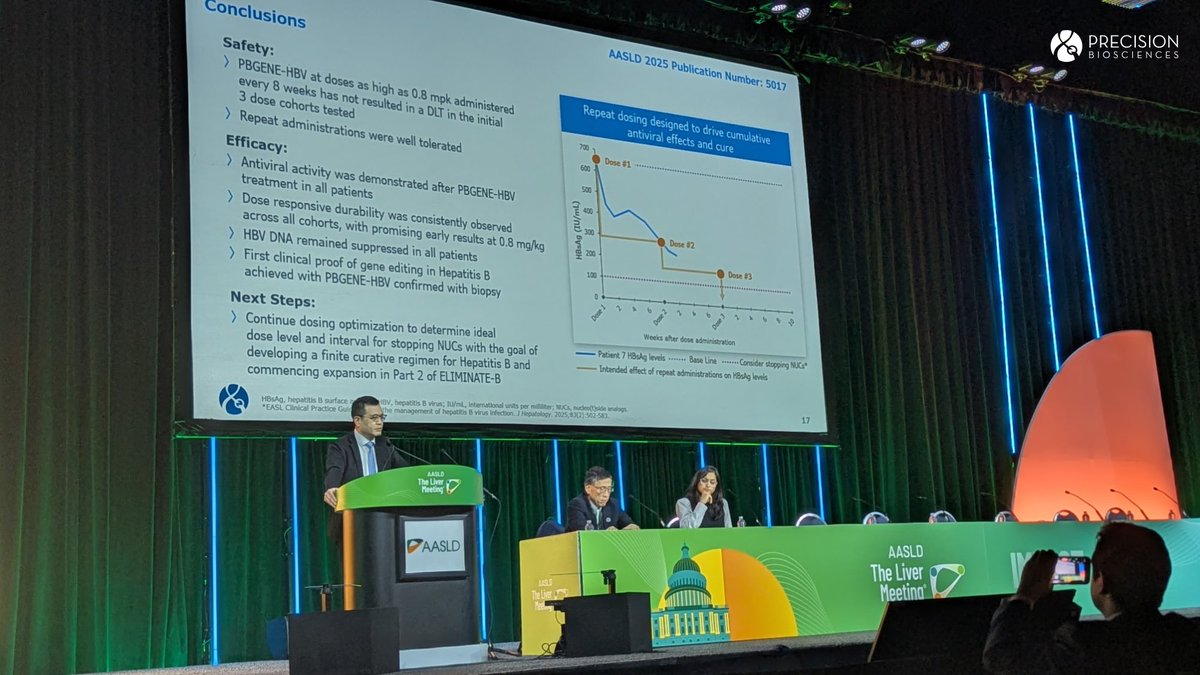
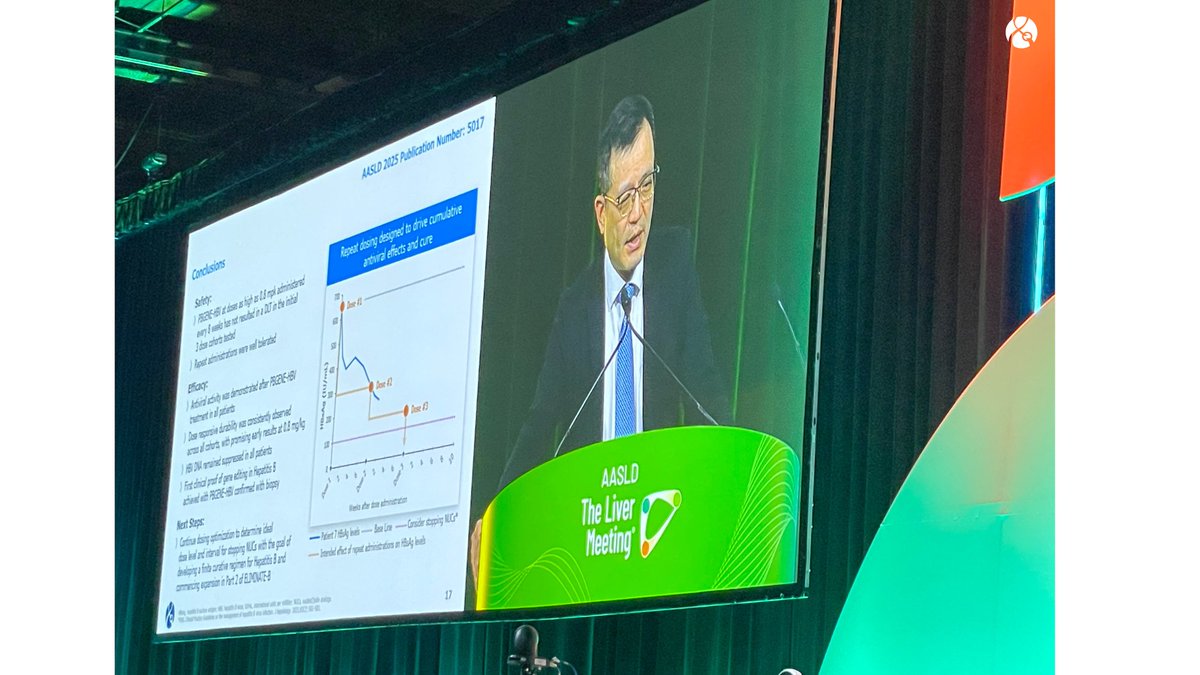
Precision BioSciences announced its Q4 and fiscal year 2025 earnings and provided a business update!
CFO, Alex Kelly, tells you what you need to know.
Read the full press release, investor.precisionbiosciences.com/news-releases/…
#GeneEditing #Biotech $DTIL
English