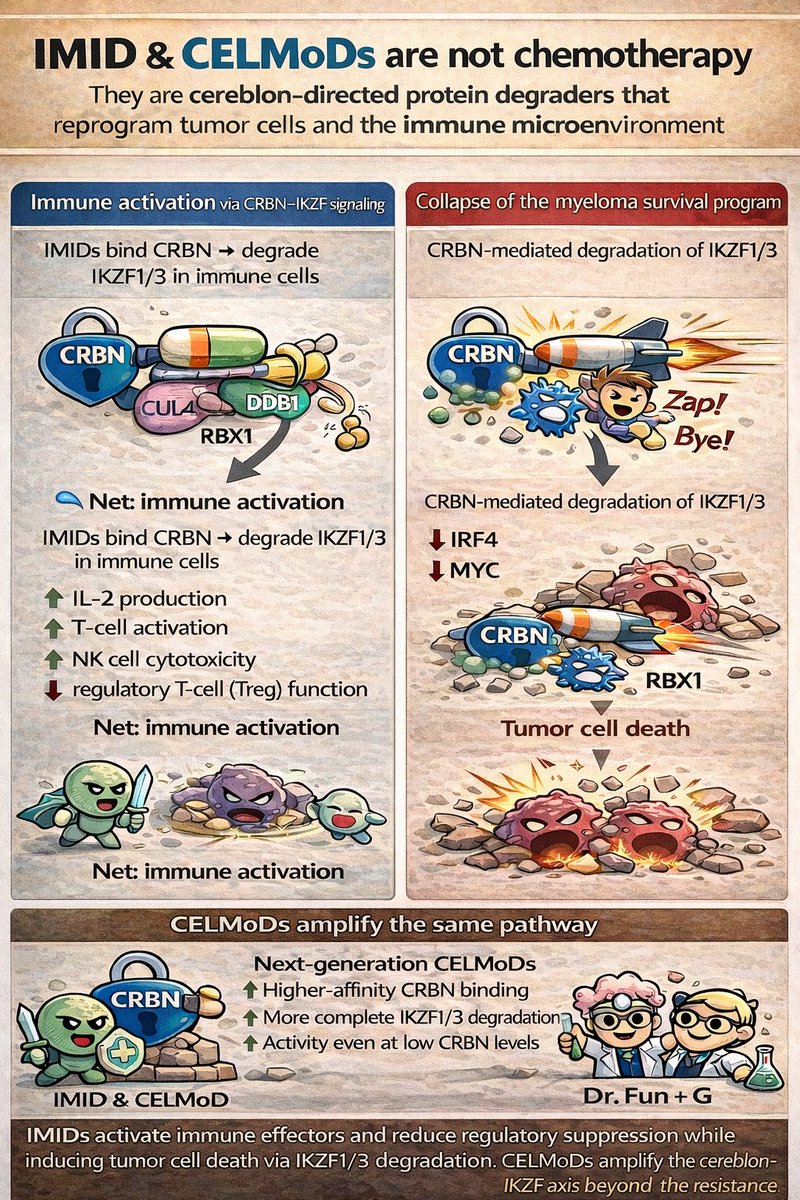
Jonas Moreno P retweetledi

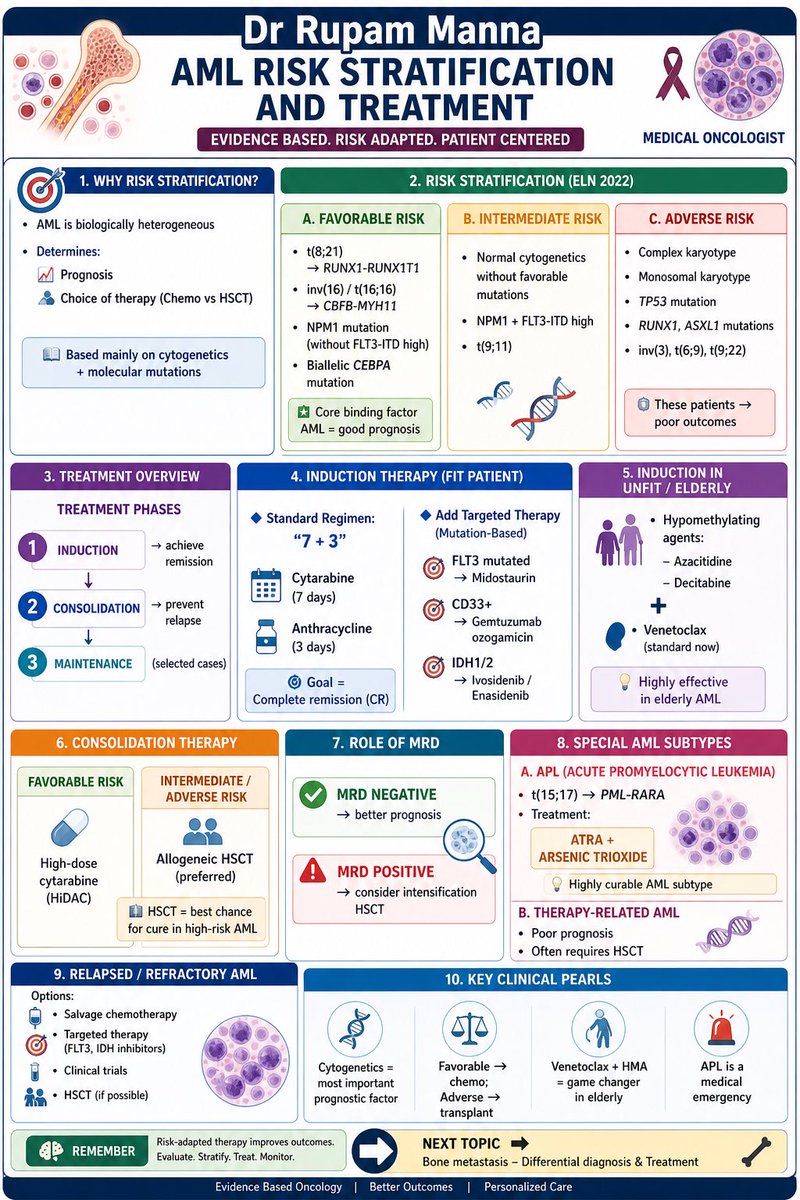
🚨 AML is NOT one disease — it’s a spectrum.
Risk stratification drives EVERYTHING:
🔬 Prognosis
💊 Treatment choice
🧬 Transplant decisions
From ELN 2022 risk groups → to 7+3, targeted therapy & HSCT, mastering this algorithm is key for every oncologist.
💡 Key takeaways:
✔️ Cytogenetics = backbone
✔️ Favorable → chemo
✔️ Adverse → transplant
✔️ Venetoclax + HMA = game changer
✔️ APL = oncologic emergency
📌 Save this for revision. Teach it. Use it.
— Dr Rupam Manna
#AML #Leukemia #Hematology #MedicalOncology #Oncology #CancerEducation #ELN2022 #MRD #HSCT #Venetoclax #MedEd #FOAMed
English