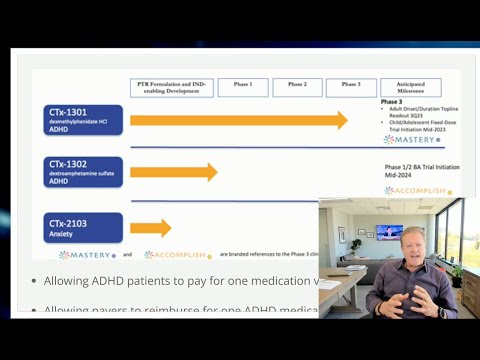
Exciting News from Cingulate: We’ve received a $4.3M PDUFA fee waiver from the FDA ahead of our imminent NDA submission for CTx-1301, our lead asset for the treatment of ADHD.
Learn more: finance.yahoo.com/news/cingulate…
#CTx1301 #ADHD #FDA #PDUFA #NDA #CING #PrecisionTimedRelease #PTR
English