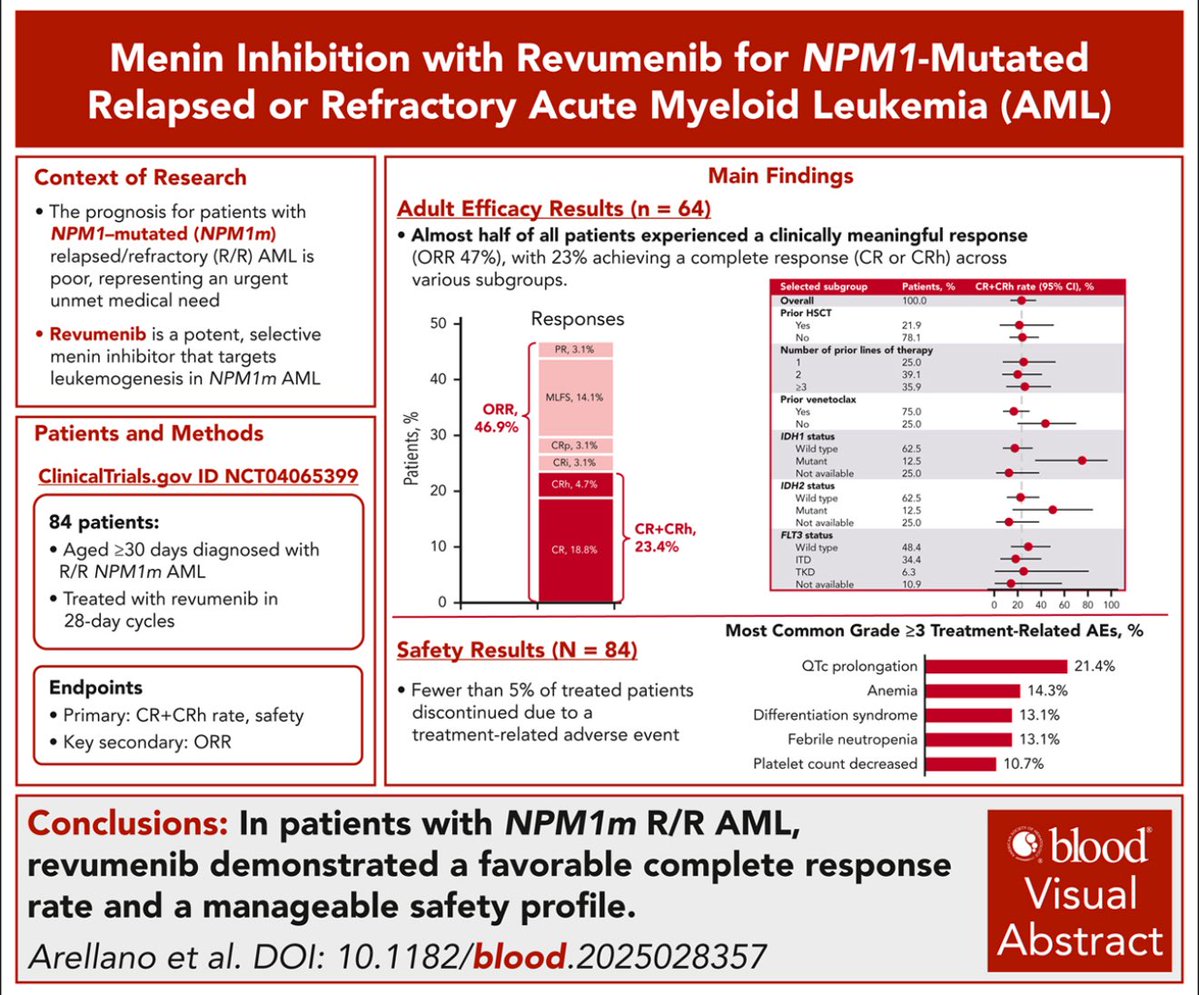
Kyle Gash, PharmD retweetledi

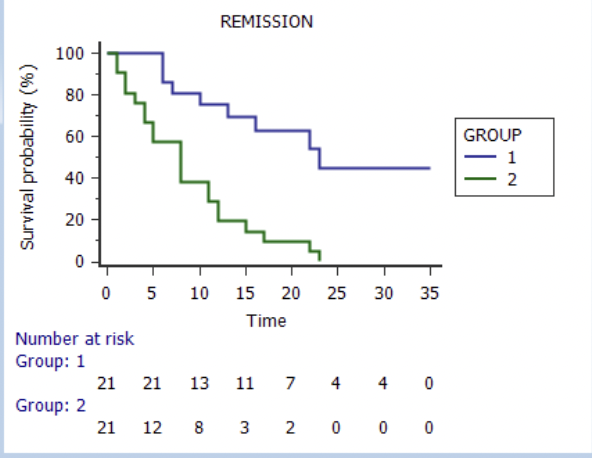
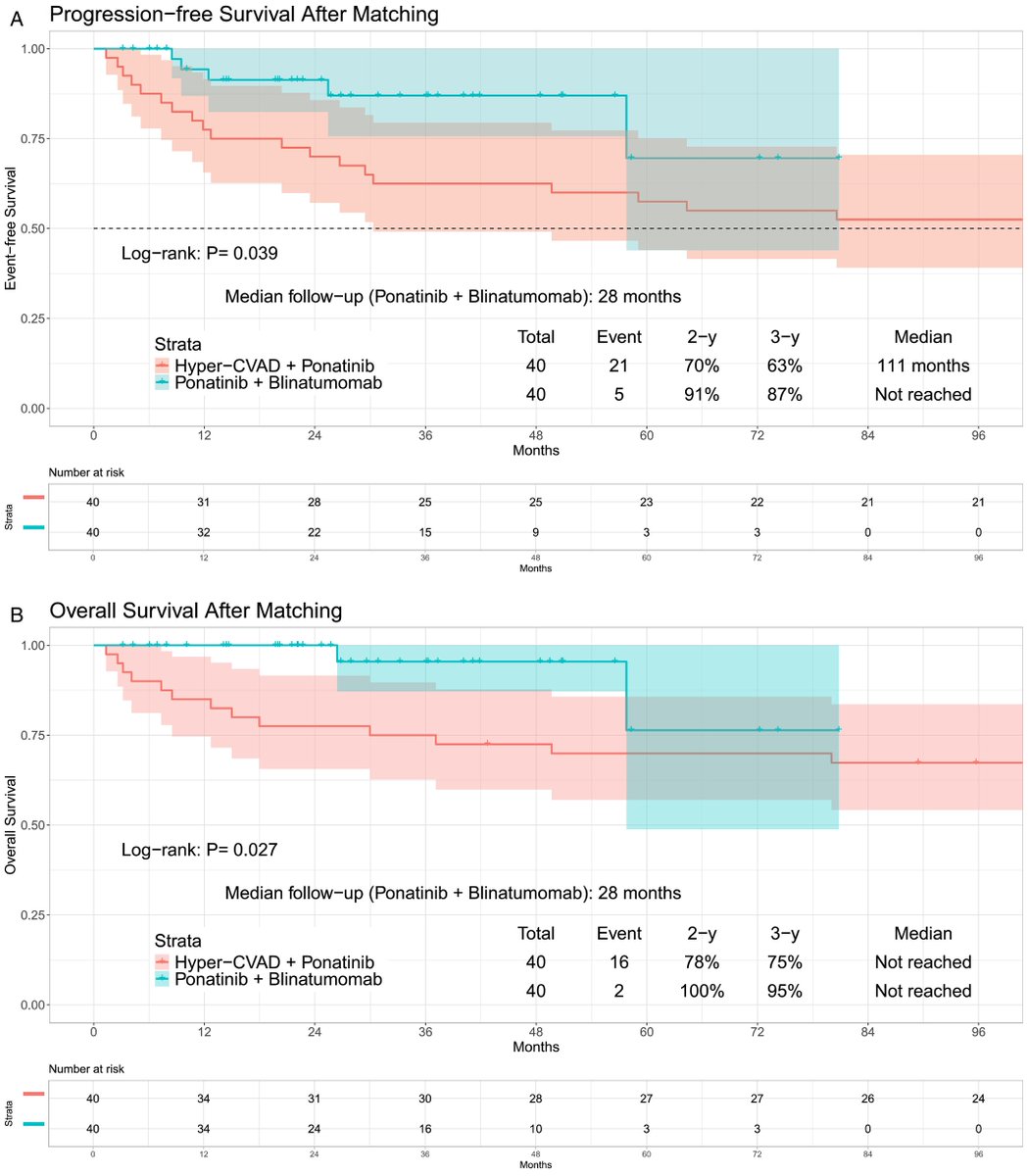
The chemotherapy-free regimen blinatumomab and ponatinib is associated with significantly improved PFS and OS compared to Hyper-CVAD with ponatinib, with less non-relapse morality and a reduced reliance on allo-SCT in Ph+ ALL.
doi.org/10.1002/ajh.70…
@AjHematology @UTMDAnderson
English