
IslaySmoke
1.3K posts


IslaySmoke
@IslaySmoke
I watch the UFC and say, "I could beat that guy" after every fight.







$LLY | Eli Lilly Q1 Earnings Highlights 🔹 Revenue: $19.80B (Est. $17.77B) 🟢; +56% YoY 🔹 Adj. EPS: $8.55 (Est. $7.06) 🟢; +156% YoY 🔹 Mounjaro: $8.66B (Est. $7.21B) 🟢; +125% YoY 🔹 Zepbound: $4.16B (Est. $4.03B) 🟢; +80% YoY 🔹 FDA Approval: Foundayo, Lilly’s oral GLP-1 pill for obesity Raises FY26 Guide: 🔹 Revenue: $82B-$85B (Est. $80B) 🟢; raised from $80.0B-$83.0B 🔹 Adj. EPS: $35.50-$37.00 (Est. $33.50) 🟢; raised from $33.50-$35.00 🔹 Performance Margin: 47.0%-48.5%; raised from 46.0%-47.5% 🔹 Tax Rate: 18%-19%; unchanged Other Metrics: 🔹 Key Products Revenue: $13.4B 🔹 U.S. Revenue: $12.1B; UP +43% YoY 🔹 Revenue Outside U.S.: $7.7B; UP +81% YoY 🔹 Worldwide Volume Growth: +65% 🔹 Realized Prices: DOWN -13% 🔹 Acquired IPR&D Charges: $584M vs $1.6B YoY 🔹 Q1 EPS included $0.52 of acquired IPR&D charges vs $1.72 YoY Financials: 🔹 Non-GAAP Gross Margin: 82.6%; DOWN -0.9 pts YoY 🔹 R&D Expense: $3.5B; UP +28% YoY 🔹 Marketing, Selling & Admin Expense: $2.9B; UP +19% YoY 🔹 Non-GAAP Net Income: $7.7B; UP +155% YoY 🔹 Non-GAAP Tax Rate: 16.5% Commentary: 🔸 “2026 is off to a strong start, we delivered 56% revenue growth in the first quarter and raised our full-year revenue guidance by $2 billion.” 🔸 “A key milestone was the U.S. FDA approval of Foundayo—the only approved GLP-1 pill that can be taken any time of day, without food and water restrictions.” 🔸 “Foundayo will meaningfully expand the number of people who can benefit from GLP-1s.”























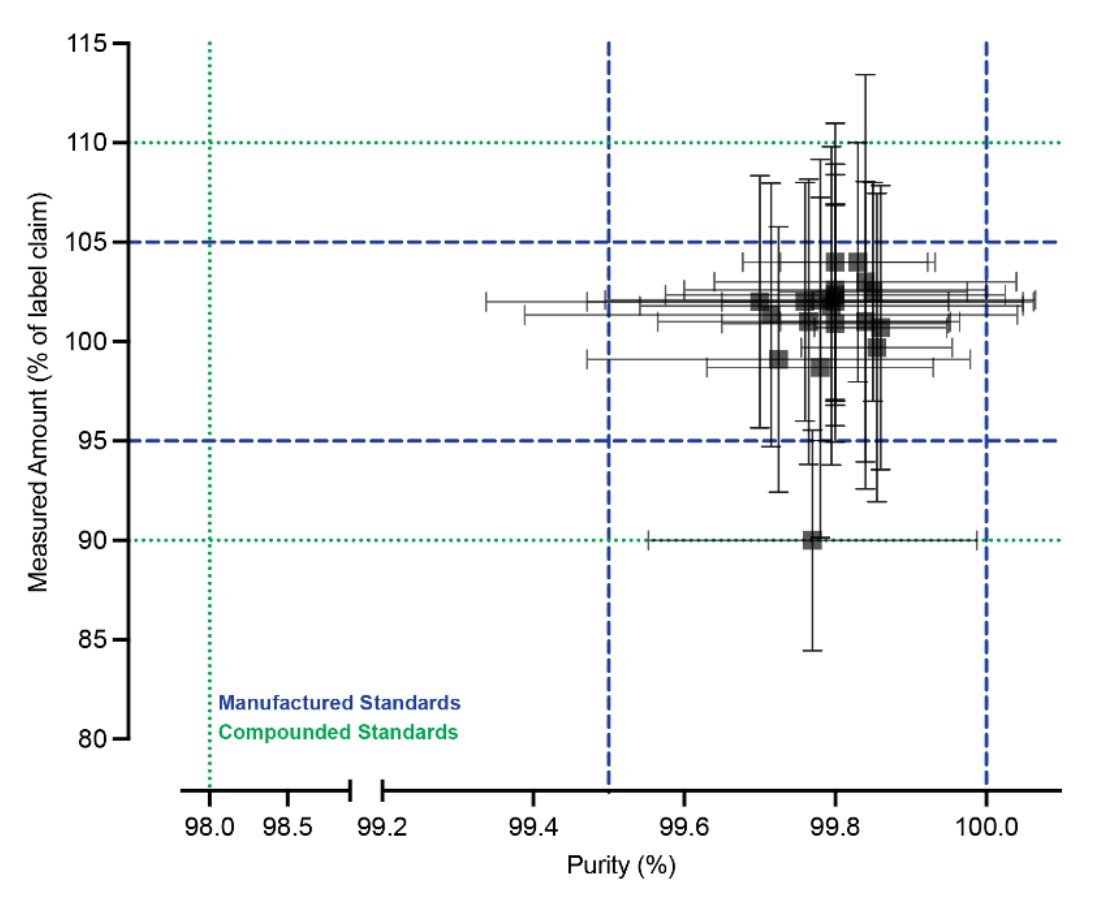
A new study has just come out vetting the quality of gray market peptides! What did it show?🧵 First result: The median and the 25th/75th-percentile peptide vials tend to be filled correctly, with between ~95% and ~110% of the amount they say they contain:




FDA proposes to exclude semaglutide, tirzepatide, and liraglutide on the 503B bulks list. After careful review, we did not identify sufficient clinical need for outsourcing facilities to compound these drugs from bulk substances. Public comment period now open. Learn more: fda.gov/news-events/pr…



