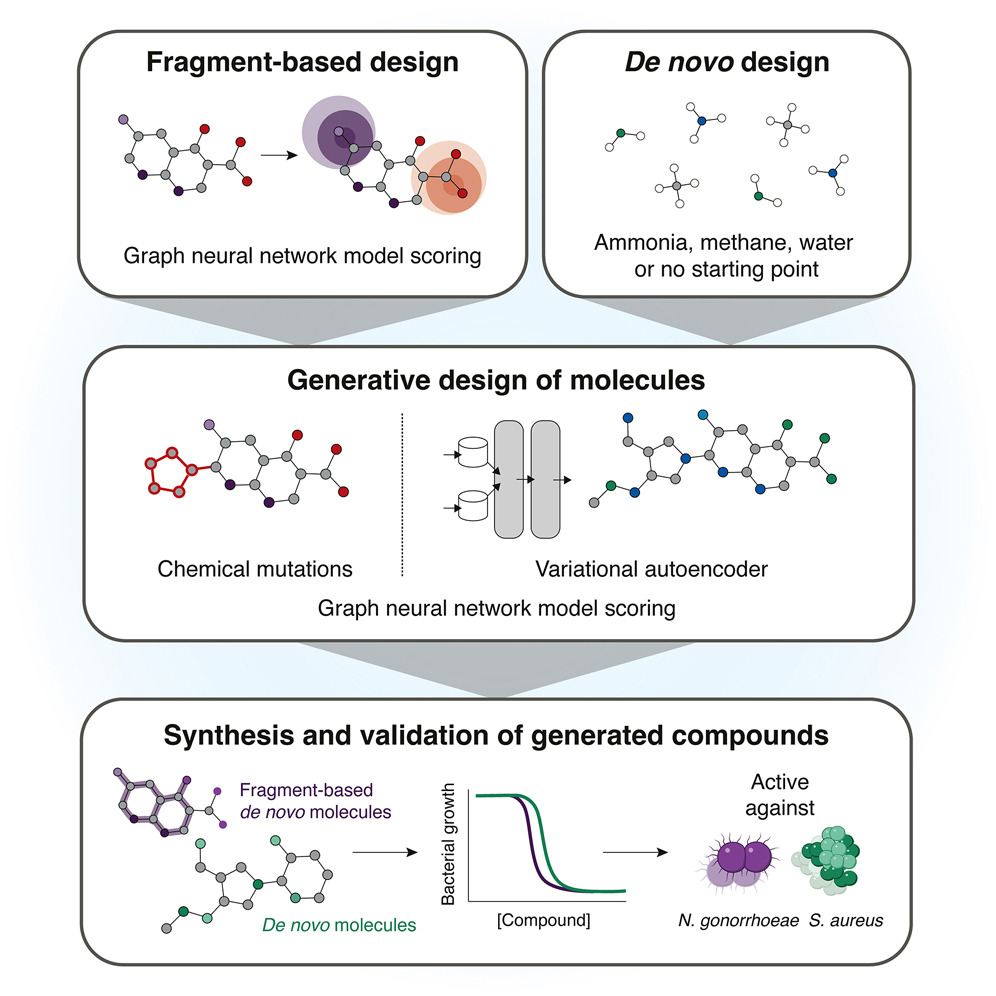
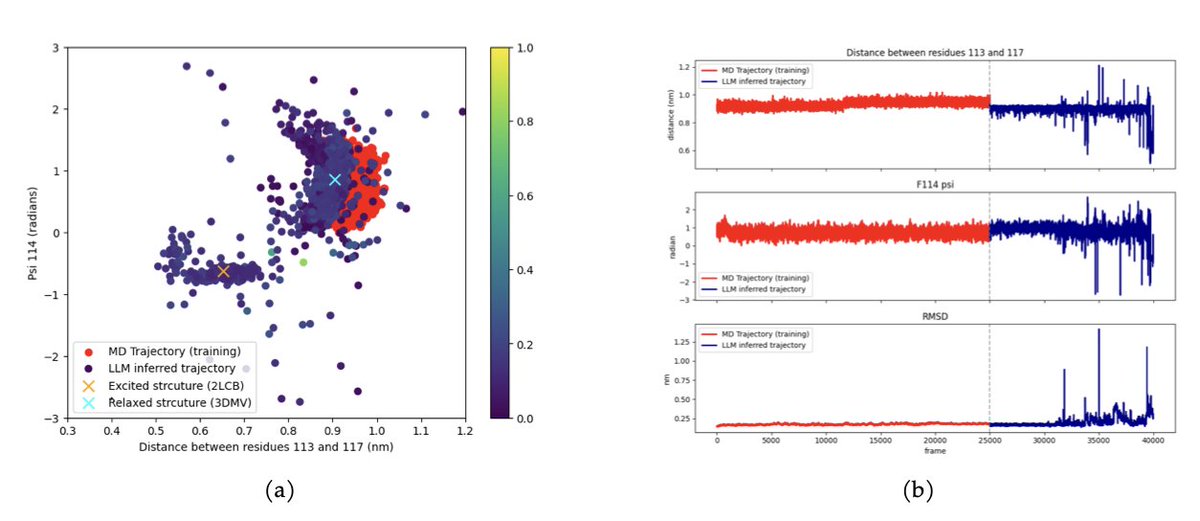
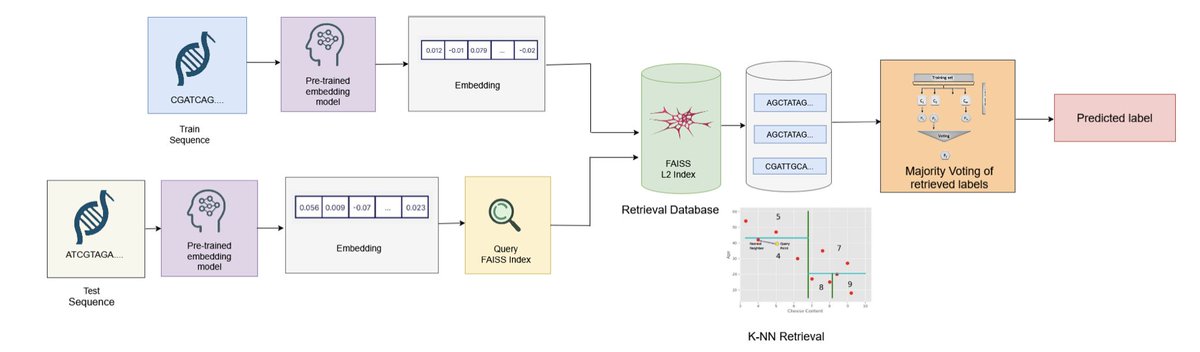
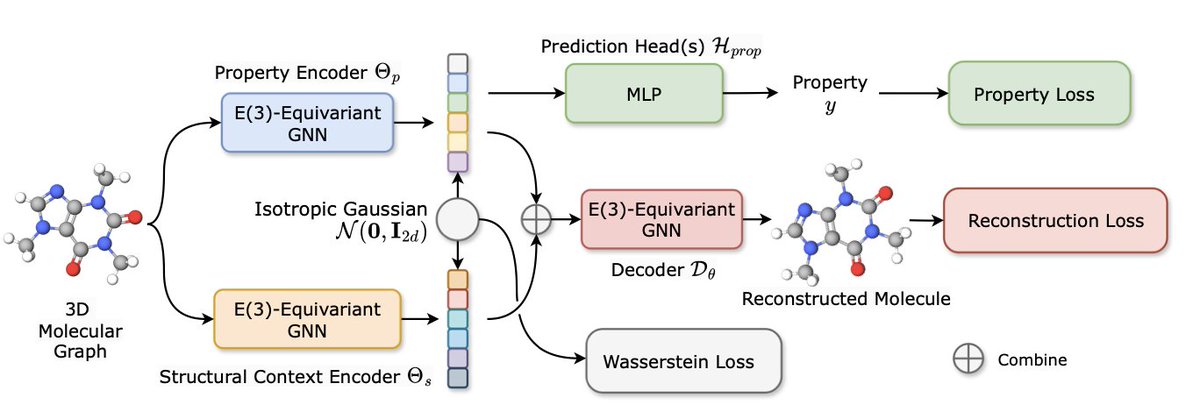
Yu Zhang retweeté

At @GoogleDeepMind, we believe AI is the ultimate catalyst for science. 🧬
The best example of this has been the AlphaFold database (AFDB) of protein structure predictions which has been used free of cost by more than 3.3 millions researchers across the world!
Today, in collaboration with @emblebi, @Nvidia and @SeoulNatlUni, we are expanding the database by adding millions of AI-predicted protein complex structures to the AlphaFold Database. To maximise global health impact, we’ve prioritised proteins that are important for understanding human health and disease, including homodimers from 20 of the most studied organisms, including humans, as well as the @WHO’S bacterial priority pathogens list.
Read more here:
embl.org/news/science-t…
English