

PubMed.ai
2.4K posts


@AIpubmed
https://t.co/IUnIwoDvGc Deep Search Get a Free Comprehensive Research Report Search, Chat, and Generate Reports with Ease! Check https://t.co/y9Xd2dBj3s










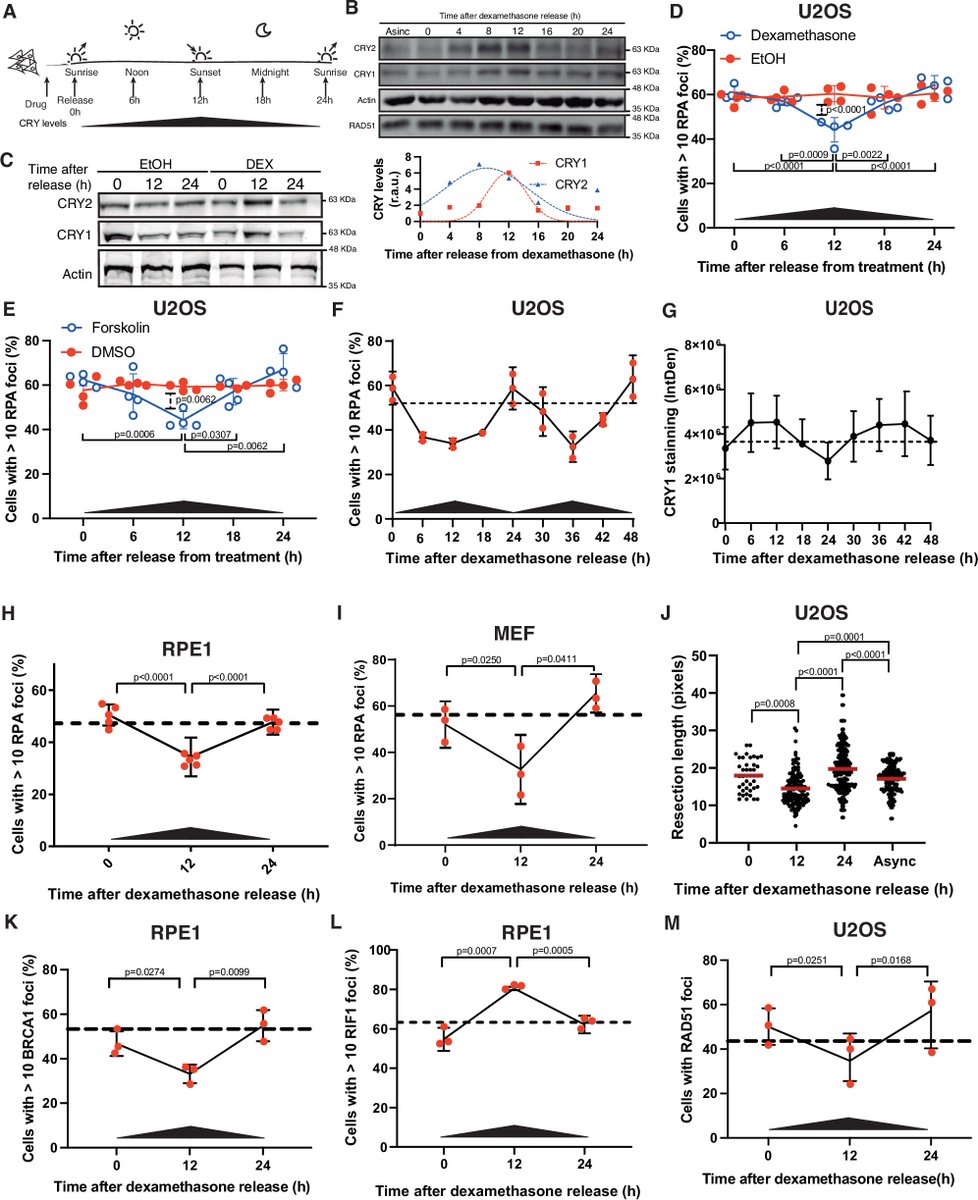
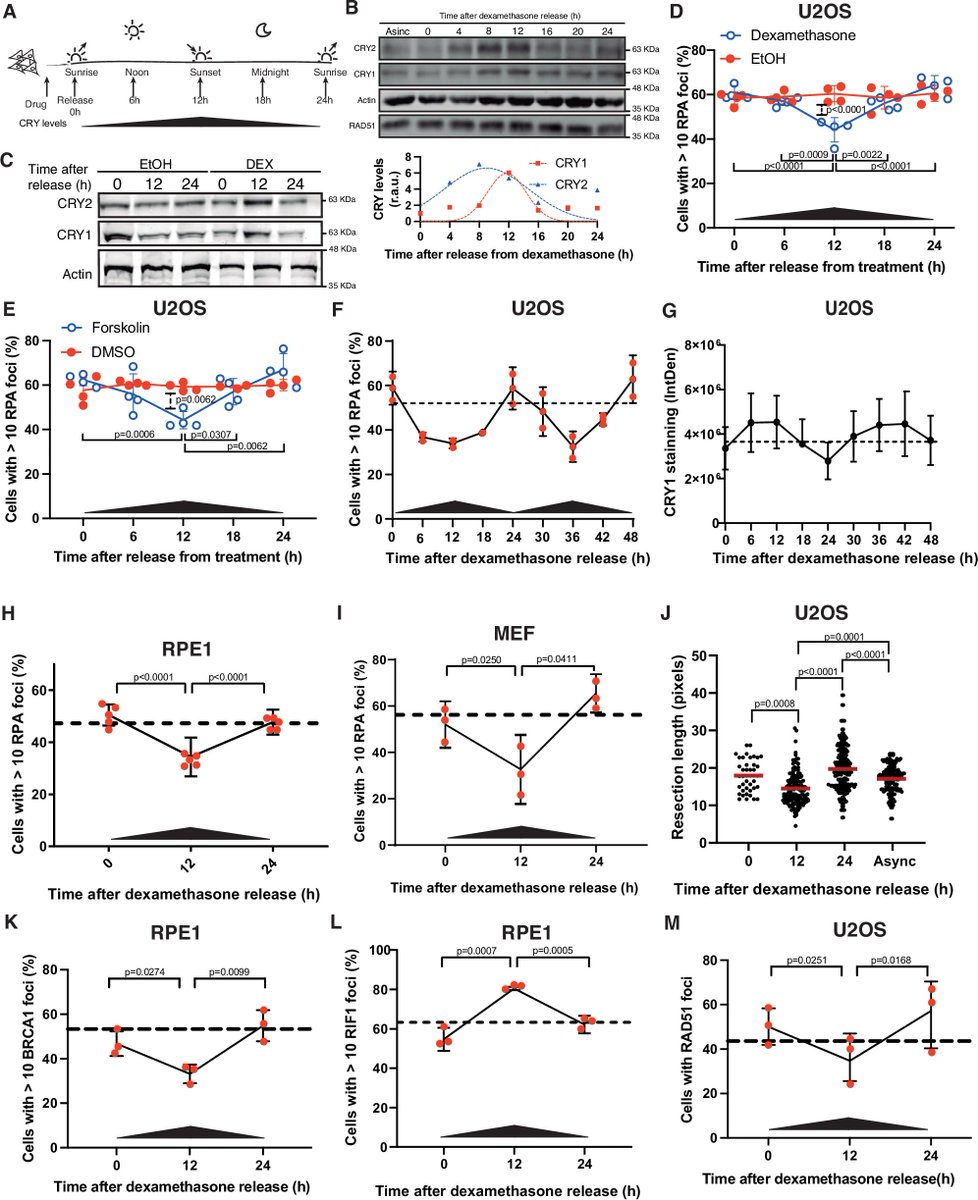
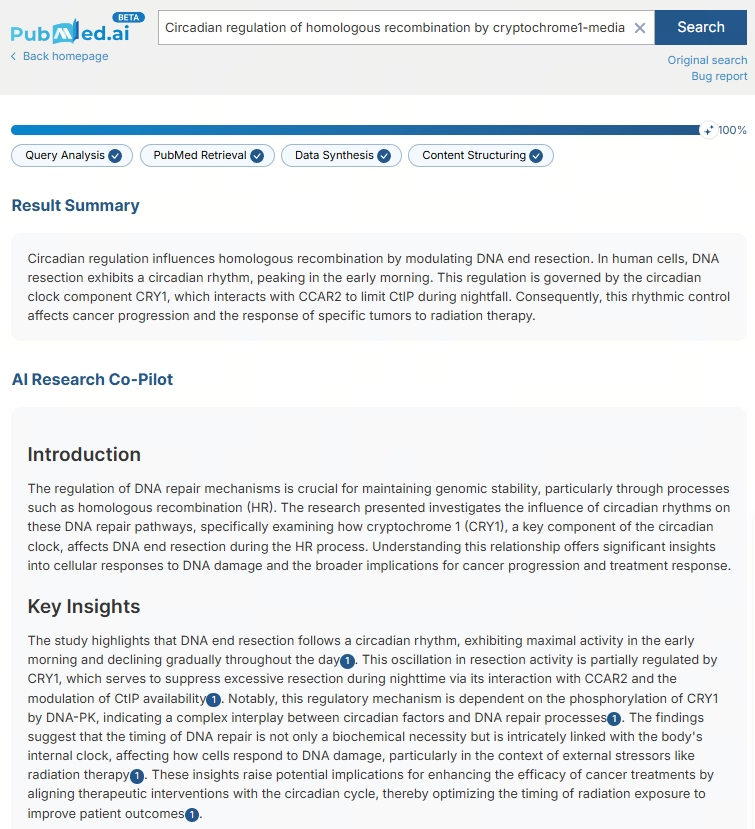
Circadian control of DNA repair may shape cancer outcomes Find More Research: pubmed.ai/?utm_source=X&… New research reveals that homologous recombination is not constant across the day. In human cells, DNA end resection follows a circadian rhythm, peaking in the early morning. This process is tightly regulated by the clock protein CRY1, which interacts with CCAR2 to restrain CtIP-mediated resection during nighttime. The result is a rhythmic modulation of DNA repair capacity. These findings link the circadian clock directly to genome stability — with important implications for tumor progression and radiotherapy timing. #CircadianBiology #DNARepair #CancerResearch #Radiotherapy #Genomics








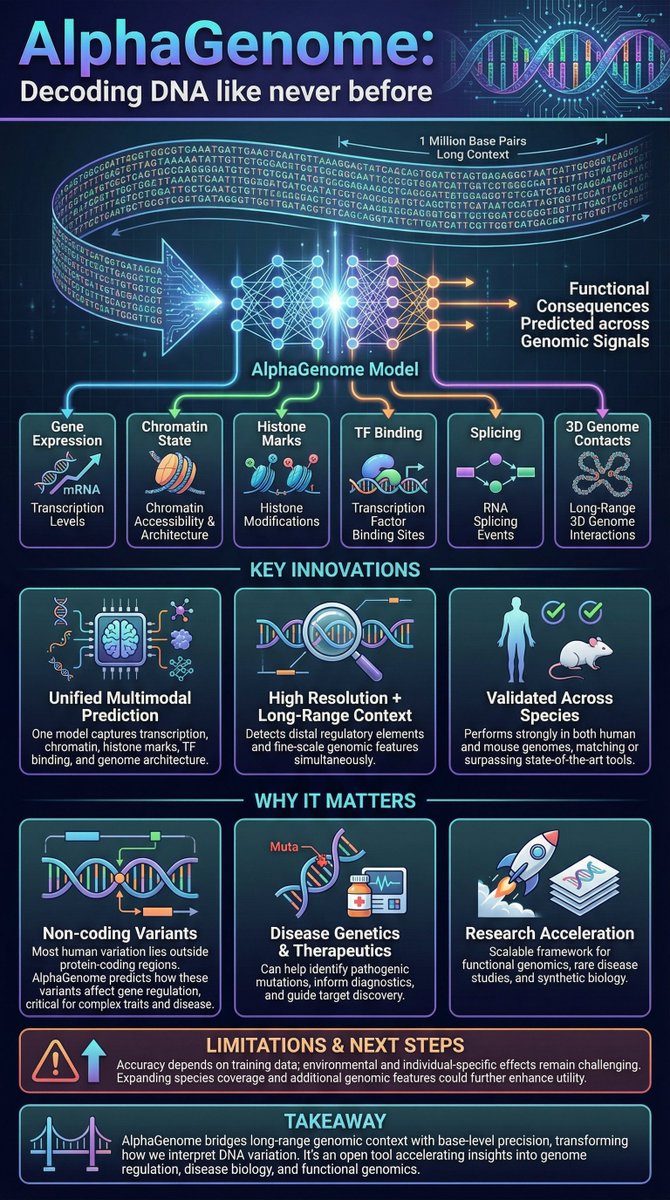
BREAKTHOUGH: Google's AI can now read 1 million DNA letters at once. Google's DeepMind has unveiled a revolutionary deep learning model, AlphaGenome, which can analyze long sequences of DNA with remarkable accuracy. A new peer reviewed study published in Nature, AlphaGenome can process up to 1 million base pairs (1 megabase) in a single input, capturing long range genetic interactions that previous models could not. The system predicts how single letter DNA changes affect gene expression, RNA splicing, and chromatin regulation across 11 genomic signals, even within the 98% of the human genome that does not code for proteins. In benchmark tests, AlphaGenome matched or outperformed previous state of the art models at identifying functionally important genetic variants. By making large sections of the non-coding genome interpretable, AlphaGenome could significantly accelerate disease variant discovery, cancer research, and precision medicine, moving genomics from sequence reading toward functional understanding.

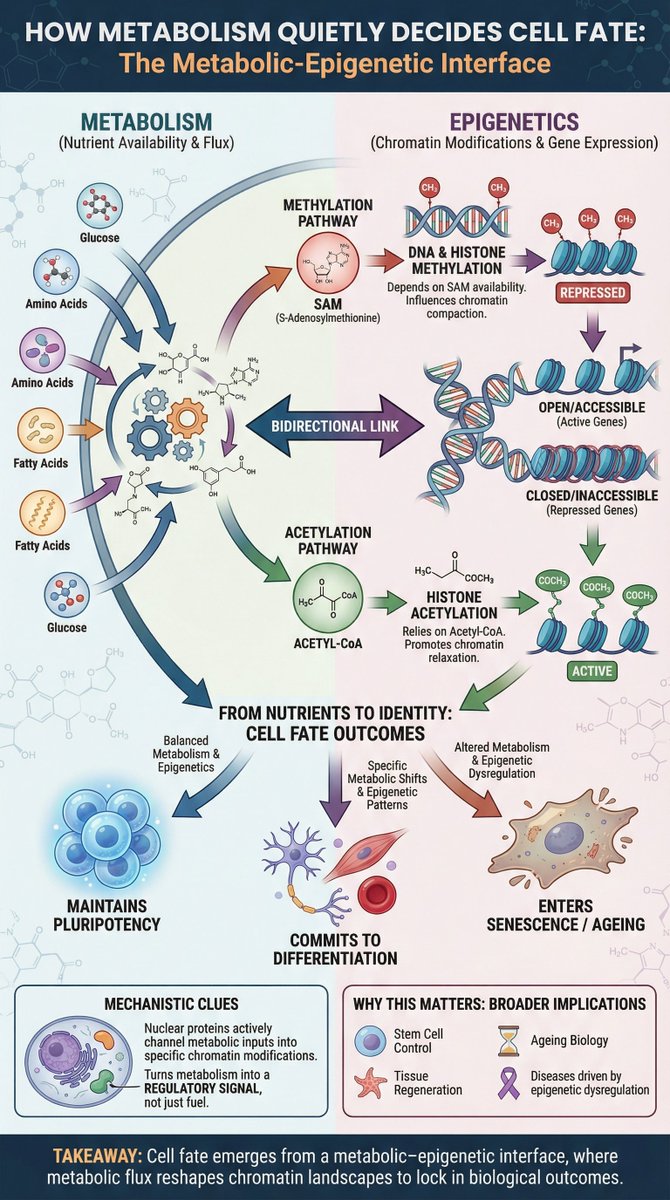
Methylation, acetylation and cell fate dlvr.it/TQcHxX