Rupert D.
926 posts


Rupert D.
@Sports_BioTech1
I offer my opinion, take it with a grain of salt.






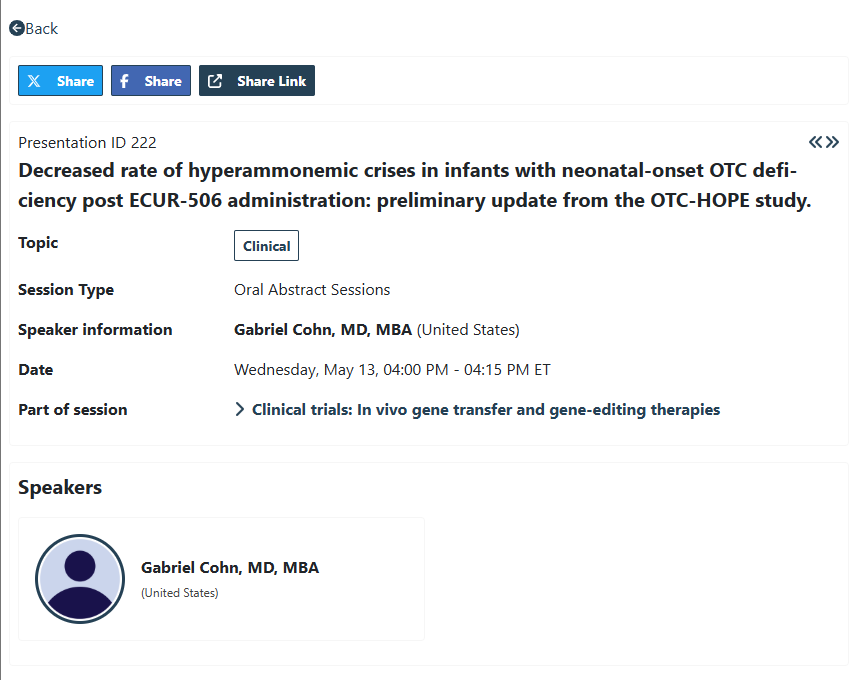
Found myself thinking about $DTIL this morning (as is often the case). I have my eye on EASL (May 27-30 in Barcelona) for a significant data update on Precision’s Hepatitis B program. In their January 12th update, outlining 2026 priorities — they made it a point to call out that they plan to share more details about the program @ Hepatitis focused medical conferences in 2026. To my knowledge, EASL & AASLD are the most prevalent & prestigious Hepatitis focused medical conference (among other liver related diseases as well). The timing of EASL and AASLD are convenient as well for patient data accrual (~mid year and ~year end). As for EASL 2026, I have my eye on a late breaker for $DTIL. In November 2025 (data cut 10/31/25), they shared promising data at AASLD that demonstrated Cohort 3 making strong progress toward a cure. If my math is correct, all patients in cohort 3 have received all three doses @ 0.8 MG/KG by now. I spoke to Precision in November of last year — they mentioned that a fourth dose was on the table for Cohort 3 participants, pending HBV elimination data from the first 3 doses. This, in tandem with Cohort 4 (0.4 MG/KG every 4 weeks) & Cohort 5 (0.65 MG/KG every 4 weeks) — both cohorts did not specify # of doses — should give us a clear sense of the dose level and regiment that it takes to cure a patient of Hepatitis B (IMO). From here, as Precision advances to the expansion phase with their go-forward dose — I’m very interested to see how the approach partnerships. They’ve been forthcoming in the sense that they’ve stated that they are open to Hep B partnership talks now — but can be patient and go-it-alone until after Phase 2. I’d imagine that players like Gilead (early HBV partner), GSK, Roche, etc. would and will be engaging as the cure picture becomes clearer. All that to say, I’m imagining the timing lining up nicely to an EASL readout and an expansion phase update. Certainly hoping we get some more breadcrumbs before May (additional biopsy data?). Between this and the initiation of the DMD trial, I remain excited for 2026.



Found myself thinking about $DTIL this morning (as is often the case). I have my eye on EASL (May 27-30 in Barcelona) for a significant data update on Precision’s Hepatitis B program. In their January 12th update, outlining 2026 priorities — they made it a point to call out that they plan to share more details about the program @ Hepatitis focused medical conferences in 2026. To my knowledge, EASL & AASLD are the most prevalent & prestigious Hepatitis focused medical conference (among other liver related diseases as well). The timing of EASL and AASLD are convenient as well for patient data accrual (~mid year and ~year end). As for EASL 2026, I have my eye on a late breaker for $DTIL. In November 2025 (data cut 10/31/25), they shared promising data at AASLD that demonstrated Cohort 3 making strong progress toward a cure. If my math is correct, all patients in cohort 3 have received all three doses @ 0.8 MG/KG by now. I spoke to Precision in November of last year — they mentioned that a fourth dose was on the table for Cohort 3 participants, pending HBV elimination data from the first 3 doses. This, in tandem with Cohort 4 (0.4 MG/KG every 4 weeks) & Cohort 5 (0.65 MG/KG every 4 weeks) — both cohorts did not specify # of doses — should give us a clear sense of the dose level and regiment that it takes to cure a patient of Hepatitis B (IMO). From here, as Precision advances to the expansion phase with their go-forward dose — I’m very interested to see how the approach partnerships. They’ve been forthcoming in the sense that they’ve stated that they are open to Hep B partnership talks now — but can be patient and go-it-alone until after Phase 2. I’d imagine that players like Gilead (early HBV partner), GSK, Roche, etc. would and will be engaging as the cure picture becomes clearer. All that to say, I’m imagining the timing lining up nicely to an EASL readout and an expansion phase update. Certainly hoping we get some more breadcrumbs before May (additional biopsy data?). Between this and the initiation of the DMD trial, I remain excited for 2026.