
OPDP Resources
4K posts


OPDP Resources
@OPDPresources
Instantly search all OPDP/DDMAC enforcement letters since 1997 with curated filters and smart summaries. https://t.co/7kZY1ILJPp





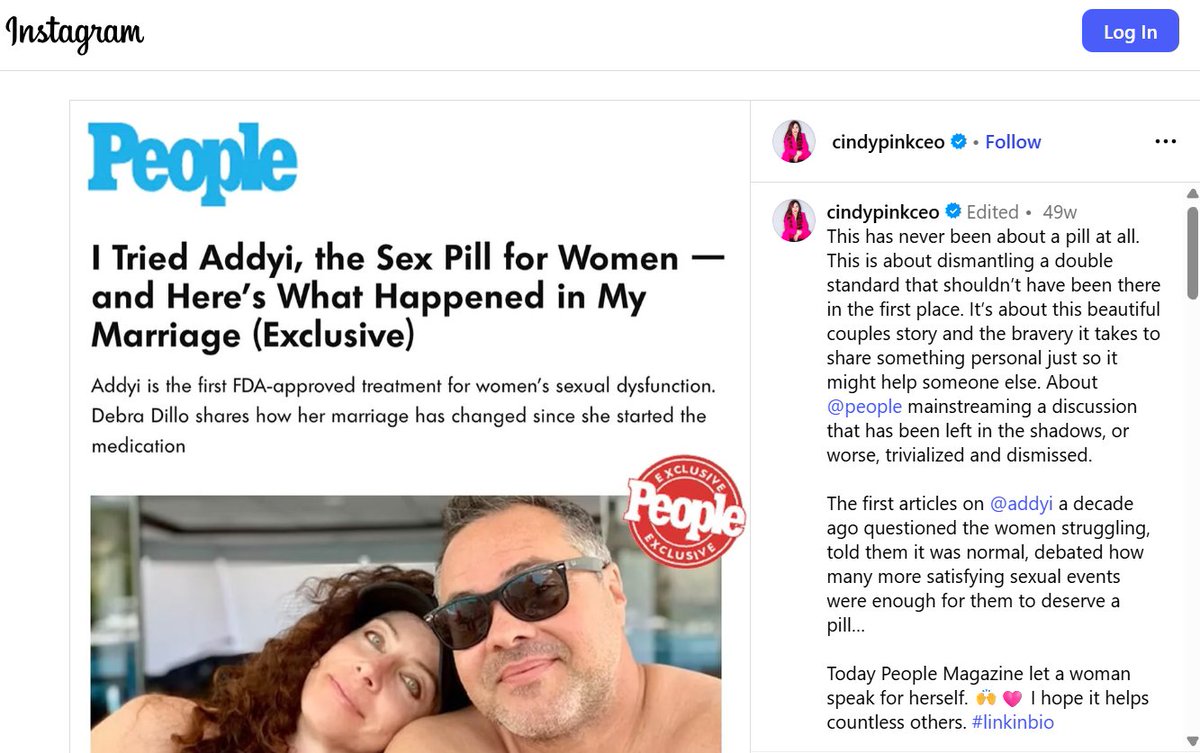
Today we’re taking long-overdue action to rein in pharmaceutical ads. For too long, misleading ads have distorted the doctor-patient relationship and driven costly, inappropriate care. Our actions will encompass online pharmacies promoting drugs with no mention of side effects, and paid social media influencers advertising drugs.



A massive, massive deal. Industry, watchdogs, lawyers and media have been wanting access to these for years. I'm not sure how FDA's lawyers suddenly decided that they had the legal authority to release these, but it's going to be hard to put the genie back in the bottle.


Just published a rather significant scoop for readers of AgencyIQ's "FDA Today" newsletter: The top leaders of FDA's drug advertising division, known as OPDP, are stepping down from the agency. Catherine Gray and Mark Askine informed FDA staff yesterday that they are leaving.













