

MitohormesisClub
18.9K posts


@MitohormesisAct
I help high-performers reclaim 2 hrs of deep sleep in 30 days using sunlight & quantum biology. DM “SUN” for free Light Audit. #CircadianCEO #QuantumSleep”



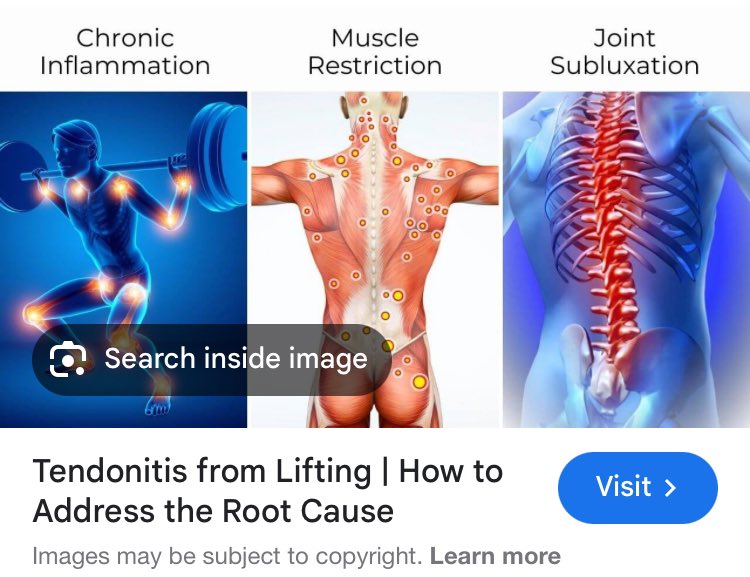
Intrinsic factors: Diabetes, metabolic syndrome, menopause, certain medications (fluoroquinolone antibiotics), genetics — all impair tendon health. This is the primary reason menopausal women see a significant increase in tendon pain. The tendon's biology is not isolated from the body's systemic environment. Glucose and glycated end products (AGEs) are serious troublemakers... same with uric acid.




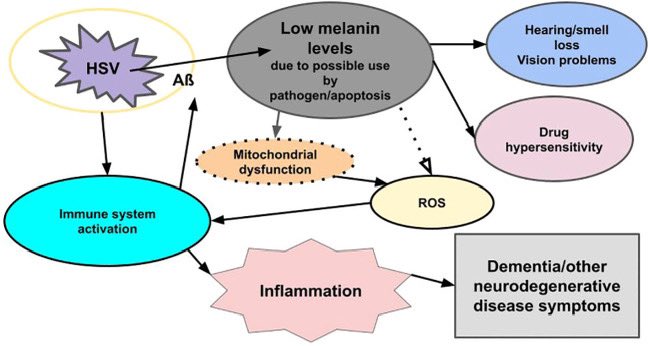

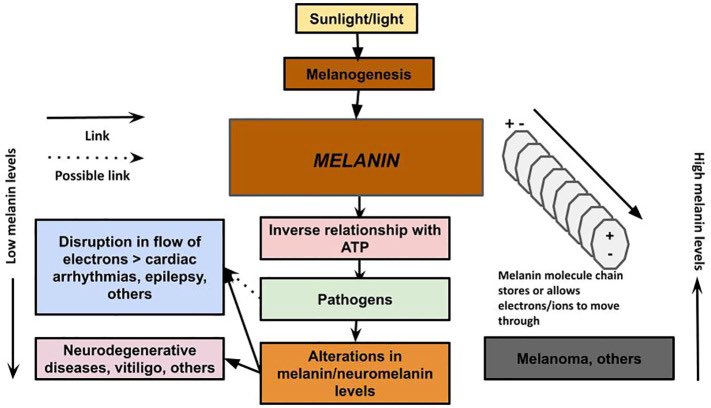
New research finds that depression is strongly linked to both Parkinson’s and Lewy body dementia and may presage a diagnosis years in advance. Depression is often a harbinger of toxin-related neurodegeneration, such as we see in type 3 Alzheimer’s disease. sciencealert.com/depression-may…






Ozempic has a new side effect: divorce New research shows people on ‘fat jabs’ are twice as likely to end their marriages. What’s going on?












Sleep and schizophrenia spectrum sciencedirect.com/science/articl…

Sam Altman has admitted he is on a waitlist for a procedure that would digitize his brain. The procedure would kill him. He considers this an acceptable trade for digital immortality. This is the person making decisions about the future of artificial intelligence for hundreds of millions of users. A man who views ending his own biological life as a reasonable step toward uploading his consciousness to the cloud. These are not the priorities of a stable leader.


The Trump administration has announced it plans to dismantle the US Forest Service. All 10 regional offices are set to close in addition to over 50 research labs.

