

Martins S. Oderinde, Ph.D
210 posts

@oderinde_m
Scientific Associate Director. Discovery Chemistry. Bristol Myers Squibb. Photocatalysis Expert. IAB Member of Angewandte Chemie.



Finally, RADICAL CROSS COUPLING without the exogenous REDOX. We disclose in @ChemRxiv (chemrxiv.org/engage/chemrxi…) a broadly general platform for achieving transformations that normally required excess metallic or chemical reducing agents or photochemical setups (and their requisite catalysts) or potentiostats for electrochemistry. Now, such transformations can be accomplished with the same ease that one conducts a Suzuki coupling. You can scale up (its homogeneous) or scale down (use cheap parallel screening plates). Moisture is tolerated and base metal catalysis is used. As the best engineers like to say, "The best part is no part". Now, chemists have the option to remove the redox part from radical couplings. Quick Summary: Sulfonyl hydrazides are disclosed as versatile radical precursors as exemplified with seven new C–C bond forming, redox-neutral cross-couplings with: (1) activated olefins, (2) alkyl halides, (3) redox active esters, (4) aryl halides, (5) alkenyl halides, (6) alkynyl halides, and (7) a trifluoromethylating reagent to forge C(sp3)-C(sp3), C(sp3)-C(sp2), and C(sp3)-C(sp) bonds. Sulfonyl hydrazides are stable and usually crystalline substances that can be accessed in a variety of ways including transiently from hydrazones to achieve a net reductive arylation of carbonyl compounds. Exogenous redox (chemical, photo/electrochemical) additives are not necessary as these functional groups serve the dual role of radical precursor and electron donor. The operational simplicity (homogeneous, water tolerant, dump-and-stir) and practicality of the method are demonstrated as well as applications to streamlining synthesis and mild late-stage functionalization.


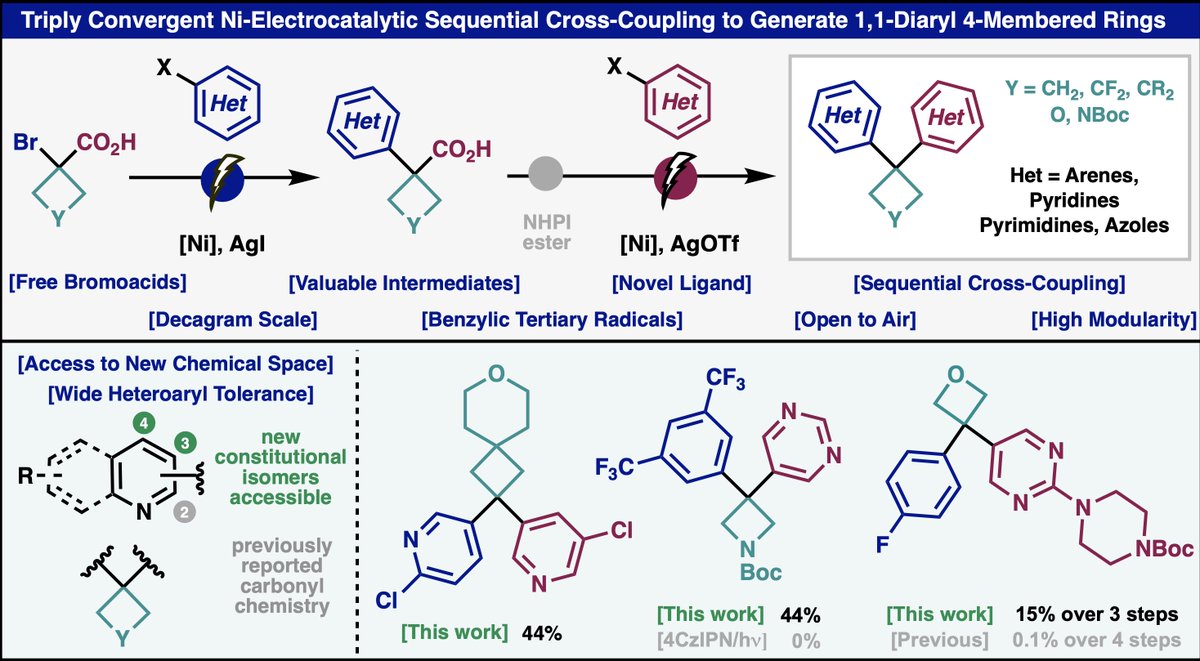


Radical retrosynthesis strikes again. Primary RAEs can be mixed and matched with tertiary RAEs or olefins to make quaternary centers with a comical level of simplicity. Check out these applications. A fun collaboration with the great and powerful @Shenvi_Lab: tinyurl.com/7heyrvka







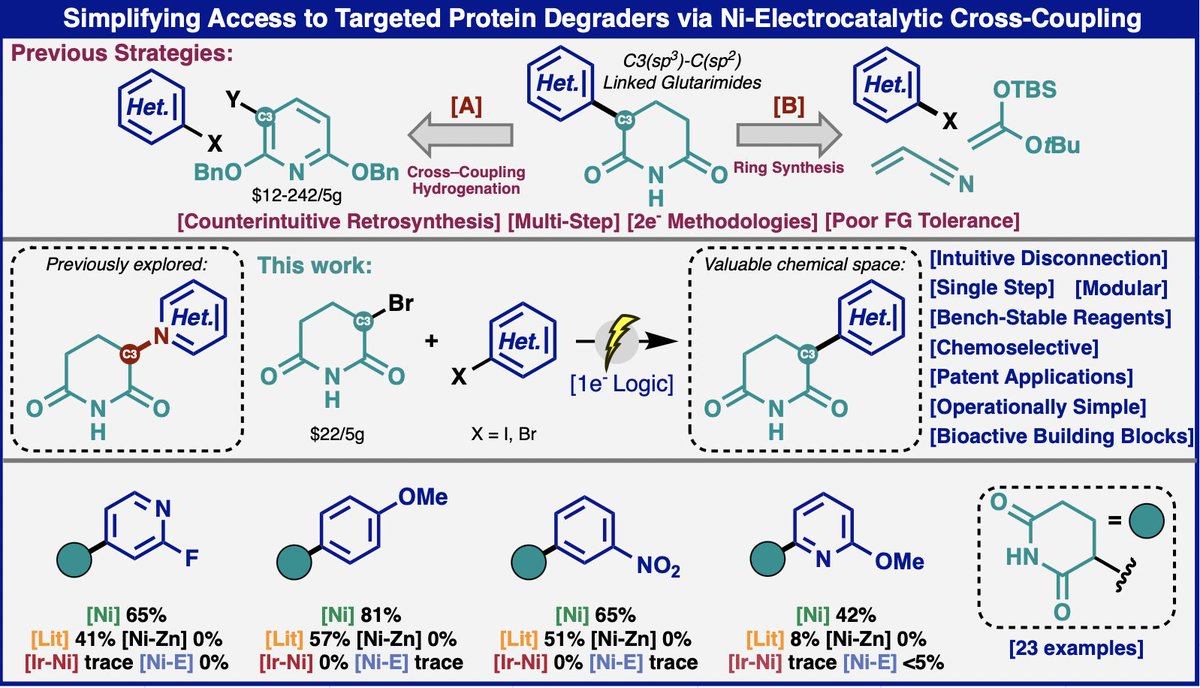
Degrade proteins, not your work schedule! A new method appearing today @ChemRxiv (chemrxiv.org/engage/chemrxi…) simplifies access to molecular glues (like PROTACS) to save medicinal chemists valuable time. One-step access from cheap commercial materials, a trivial experimental setup, and superb chemoselectivity enabled by Ni-electrocatalysis. Another fun collaboration with BMS.



The updated version of this work has appeared today @ChemRxiv now with applications, scale-up, and mechanistic studies.

Degrade proteins, not your work schedule! A new method appearing today @ChemRxiv (chemrxiv.org/engage/chemrxi…) simplifies access to molecular glues (like PROTACS) to save medicinal chemists valuable time. One-step access from cheap commercial materials, a trivial experimental setup, and superb chemoselectivity enabled by Ni-electrocatalysis. Another fun collaboration with BMS.


Chiral amino alcohols without the hassle, radically simplified appearing today in @ChemRxiv : chemrxiv.org/engage/chemrxi…