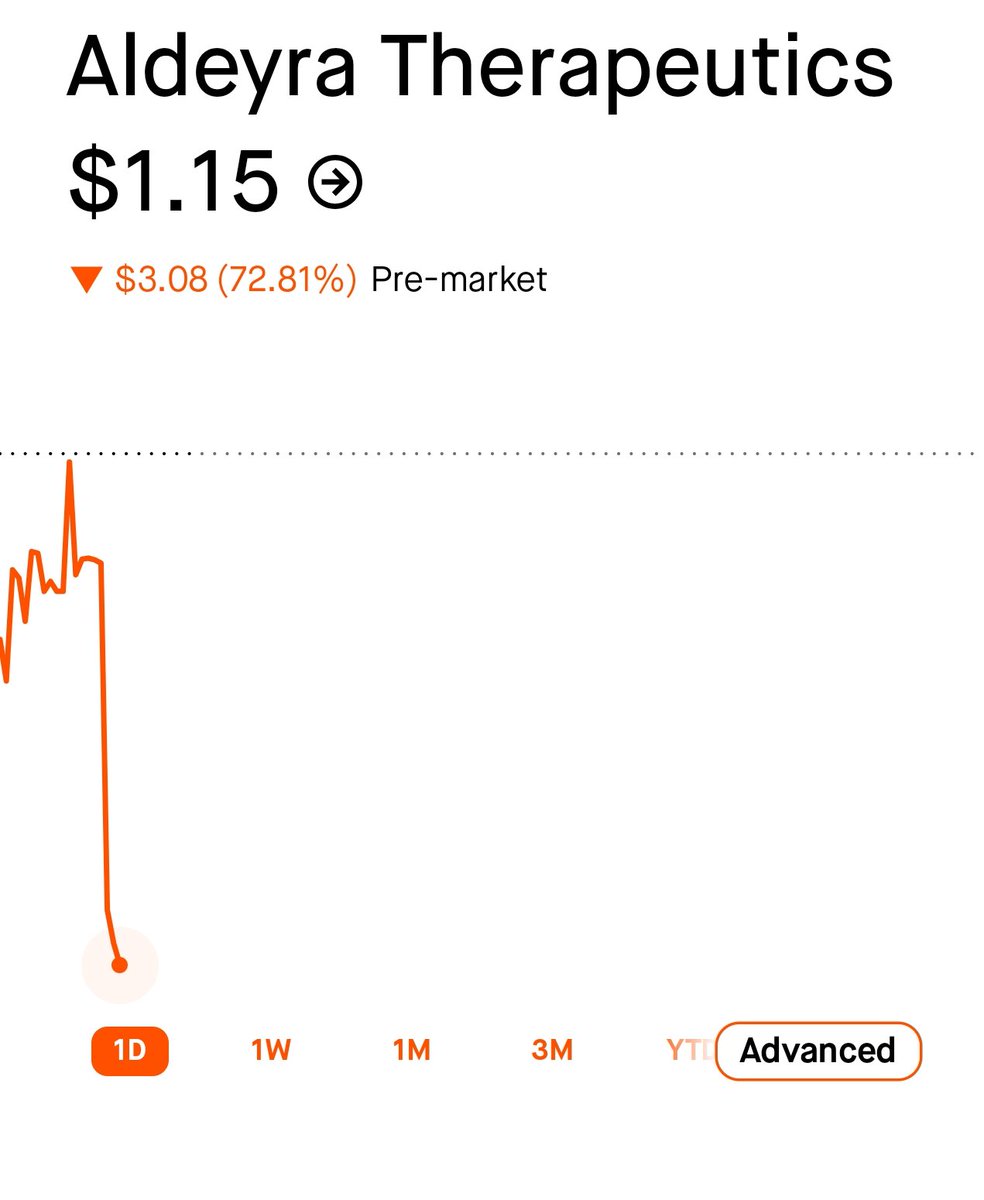
Sally struthers returns
3K posts

Sally struthers returns ری ٹویٹ کیا

$IOVA is a gift at the current levels $3.80-$4. It will be over $10 by end of year. There are so many massive catalysts coming. Most importantly, Amtagvi adoption is accelerating and that means when NSCLC is approved sales will be through the roof. The sales and marketing infrastructure is already being built and fine tuned with the advanced melanoma indication. It will be a well oiled machine soon.
Patience…
English
Sally struthers returns ری ٹویٹ کیا
Sally struthers returns ری ٹویٹ کیا

@BIOTECHSCANNER Amazing year ahead! The Q4 was my biggest prove for $IOVA in 2026 and beyond ☝🏽
English
Sally struthers returns ری ٹویٹ کیا

$IOVA - These numbers are staggering. 234,600 new cases in the US ALONE. Iovance will be there. This is why I don’t ever worry about “competition.” We actually need more treatment options to meet demands. It’s about the patient first and foremost. Remember, 90% of all new cancers are solid tumors. $IOVA can’t do it alone. Iovance Biotherapeutics is becoming a household name in this space. 2026 and beyond will be amazing for this company and the patients who will be saved! Iovance will become big pharma soon.
Melanoma Research Foundation@CureMelanoma
In 2026, more than 234,600 Americans are expected to be diagnosed with melanoma. Thousands will face advanced disease. Progress has been made, but we are not done. Research funding fuels: • New immunotherapies • Genetic discoveries • Earlier detection tools • Clinical trials for advanced melanoma Without funding, progress slows. With you, it accelerates. Donate now to power the next breakthrough: buff.ly/aNqYPiQ #ResearchSavesLives
English
Sally struthers returns ری ٹویٹ کیا

$IOVA Taking a look at Iovance thru the lens of current world events:
1) Higher oil prices - energy is a very small input for Iovance so no impact on margins
2) Tariffs - manufacturing is in US and infrastructure built out to handle 5,000 patients/ year so no need for material rise in input costs
3) War with Iran/potential recession - Amtagvi approved and all treatment in US so world events limited. Cancer does not wait for a war to end or for the economy to improve.
Healthcare and biotech are usually the safe haven when the market gets dicey because they are, for the most part, a non-cyclical sector.
English
Sally struthers returns ری ٹویٹ کیا
Sally struthers returns ری ٹویٹ کیا
Sally struthers returns ری ٹویٹ کیا

$IOVA We thought the week was set, but it wasn’t!
Last week we discussed about Immutep discontinuing their NSCLC trial, a near direct competitor to Iovance’s TIL therapy.
And here we are today before the news that this time it is AstraZeneca discounting a trial that could have been direct competition to TIL in the solid tumour realm.
Things are aligning across the board! Internally and externally.
And here we are, in a Triple Witching day that could bring us further discounts!
Source: fiercebiotech.com/biotech/pfizer…
🤍 Thank you babariaron on StockTwits for the awesome share!
English
Sally struthers returns ری ٹویٹ کیا

@Henry431171040 @BiotechAutist @Investorclimber Absolutely but short term could create a dip. I think it two years they will struggle on the supply side unless they get bought out. Demand will be so massive.
English

@Dancingtapas @BiotechAutist @Investorclimber They also will only compete for 2l so once tilvance 301 is unlocked , its a nothing burger
English

@KingachillesE @A_May_MD They finally started to have decent margins in 2026.
English

@A_May_MD I’m with you on COGS being an issue, however still early days. They have tightened things up in terms of process. As you note it’s quite complicated. I am a trader in this name mostly (because of opportunity), but long term investor as well. THIS MOVE IS OVERDONE.
English

If you’re long $IOVA and only listening to the permabull biotwitter cult leading pumpers who won’t stop tweeting about how positive the earnings results were, you may be wondering why you’re down 20% right now.
Well, it *does* happen that stocks irrationally trade down on good news sometimes. But you may want to be open to the idea that the earnings actually weren’t positive and take a look at how expensive these $IOVA revenues are.
Especially the fact that $8.3M was “wasted” on manufacturing Amtagvi that was never given to patients. This is an EXTREMELY intense and complicated regimen to administer. There will be manufacturing failures, people will drop out electively or even die during preconditioning (or from advanced disease) before getting Amtagvi. The result of these problems is spending a LOT of money on making product that never generates any revenue. A double whammy.
Sure, total revenue exceeded some sellside projects this quarter. But that $8.3M loss just on “wasted” manufacturing accounts for almost 20% of all Amtagvi revenues on the quarter! How many sellside projections have $IOVA immediately dumping 20% of Amtagvi revenue into the trash? Not good.
Quit listening to the permabull pumpers and consider that it’s possible things actually aren’t going well.
Fingers crossed for good NSCLC data this weekend, which could certainly turn things around if resoundingly positive.

English

@Henry431171040 @BiotechAutist @Investorclimber The only potential competition is Repl a little bit for melanoma
English

@Dancingtapas @BiotechAutist @Investorclimber Yeah 10Bil potential for Iovance. Crazy thing is Immutep gave up NSCLC study, AstraZencea failed their NSCLC . Iovance has no competitor aka the first and only immunotherapy if approved in 2027.
English

@Henry431171040 @BiotechAutist @Investorclimber Agreed just wondering how much cash to have on the sidelines for the possibility of that bump caused by the noise. Have a nice position already.
English

@Dancingtapas @BiotechAutist @Investorclimber This will be 12+ anyways the moment it filed sBLA for NSCLC so. All esle is noise and opportunity to buy. Teens is inevitable
English

@BiotechAutist @Investorclimber If Repl gets rejected Iova should get a pop and vice versa
English

@mahasr199 @JPZaragoza1 That's possible but it's so much speculation and unless you're a real insider hard to know.
English

@JPZaragoza1 Considering they submitted the exact same data is their any risk it was mostly padzur pushing the CRL and rest will now just go for it?
English

$REPL advancing a bit pm. Guess it's because Cantor's note with a positive tone for approval after meeting with mgmt. They "came away incrementally more positive in the likelihood of approval". I view the reasons for optimism as fairly weak in particular that their sales team is "pumped about a potential launch" and the support from the melanoma community (?). The roles of Prasad and Pazdur in the CRL and the presenced of new team members at the FDA involved in the review remain speculative. I expect another CRL.
English

@Heatlamp11 @jy201506 This analysis is more logical than the analysis that went something like: because FDA accepted the response as complete, strong likelihood that approves.
I have no position in $REPL.
English

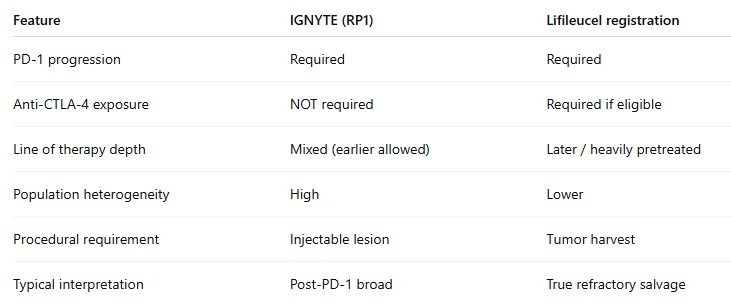
$IOVA another key catalyst coming up is the PDUFA date of April 10, 2026 for $REPL's RP1+Nivo Combo in Melanoma
I think 80% chance it will be another CRL and 20% chance it gets approved with a narrow label. Most likely FDA would want "a single adequate and well‑controlled" phase 3 to support RP1's approval.
Here are why:
FDA already issued a CRL stating the dataset was not an adequate & well-controlled investigation → that is a high-bar deficiency, most difficult to fix relative to other reasons such as CMC.
Core issue = interpretability, driven by heterogeneous “post-PD-1” population (adjuvant relapse mixed with true refractory metastatic pts). Single-arm + heterogeneous population = unclear effect size → classic scenario where FDA pushes for randomized evidence.
Single-arm registration is most persuasive in true salvage/no-good-options settings (such as Amtagvi's registration trial population in 2L+ Melanoma). In RP1's phase 2, many patients haven’t had CTLA-4 (or aren’t clearly ineligible), there’s a credible active alternative → randomization becomes more appropriate.
Recent FDA messaging: one pivotal trial is fine — but must be clean & interpretable → this cuts against weak single-arm packages.
FDA doesn’t always finish CMC review if efficacy already blocks approval. Absence of CMC in the first CRL ≠ CMC cleared. Resubmission often triggers deeper CMC review + inspections
English
Sally struthers returns ری ٹویٹ کیا

@Mahmissa Honestly never know but not sure what's taking so long. Just approve it if you're going to approve it.
English

@Mahmissa Fda has all the same data and keeps waiting the trial design probably sinks them
English
Sally struthers returns ری ٹویٹ کیا

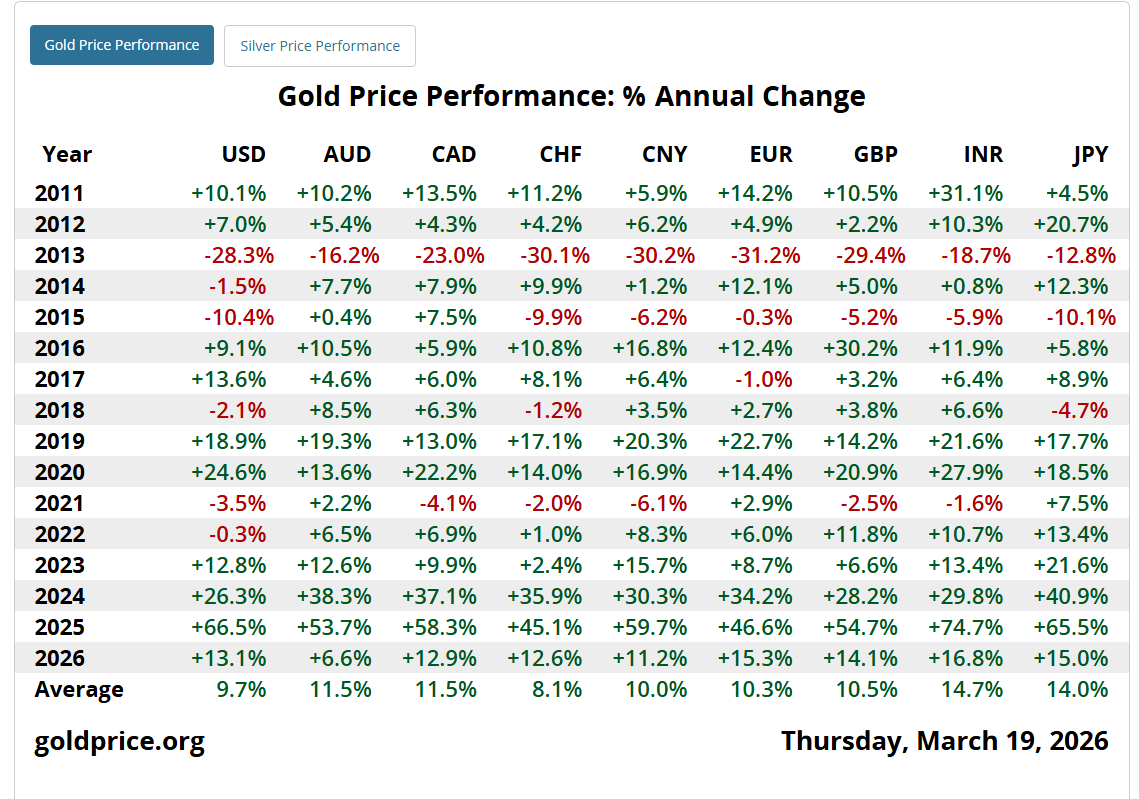
The amount of panic in my inbox over this #Gold sell-off is a good contrarian indicator.
If you are losing sleep over a short-term dip, you aren't looking at the big picture. Gold is still up double-digits in almost every major currency this year.
Don't let recency bias shake you out of a generational bull run. The data speaks for itself. 👇
English