
Research Analyst @Shea_ARK shares her top 3 takeaways from the Multiomics section of ARK's Big Ideas 2026 report.
Shea Wihlborg
398 posts


@Shea_ARK
Multiomics analyst @ARKInvest focused on identifying disruptive innovations that could reshape the future of medicine | Disclosure: https://t.co/sPOP1gVoCR

Research Analyst @Shea_ARK shares her top 3 takeaways from the Multiomics section of ARK's Big Ideas 2026 report.


ARK’s Big Ideas filters out short-term noise to reveal the signals we believe matter most for long-term growth. Explore the themes in Big Ideas 2026 by watching a detailed, section-by-section analysis from the ARK research team. ark-invest.com/videos/analyst…




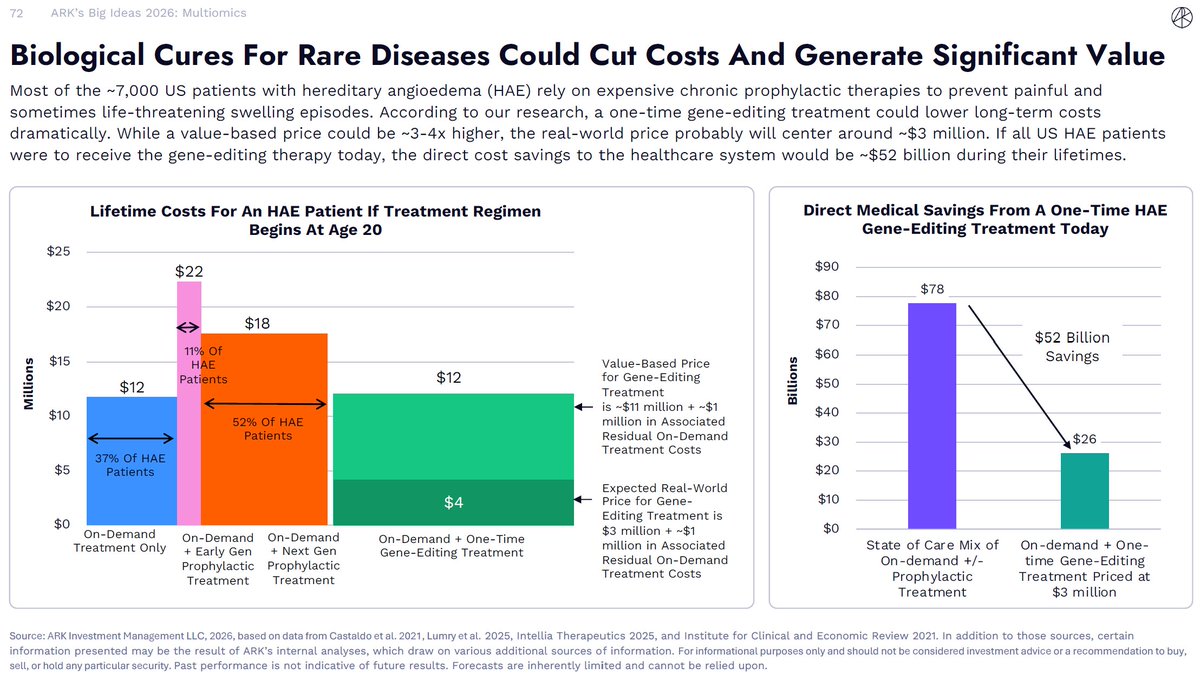
If you could choose—would you prefer a one-time treatment or lifelong chronic therapy? Innovations in gene-editing medicine could enable one-time treatments for certain rare diseases. While these therapies are likely to carry higher upfront prices than chronic treatments, they could also improve patient outcomes and lower long-term healthcare costs. This Big Ideas 2026 slide examines hereditary angioedema (HAE), where emerging clinical data indicate that a one-time gene-editing therapy could more effectively prevent painful—and sometimes life-threatening—attacks than chronic treatment. Based on modeled pricing assumptions, our analysis suggests this approach could also reduce lifetime medical spending despite a higher initial cost. ark-invest.com/big-ideas-2026




Don’t chase what was. Own what’s next. Big Ideas 2026. Download now: ark-invest.com/big-ideas-2026

You know your chronological age—but how old are you biologically, and how well can we measure it and assess mortality risk? Progress in medicine has meaningfully extended lifespans by reducing early deaths from infectious and chronic diseases. As a result, mortality is now increasingly concentrated at older ages. As understanding of the biology of aging advances, our ability to measure biological age and assess risk of mortality and functional decline has improved as well. This Big Ideas 2026 slide illustrates how approaches to measuring aging have evolved—from basic clinical markers to functional tests, molecular clocks, and digital aging measures—sharpening precision in how biological aging and mortality risk are quantified. ark-invest.com/big-ideas-2026



Don’t chase what was. Own what’s next. Big Ideas 2026. Download now: ark-invest.com/big-ideas-2026

A $2.8T market hiding in plain sight? If CRISPR captures just 8% of its total addressable market, it would rival Lipitor’s 20-year run of success. Watch the full episode of "The Brainstorm" with @Shea_ARK → ark-invest.com/podcast/the-br…

Gene editing is moving from rare diseases to major illnesses like heart disease. This could reshape the entire healthcare industry. What does this shift means for the future of medicine and long-term investment opportunities? Watch this week's episode of The Brainstorm. ark-invest.com/podcast/the-br…




