Lesley.SeymourMD
200 posts

Lesley.SeymourMD
@LSeymourMD
Medical Oncologist, Professor of Oncology Queens University, Director Investigational New Drug Program, Canadian Clinical Trials Group.
Kingston, Ontario Katılım Kasım 2017
53 Takip Edilen154 Takipçiler
Lesley.SeymourMD retweetledi

@CDNCancerTrials Wonderful to have Carol join us in early clinical trials research
English

Congratulations Joe - it’s been an enormous privilege to have (and continue to) work with you - your contributions to cancer research nationally and globally cannot be overstated
CCTG@CDNCancerTrials
Dr. Joseph Pater #CCTG's founding director recognized for a lifetime of work in cancer research with his appointment as an Officer of the Order of Canada - his clinical research has improved the lives of Canadians with cancer. #OrderOfCanada #GGHonours ctg.queensu.ca/cctg_news/foun…
English
Lesley.SeymourMD retweetledi

New feature ESMO-MCBS scorecards: visualization of the score. When interested, please give your feedback. @myESMO @curzua_28 @rsnfehrmann esmo.org/guidelines/esm…

English
Lesley.SeymourMD retweetledi

Pembro Plus Platinum-Based Chemotherapy Improves Survival in Advanced Pleural Mesothelioma ascopubs.org/doi/10.1200/AD…
English
Lesley.SeymourMD retweetledi

Dr Quincy Chu concluding his presentation of IND.227, a collaboration of
@istitutotumori
@IFCTlung
@CDNCancerTrials

English
Lesley.SeymourMD retweetledi

#ASCO23 is this weekend stop by and see @ALLIANCE_org NCCTG N0147 (#CCTG CRC2): Artificial intelligence–derived immune phenotypes for prediction of prognosis in patients with stage III colon cancer
Poster Session 3542: meetings.asco.org/abstracts-pres…

English
Lesley.SeymourMD retweetledi

If you are attending #ASCO23 make sure to visit this poster presentation - #CCTG CO26: Reversion of RAS mutations in metastatic colorectal cancer in the CCTG CO26 clinical trial.
Poster Session 3567: meetings.asco.org/abstracts-pres…
#SolvingCancerTogether

English
Lesley.SeymourMD retweetledi
Lesley.SeymourMD retweetledi
Lesley.SeymourMD retweetledi

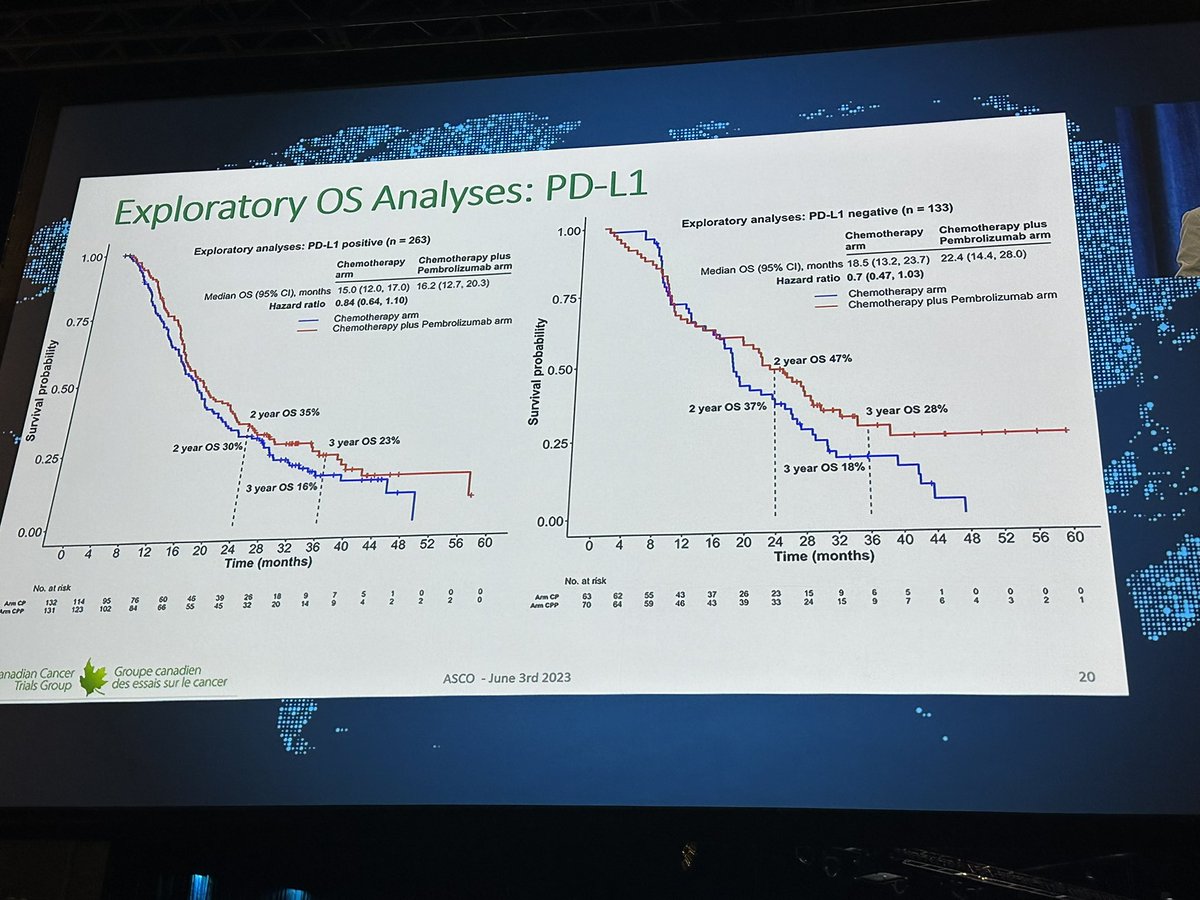
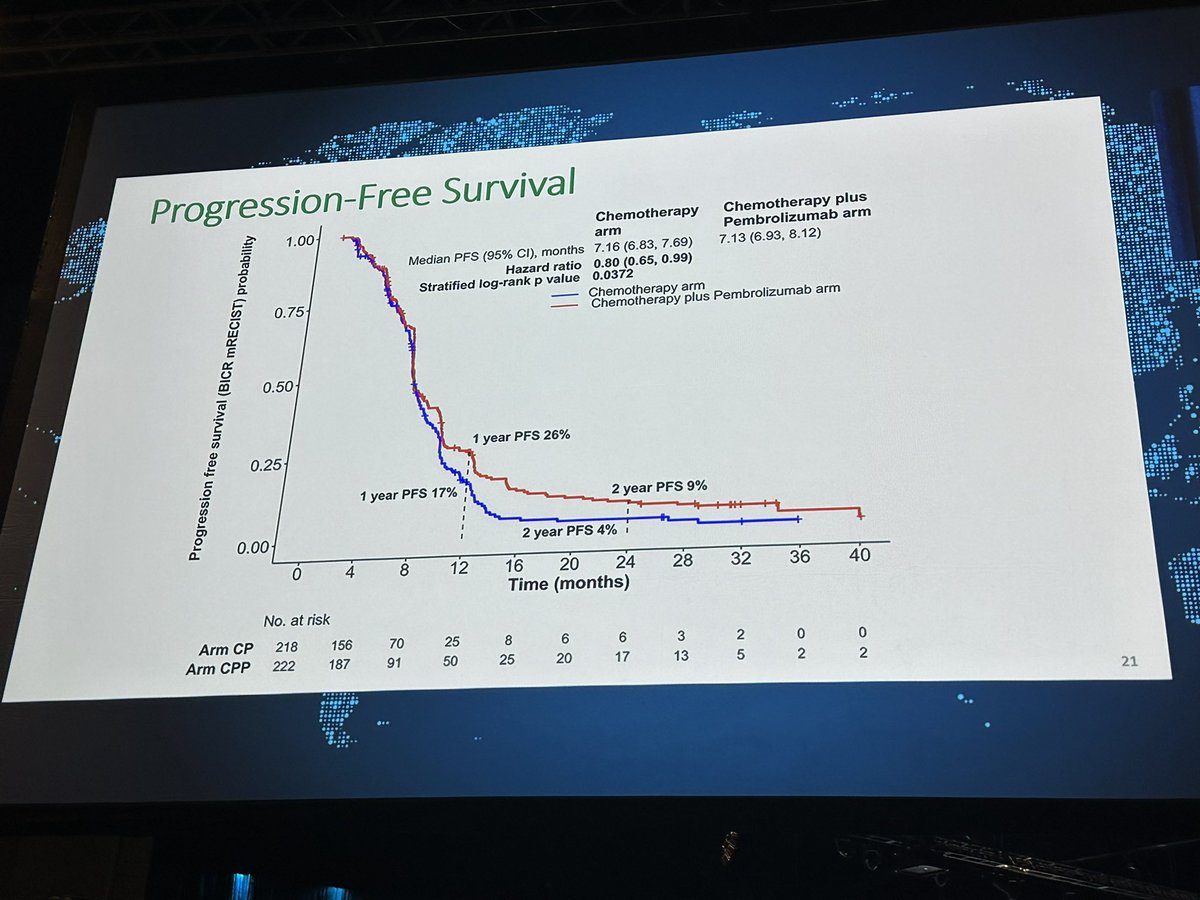
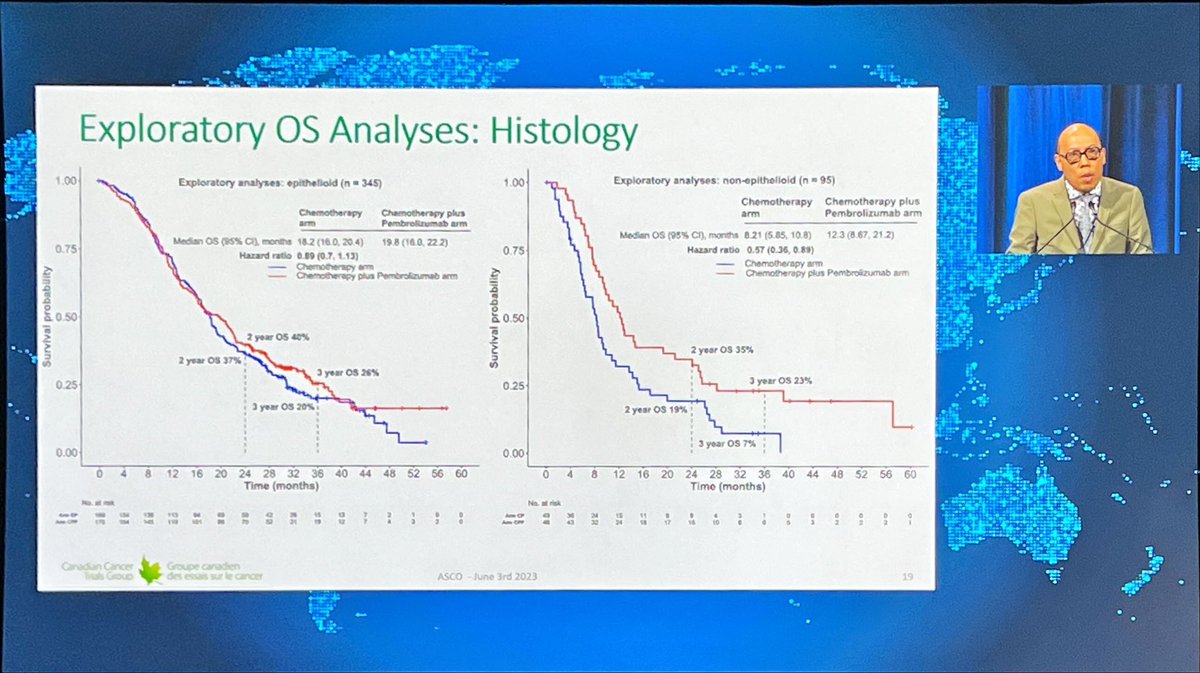
🧐IND.227: fase III randomizado comparando CIS/PEM X CIS/PEM/PEMBRO. N=440 ⬆️OS HR: 0,79. >>Impacto histologia ñ-epitelioide e > EORTC prognostic risk, contudo mediana numericamente inferior ao CK743. ↔️PFS ↔️TRO #ASCO23
Português
Lesley.SeymourMD retweetledi

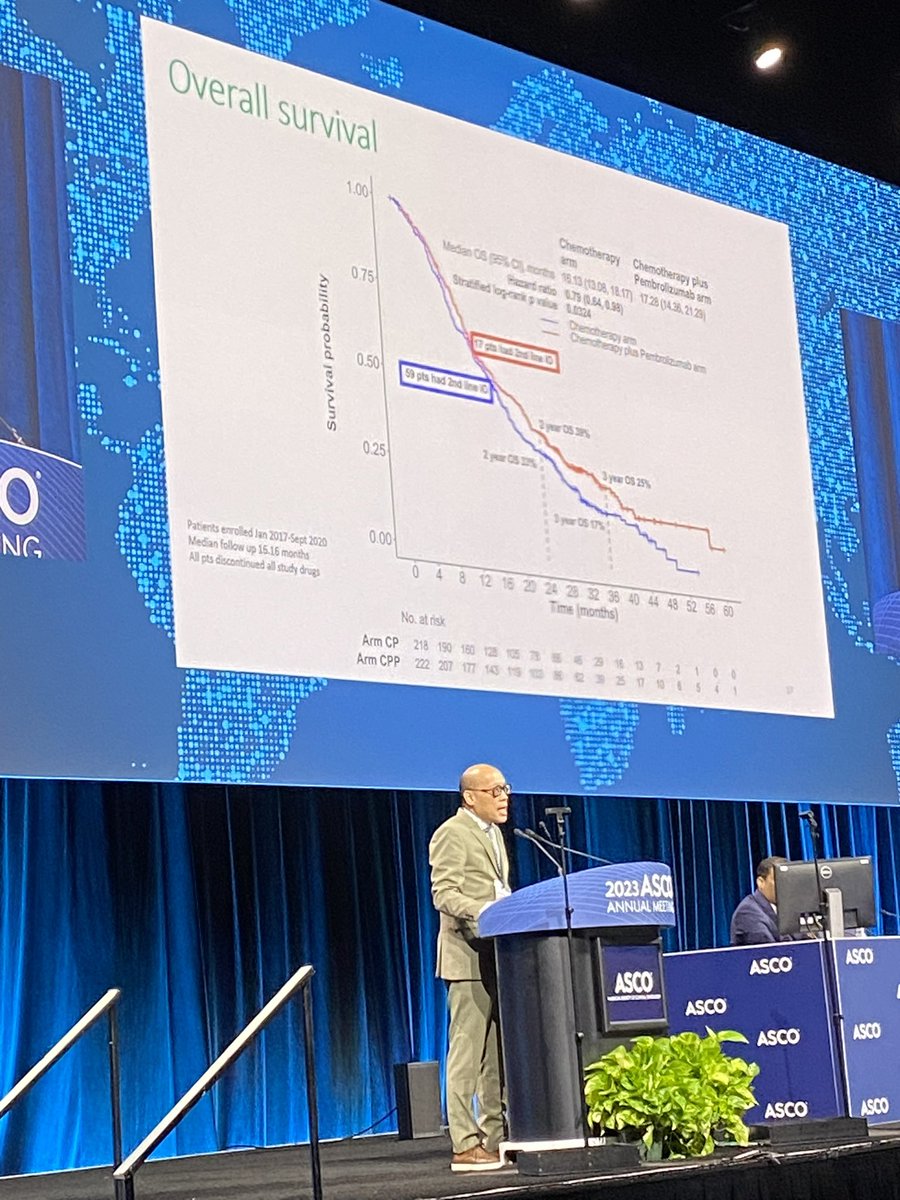
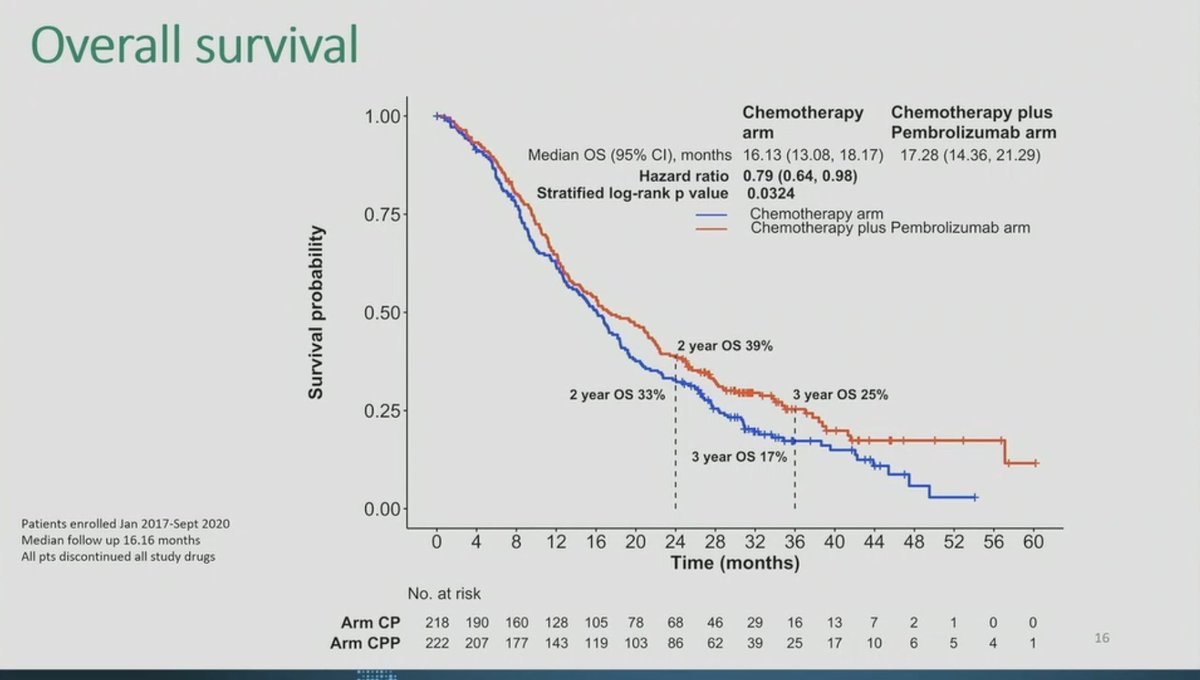
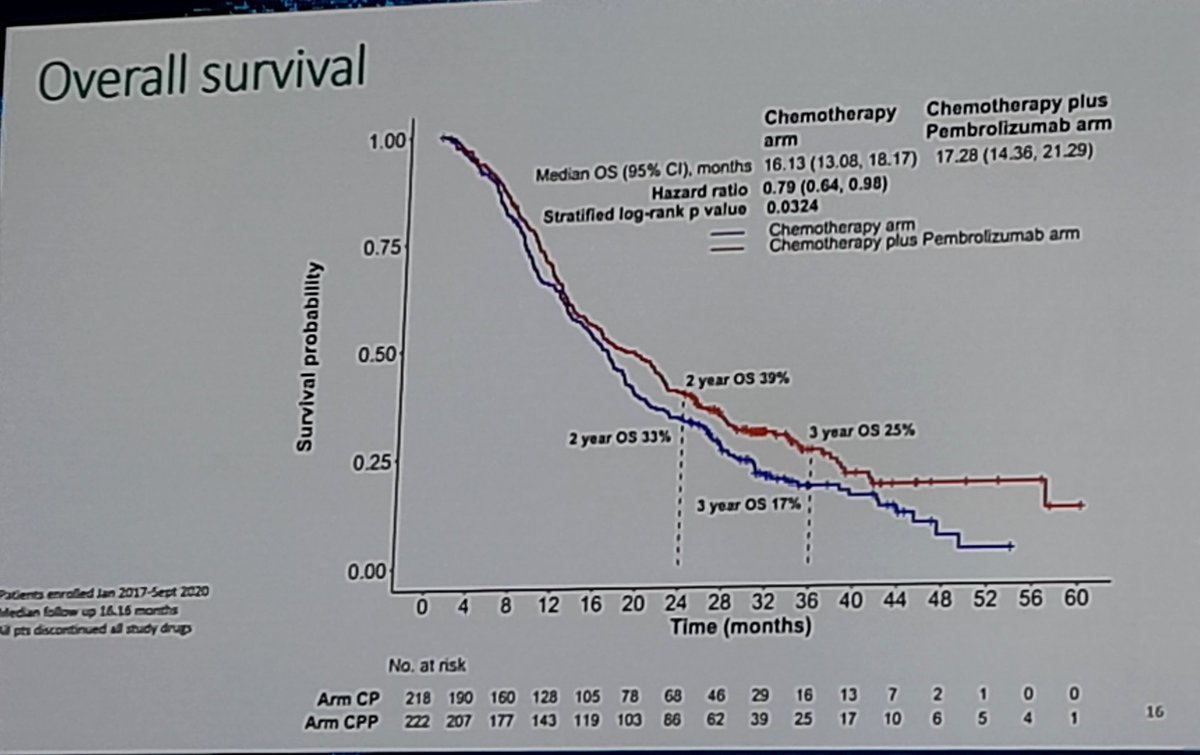
IND.227: PhIII, Platinum + Pem + Pembro x Platinum + Pem in mesothelioma. mOS 17.28 x 16.13 months, HR 0.79, p 0.0324. Pronounced benefit in patients with non-epithelial subtypes. #ASCO23 #LCSM @GBOT_Alerta
English
Lesley.SeymourMD retweetledi

Novità per il trattamento del #mesotelioma dai risultati dello studio IND.227, presentati ad @ASCO. L’articolo: medinews.it/comunicati/i-r…
#ascodailynews #ASCO23 #specialeASCO23

Italiano
Lesley.SeymourMD retweetledi
Lesley.SeymourMD retweetledi

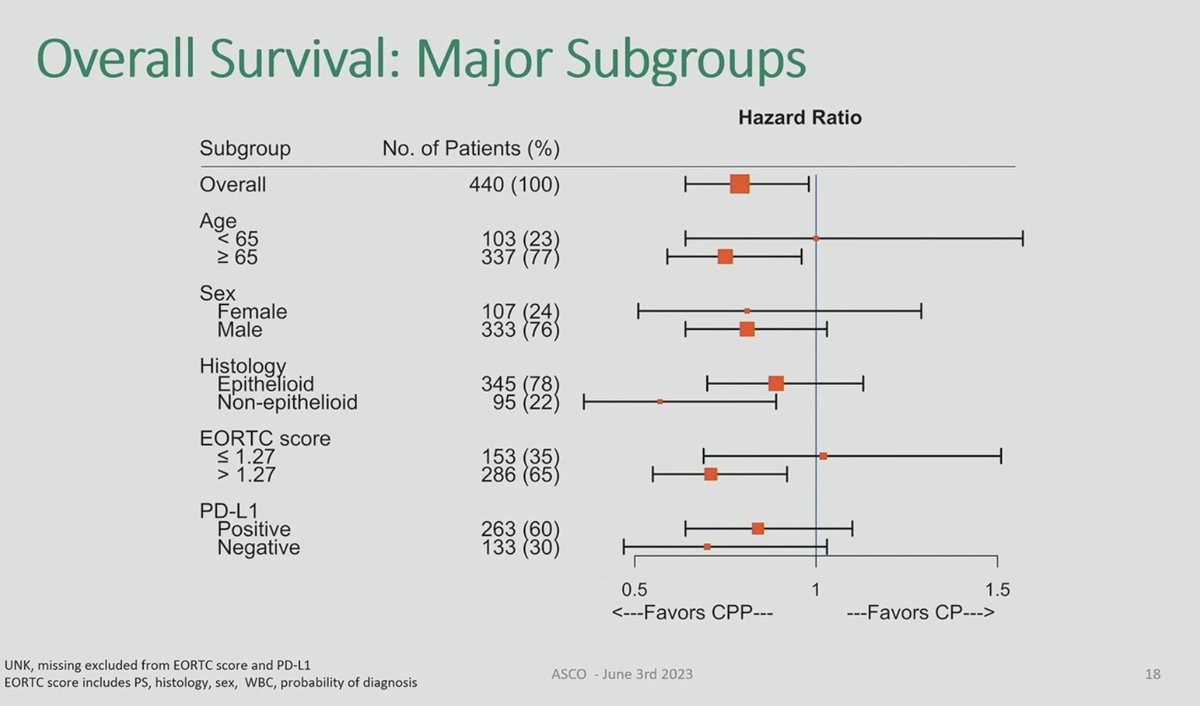
Great to see IND.227: long awaited. Chemo-pbro vs chemo. Mostly epithel subtyp. 61% CPS+ OS improved HR0.79, 16 to17mo median; PFS benefit. CPS not predictive.
Great work @CDNCancerTrials
Will the addition of beva improve this? Let’s see… #ASCO23 @etop_ibcsg


English
Lesley.SeymourMD retweetledi

IO+Chemo work in all thoracic 🫁 malignancies including pleural mesothelioma: IND227 trial. Results mirror CheckMate 743. Today the dilemma in 1st L: IO+IO vs IO+CT?
We need predictive mrk for choosing 1st L or decide based on histology subtype:IO+IO non-epithelioid? #ASCO2023



English