year. *no position in stocks mentioned *

Putting criticism about timing and data presentation aside, $ERAS' ERAS-0015 seems very similar to $RVMD's daraxonrasib imo. ERAS-0015 is more potent which explains the lower exposure required for efficacy (and associated with on target toxicity of course). It might have a slight advantage on GI side effects (to be seen) but other than that it is likely to have a similar therapeutic index. If we compare just the PDAC data sets, $ERAS reported 4 PRs in 21 patients (~19% ORR) in the US at relevant doses which is lower than dara's 35% ORR but that gap will likely close with more follow up as $ERAS has multiple near PRs at the first scan than can convert in the future (They need 3 such conversions to reach 33% ORR). The limited follow up probably has a confounding effect on safety as well (longer treatment typically leads to more side effects) which should take $ERAS to a similar dose interruption/reduction rate in the future. Gr5 pneumonitis case requires attention but it could be an isolated case. Is $ERAS cheap at $3B? Not sure but for pharmas who cannot afford or simply won't get $RVMD this is the closest fast follower program that could be P3 ready later this year. *no position in stocks mentioned *


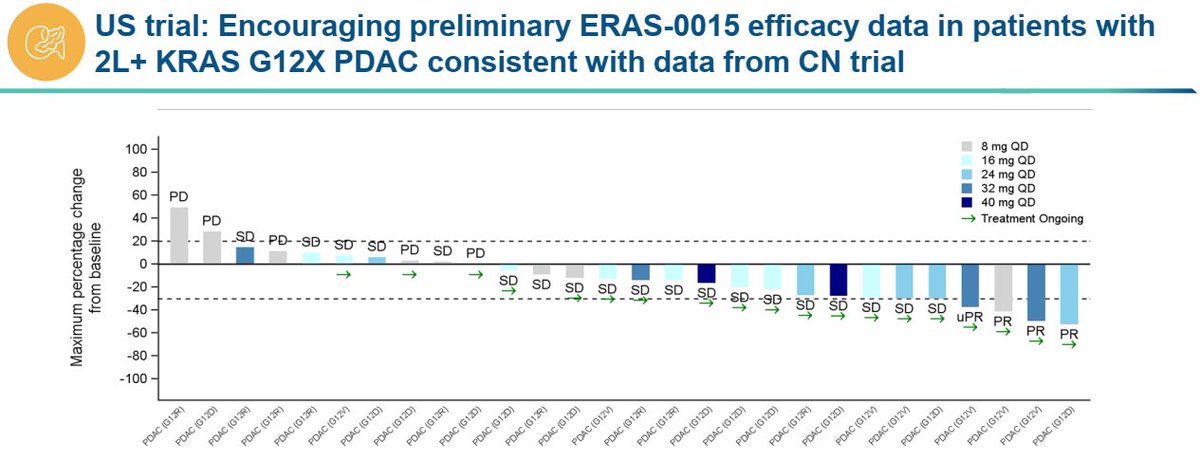

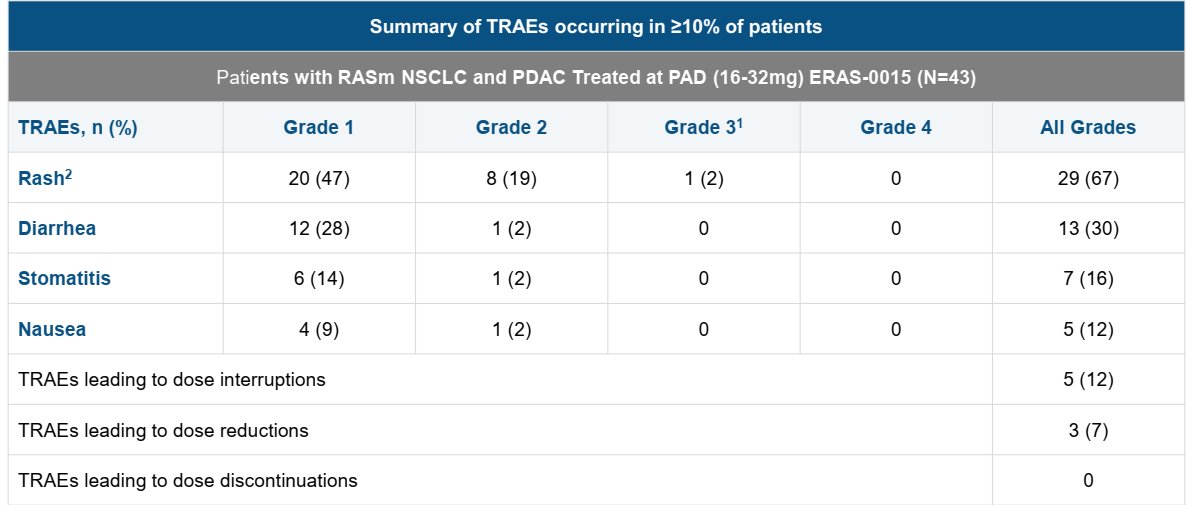

have to say really not a great look to not disclose a death in the PR and only mention it in slides. If there really is no correlation, what's the issue

$ERAS pan-Ras data update this afternoon after market close. also, $RVMD sent Erasca a letter claiming ERAS-0015 violates certain RevMed patents. sec.gov/Archives/edgar…

$IDYA PFS curves from Optimum-02


$idya easy choice for patients, docs and payors. hr = 0.42💥💊 4x fewer g3+ traes.


@anthonystaj I'm not overly worried about this. Prophylaxis should help. We've seen in trials as recent as HERIZON-GEA-01 that you can bring gr3 diarrhea way down. That one still had 24.5% gr3 diarrhea with a 11.9% d/c rate after prophy. Not atrocious all things considered.