Sabitlenmiş Tweet
Market Pathways | MedTech Regulatory & Policy
3.2K posts


Market Pathways | MedTech Regulatory & Policy
@Market_Pathways
Exploring the people, challenges, and opportunities impacting the global #medtech regulatory, reimbursement and policy communities. Likes,RTs,etc ≠ endorsement.
Stay informed ➡️ Katılım Nisan 2019
143 Takip Edilen262 Takipçiler

Postmarket surveillance is evolving fast. Regulators now expect manufacturers to actively analyze device safety data, not just submit it. Read more here: bit.ly/4doCmYV #medtech
English

Three novel FDA device actions and multiple Class I recalls marked the first half of May 2026. Our latest #FDA Output roundup tracks approvals, recalls, warning letters, and more: bit.ly/4tsfVHY

English

Faster coverage is the goal, but questions remain. @FDADeviceInfo and @CMSGov introduce RAPID to align approval and #Medicare coverage for Breakthrough Devices. Industry is watching closely as details unfold: bit.ly/4d8cvEp #medtech

English

The shift is here, and many aren’t ready. Regulators are now analyzing device safety data and expect manufacturers to do the same. Why 2026 is a turning point for surveillance: bit.ly/4euIj8F #medtech

English

Where do companies fail MOST after #FDA?
• #Reimbursement gaps
• Weak commercialization
• Not enough payer data
• Investor drop-off
~115 TAP devices aimed to fix this. What’s the real issue?

English

If you can’t explain the #medtech landscape, you’re already behind.
Market Pathways gives you the insight on global regulation, #reimbursement, and policy so you can speak with confidence.
Start your free trial today: bit.ly/4w5fyFQ

English
Market Pathways | MedTech Regulatory & Policy retweetledi

Back from lunch at #InnovationDublin 2026. Now live: is the #medtech public market still viable, or is M&A the only path? Experts from @ThisIsBTIG, @Piper_Sandler, @Market_Pathways, and more break down capital markets, deal activity, and investor sentiment.


English
Market Pathways | MedTech Regulatory & Policy retweetledi

How are #medtech innovators funding growth in today’s shifting landscape? Happening now at #InnovationDublin 2026, this panel with leaders from @ClaretCapital, @Market_Pathways, and more explores public, private, and patient capital strategies.




English
Market Pathways | MedTech Regulatory & Policy retweetledi

FDA and CMS have proposed a parallel review program to expedite certain Class II and Class III breakthrough medical devices to market for Medicare patients called the Regulatory Alignment for Predictable and Immediate Device (RAPID) coverage pathway. hubs.la/Q04dr9-p0

English
Market Pathways | MedTech Regulatory & Policy retweetledi

Today FDA approved the first-ever gene therapy for the treatment of genetic hearing loss under the National Priority Voucher Program.
✅6th approval under the National Priority Voucher Program
✅Approved 61 days after filing
fda.gov/news-events/pr…
English

Leading in #medtech starts with insight.
Market Pathways delivers global analysis on #regulatory, reimbursement, and policy trends so you can stay ahead and make smarter decisions: bit.ly/4u3m9ie
English

Struggling with #MDR/IVDR classification? The updated EU Borderline & Classification Manual (v5, April 2026) offers key guidance. Explore it and access more in Pathways’ Document Depot: bit.ly/4dZDRyB



English

This week: HHS Secretary Kennedy hints about work on Breakthrough Device coverage policies; Kenya publishes a forward-leaning #AI guidance; Europe updates its guides on risk classifications and borderline products; and more: bit.ly/48RRhsX

English
Market Pathways | MedTech Regulatory & Policy retweetledi

#FDA approval is only half the battle. Learn how to accelerate the path to #reimbursement with leaders from @CMSGov, @proverummedical, @SUBiodesign, @Market_Pathways, and more at #InnovationDublin. Join our expert panel April 28 in Dublin: bit.ly/4cFURrm

English

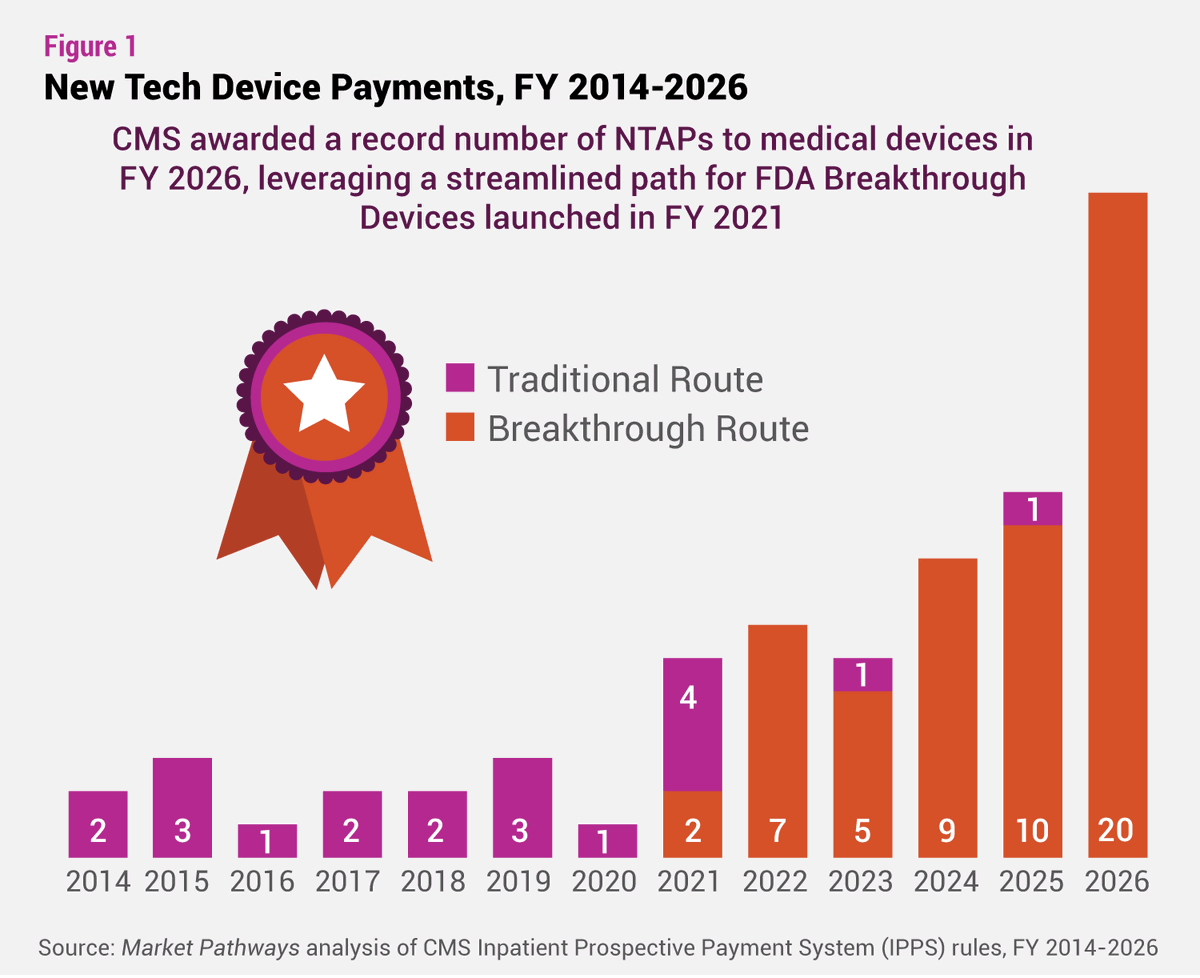
NTAP awards hit record highs, driven by the Breakthrough Device pathway. Now @CMSGov may end it. What could this mean for #medtech innovation and #reimbursement? bit.ly/4e0sIgR
English

#ICYMI: What’s shaping #medtech policy right now? The latest Market Pathways digital edition covers AI software rules, MDR updates, @bostonsci #reimbursement strategy, and expert insights. Read more: bit.ly/4sLHFqY

English

How fast will MDR/IVDR reforms move? EU leaders signal urgency, but no clear timeline. MEP Tomislav Sokol highlights the challenge of balancing speed with complex negotiations: bit.ly/4tkLxA0 #medtech

English

What happens after #FDA approval?
Former CDRH leaders are launching White Oak 66 Capital to tackle post-FDA risk, focusing on #reimbursement and market access from day one: bit.ly/4efq4UA

English

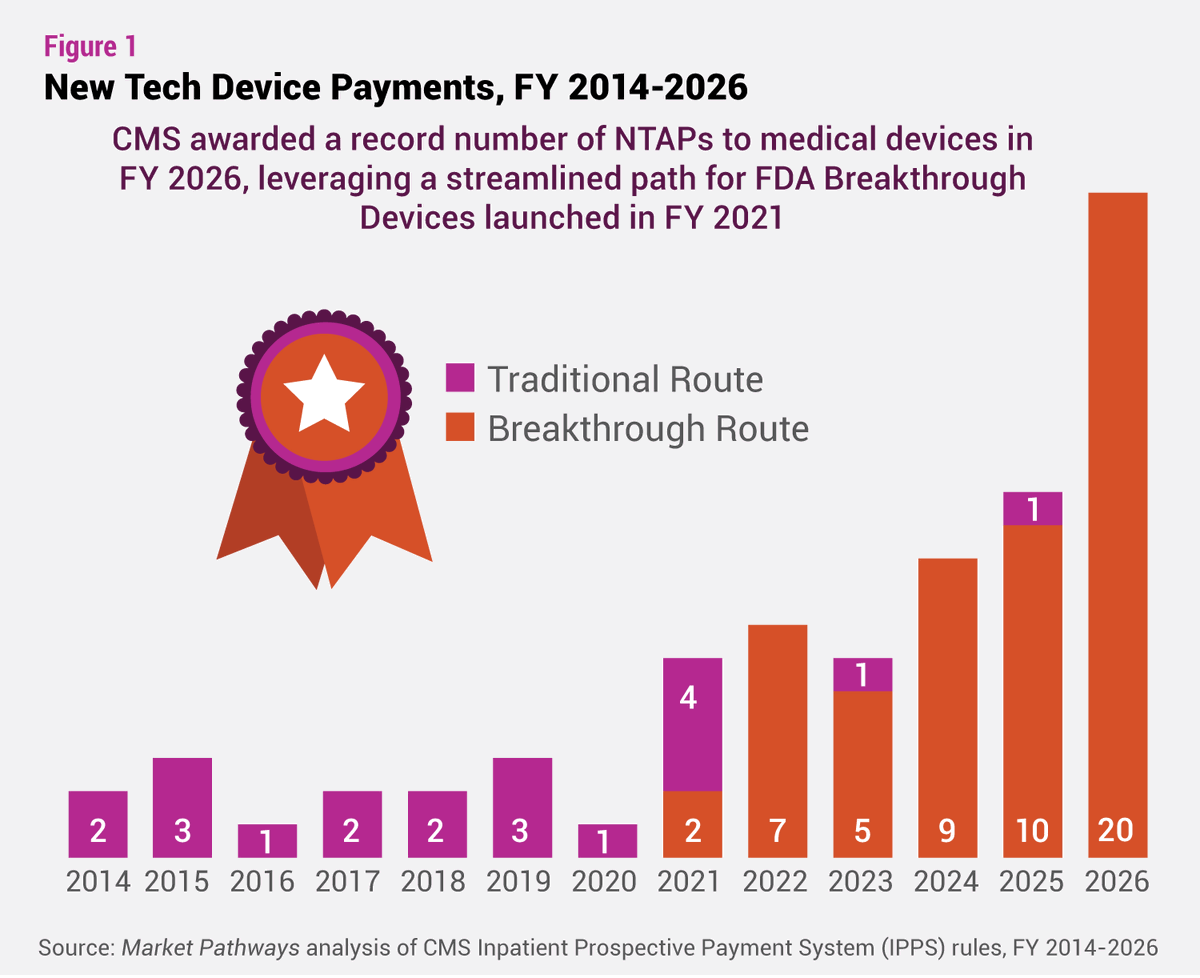
The NTAP surge may be ending. #CMS plans to remove the Breakthrough Device pathway and require proof of clinical improvement, potentially reducing awards and reshaping #reimbursement strategy: bit.ly/3OcT44R
English

The #medtech regulatory landscape is evolving fast. Stay ahead with Market Pathways, delivering global insights on regulation, #reimbursement, and market access: bit.ly/4ckhRM9

English