Angehefteter Tweet
Market Pathways | MedTech Regulatory & Policy
3.1K posts


Market Pathways | MedTech Regulatory & Policy
@Market_Pathways
Exploring the people, challenges, and opportunities impacting the global #medtech regulatory, reimbursement and policy communities. Likes,RTs,etc ≠ endorsement.
Stay informed ➡️ Beigetreten Nisan 2019
143 Folgt263 Follower

The only database of its kind in #medtech is here. Pathways’ Document Depot tracks global medtech policy documents, guidance, and rules in one searchable hub. Stay on top of the #regulatory landscape: bit.ly/4snTlAw

English

Entering the Swiss #medtech market is changing fast. Our Global Spotlight examines Swissmedic regulation, new TARDOC #reimbursement rules, and what the evolving EU-Swiss relationship means for device companies: bit.ly/4uuEbef

English

Reimbursement can make or break a #medtech innovation. @bostonsci's Intracept has grown to ~$200M in revenue with coverage for 200M+ lives. See how a targeted #reimbursement strategy drove adoption: bit.ly/4sc2Qmp

English

What do FDA staffing cuts mean for #medtech innovation? Josh Makower (@joshatstanford) discusses #regulatory slowdowns, the US “first mover disadvantage,” and ways to improve efficiency in device review: bit.ly/4bpVggJ

English

Is the EU MDR finally shifting course? Our infographic maps the #MDR timeline from 2017 to 2028 and highlights how proposed reforms could reduce bottlenecks and support #medtech innovation in Europe: bit.ly/4du93FE
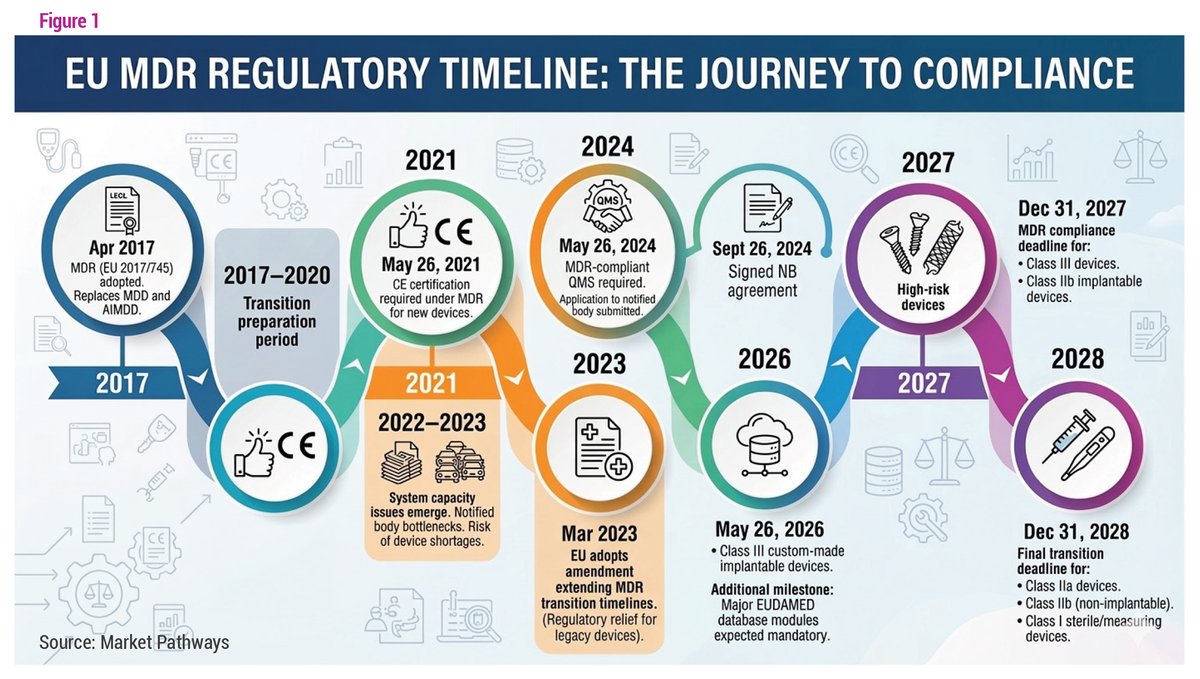
English

Where do MDUFA VI negotiations stand? #FDA and industry are nearing a tentative agreement, but key issues remain unresolved. Our latest analysis breaks down the progress, debates, and what comes next: bit.ly/4sbXB6k

English
Market Pathways | MedTech Regulatory & Policy retweetet

Criteria-independent genetic testing identified pathogenic variants in 3.1% of women without a history of #BreastCancer, including many lacking common risk factors.
ja.ma/4bfbYAx

English

Global #medtech policy activity accelerated in February.
FDA launched QMSR, regulators advanced reliance frameworks, and #digitalhealth guidance evolved worldwide: bit.ly/4csvk6t

English

China’s device approvals ticked up in 2025, with domestic and localized firms continuing to dominate.
The total number of #devices approved in China increased slightly last year as the volume of products leveraging the special Innovative Device Pathway continues to grow.
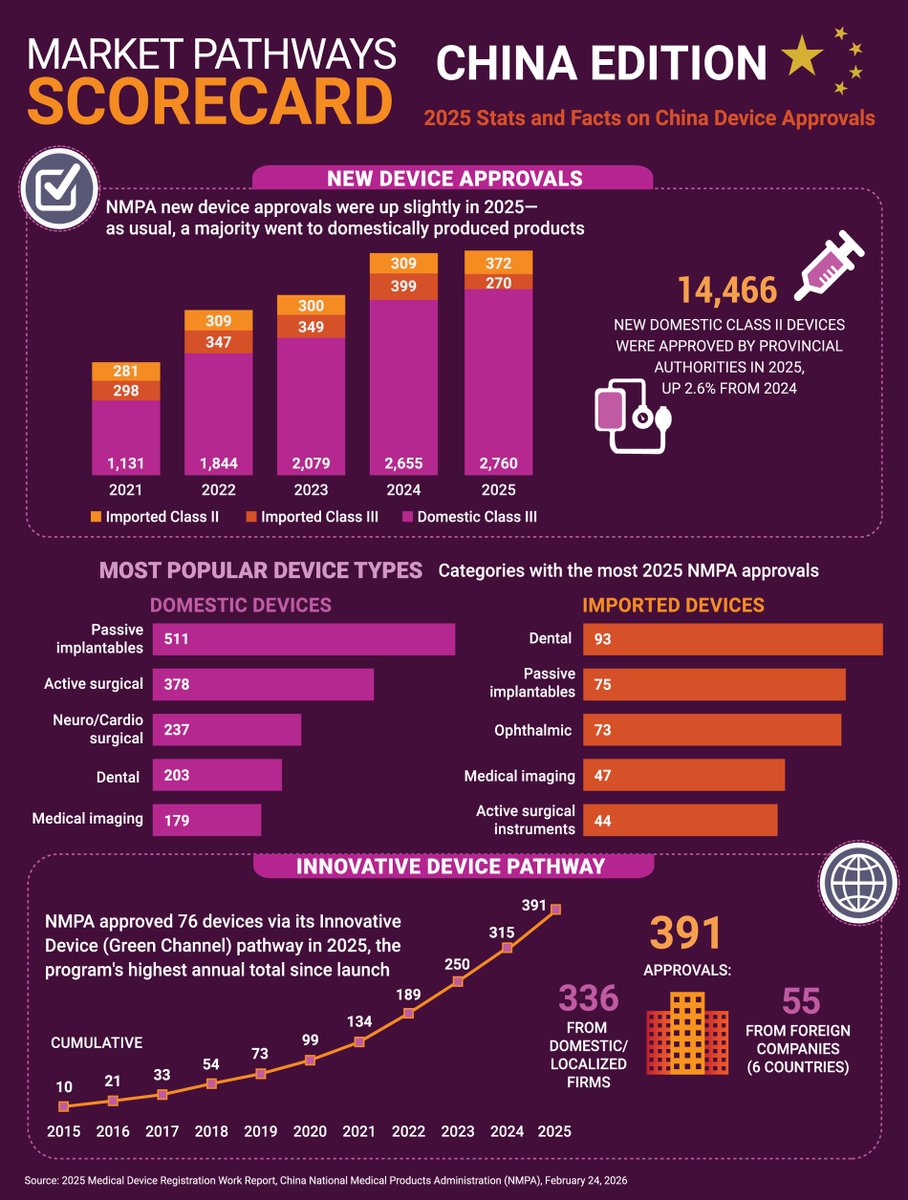
English

IP can define the value of a #medtech start-up. In this article, Adam Hines of HGF Ltd. explains how companies can build a sustainable global IP strategy to protect #innovation and attract investment: bit.ly/4ujXuXK

English
Market Pathways | MedTech Regulatory & Policy retweetet

Where is #medtech capital coming from today?
Join our #InnovationDublin 2026 panel on public, private, and patient capital featuring leaders from IHI, Anthro Ventures, @ClaretCapital, Goodbody, @Market_Pathways, and more: bit.ly/4s232o9

English

What’s shaping #medtech in March 2026? Explore the new Market Pathways digital edition covering #AI software rules, #reimbursement strategy, MDR/IVDR changes, MDUFA VI, QMSR, and more: bit.ly/3MVUPTu

English
Market Pathways | MedTech Regulatory & Policy retweetet

This Narrative Review considers postacute care settings to assist hospital-based clinicians in effectively collaborating with patients, caregivers, and interdisciplinary care teams to facilitate transitions to high-quality postacute care. ja.ma/4cQMWcf

English
Market Pathways | MedTech Regulatory & Policy retweetet

Europe just expanded its harmonized standards under MDR and IVDR. Our latest Docs of the Month breaks down 12 new MDR and six IVDR standards and what they mean for presumption of conformity: Source: bit.ly/4l3mi29 @EU_Commission

English
Market Pathways | MedTech Regulatory & Policy retweetet

A strong IP strategy can shape the future of a #medtech startup. Adam Hines explains how patents, trade secrets, and licensing strategies help companies protect innovation and create long-term value. Read more: bit.ly/4brcJFV

English
Market Pathways | MedTech Regulatory & Policy retweetet

AI is reshaping #healthcare, but regulation is struggling to keep pace. US and EU policymakers are revisiting software rules, yet major questions remain about how generative AI will fit into device frameworks: bit.ly/4sIibez #MedTech

English
Market Pathways | MedTech Regulatory & Policy retweetet

Skilled nursing facility operating capacity in the US declined by 5% since 2019, with the largest losses in rural counties and areas reporting staff shortages, impacting hospital discharge and access to facility-based postacute care. ja.ma/46SUebZ

English