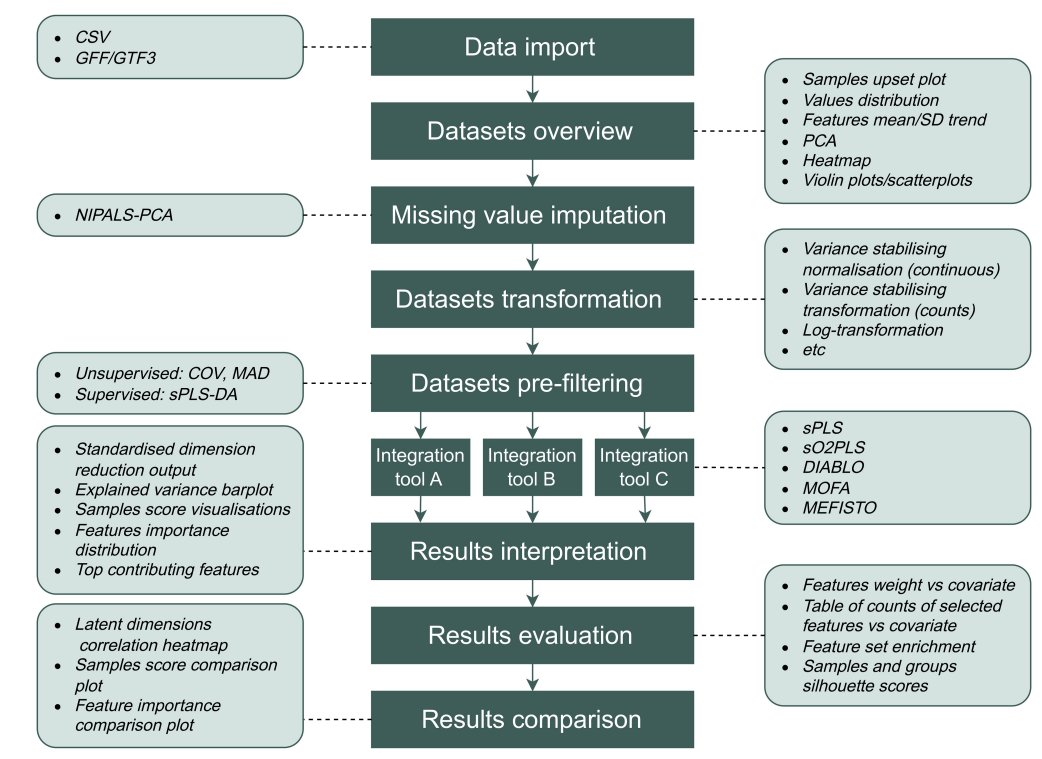
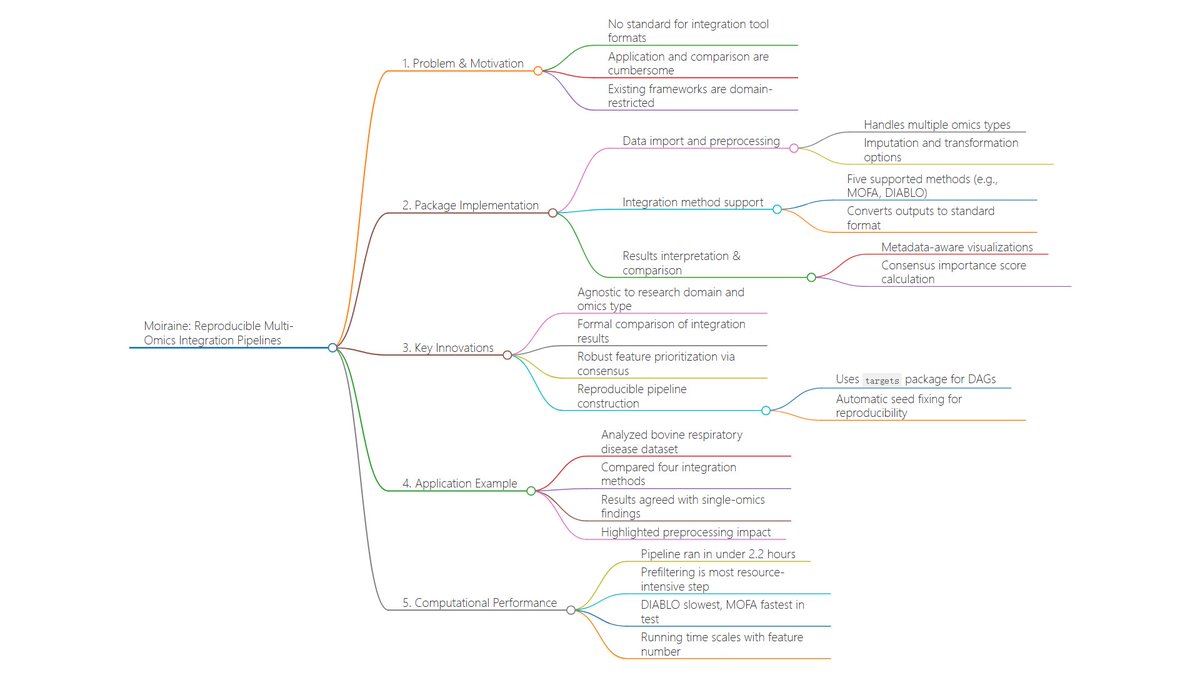
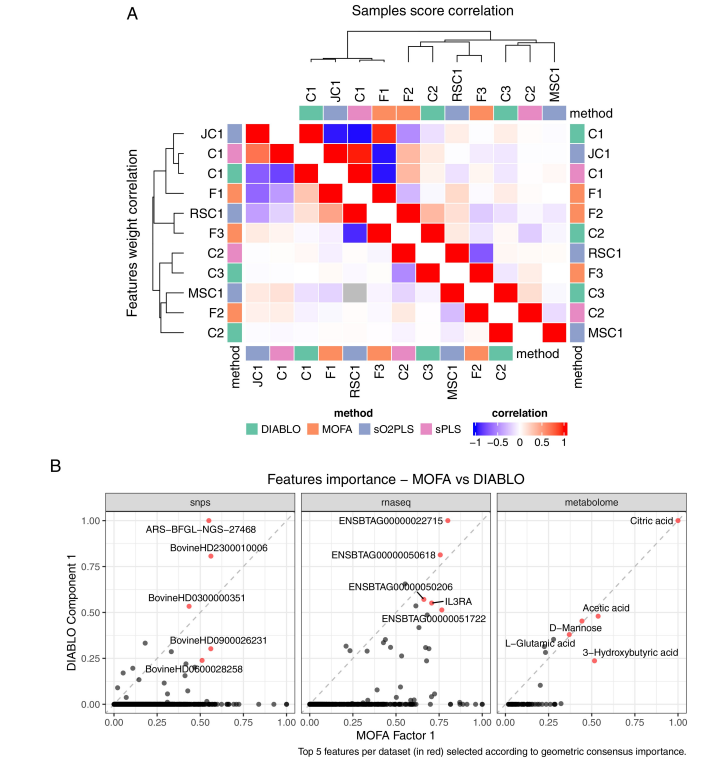
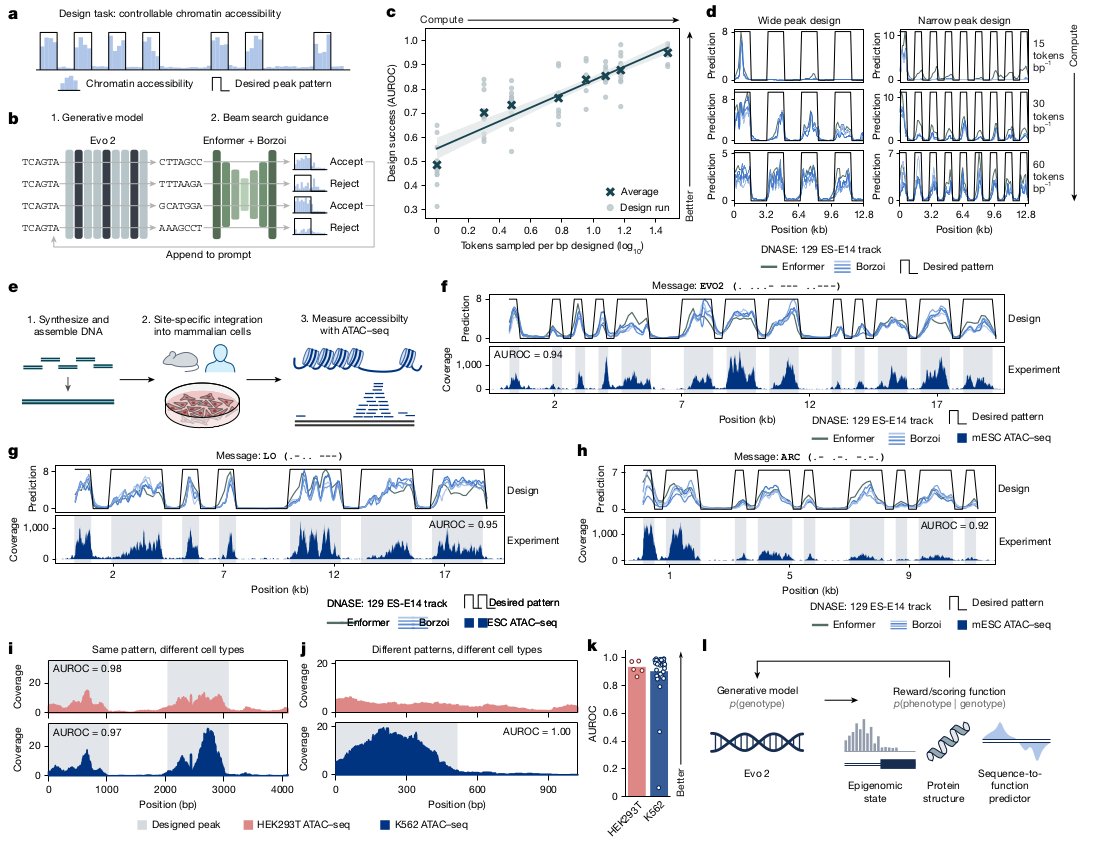
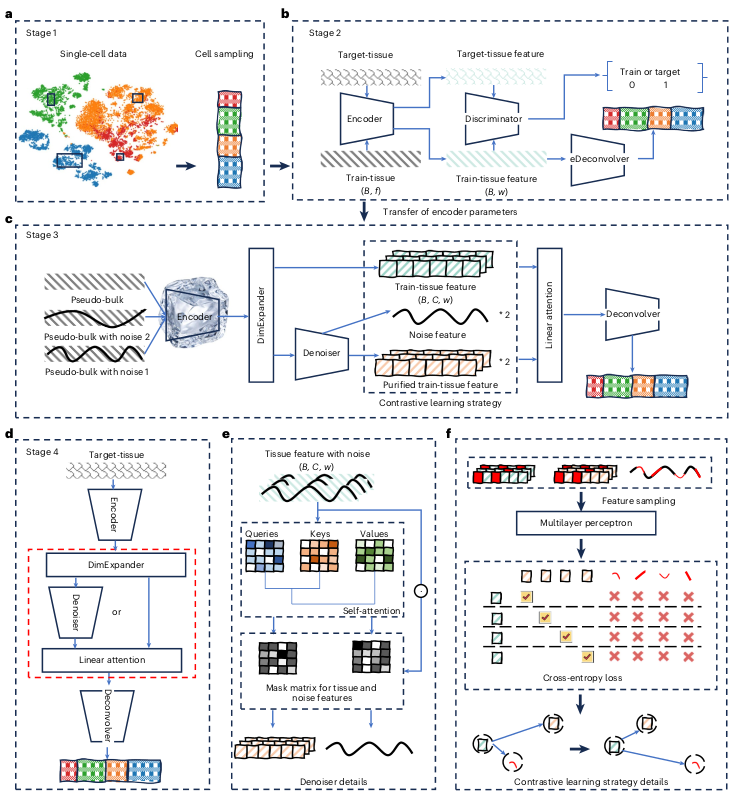
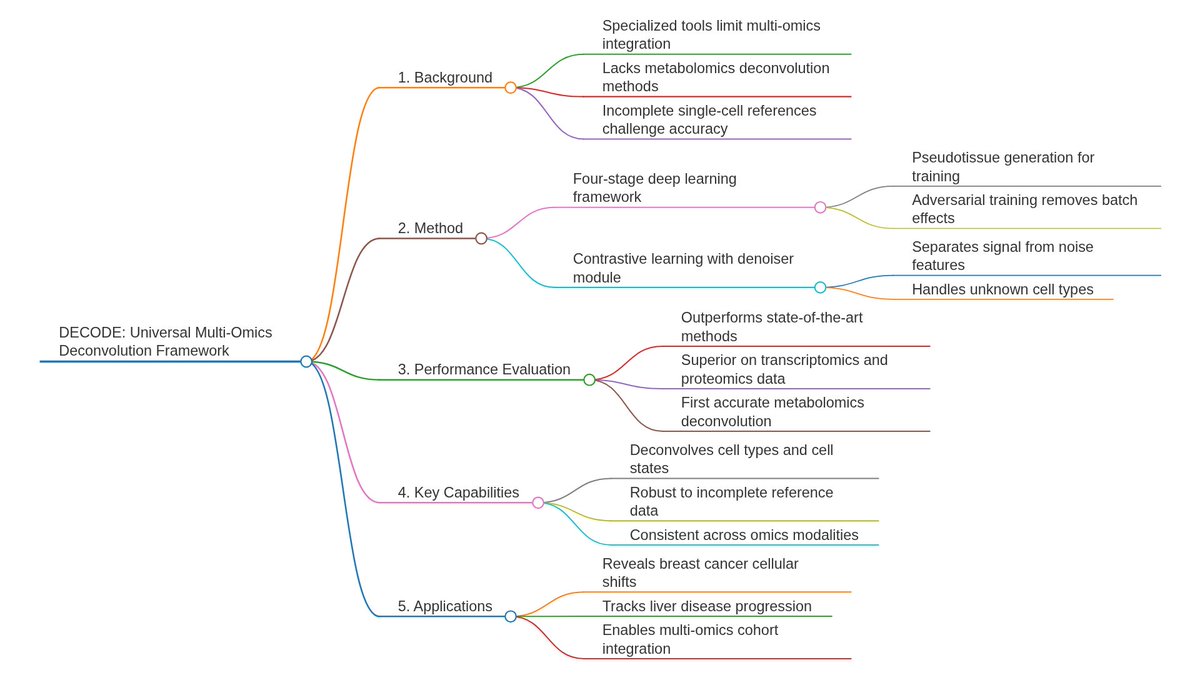
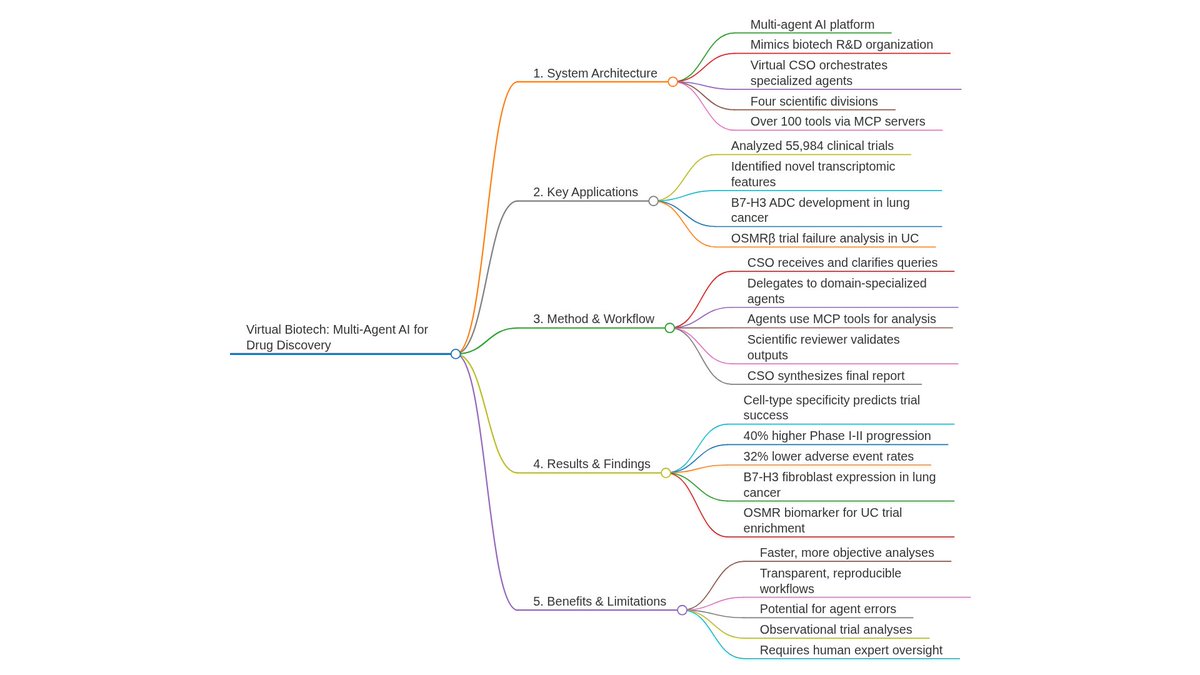
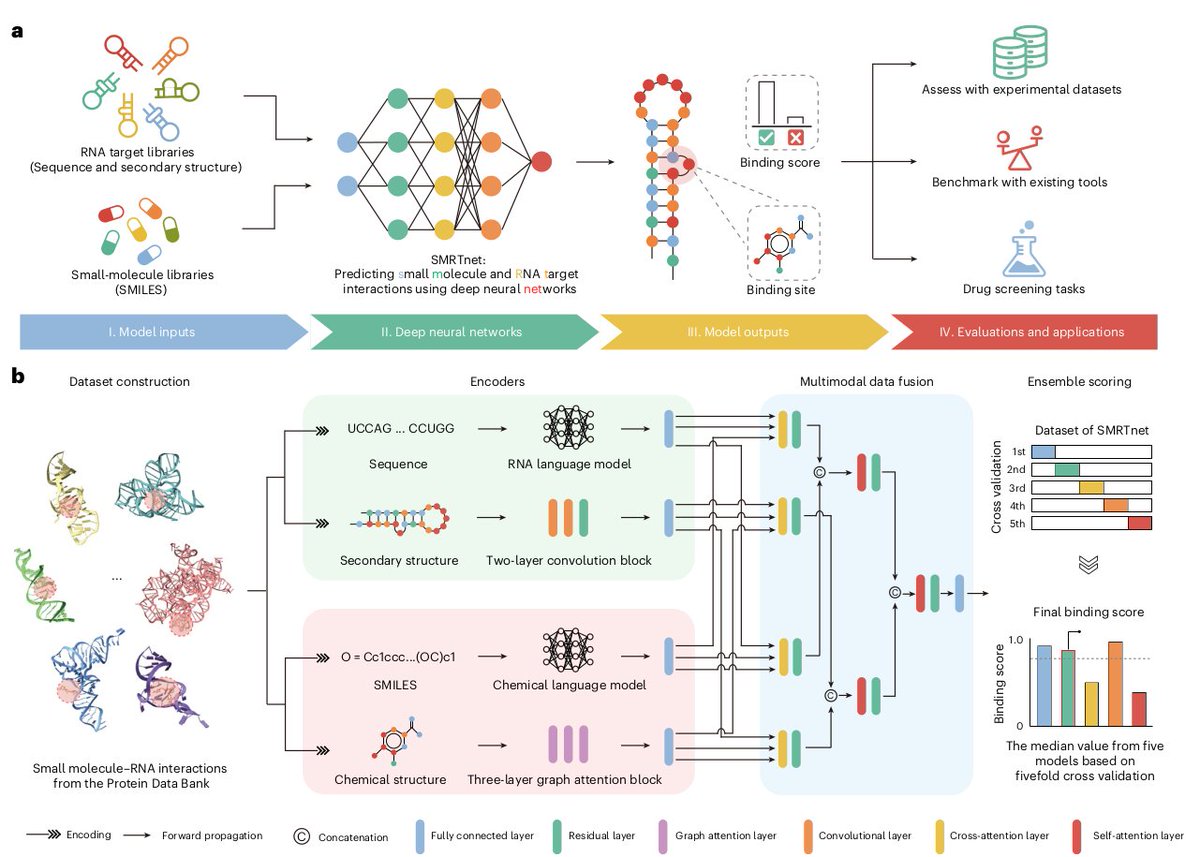
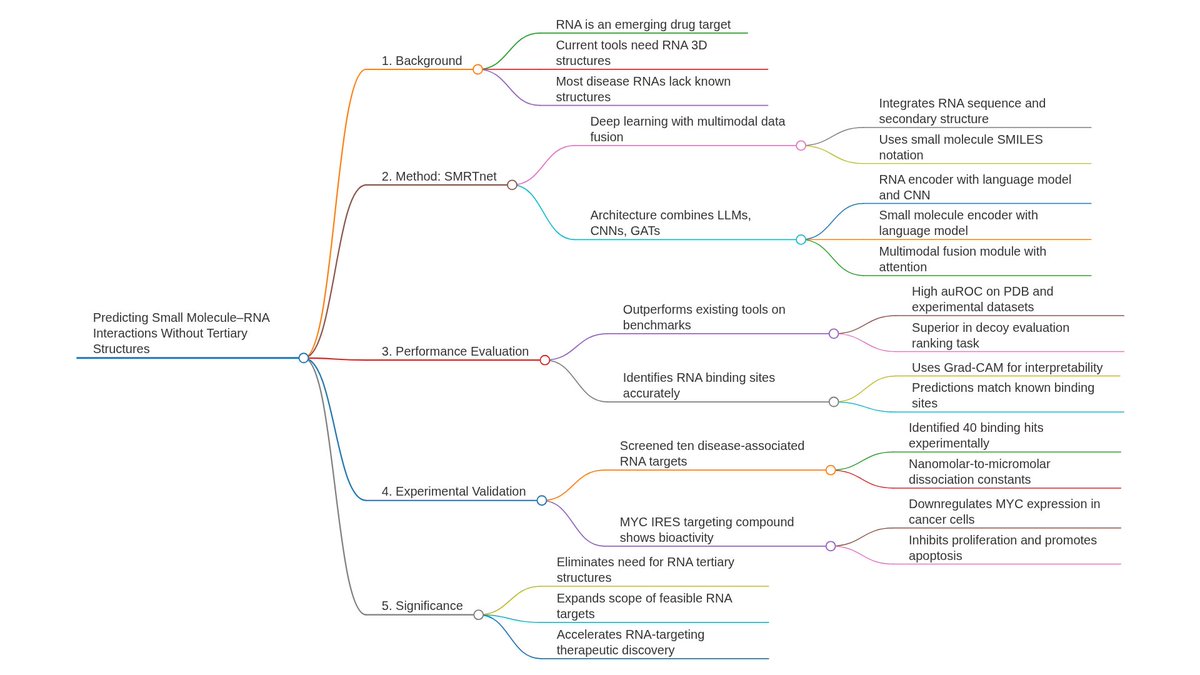
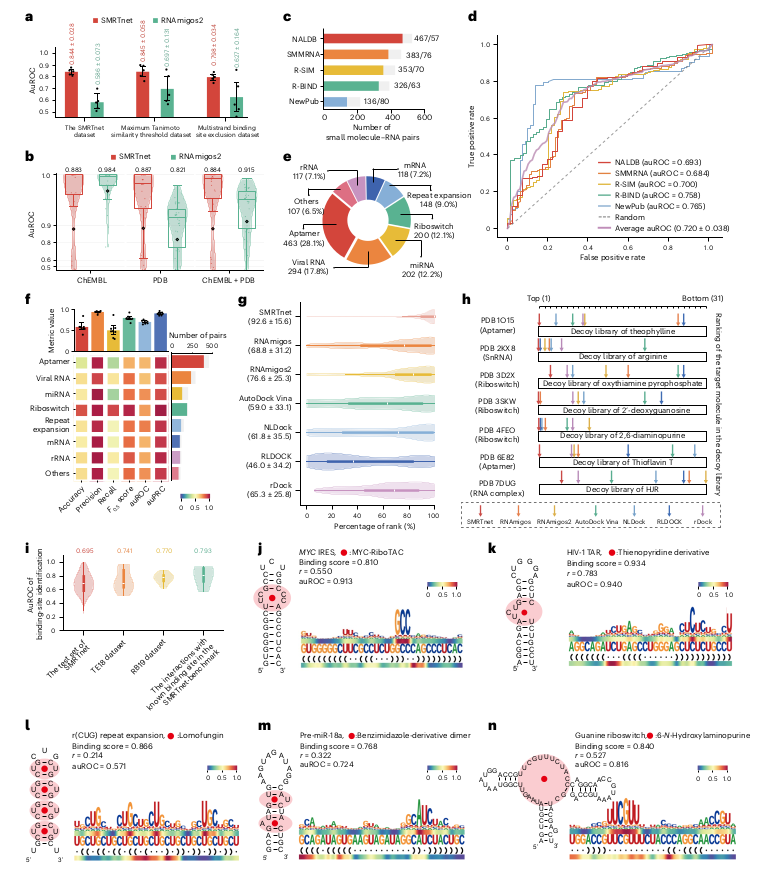
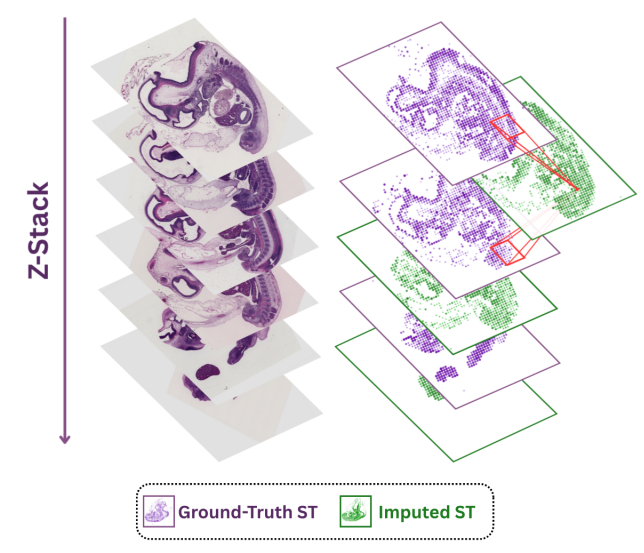
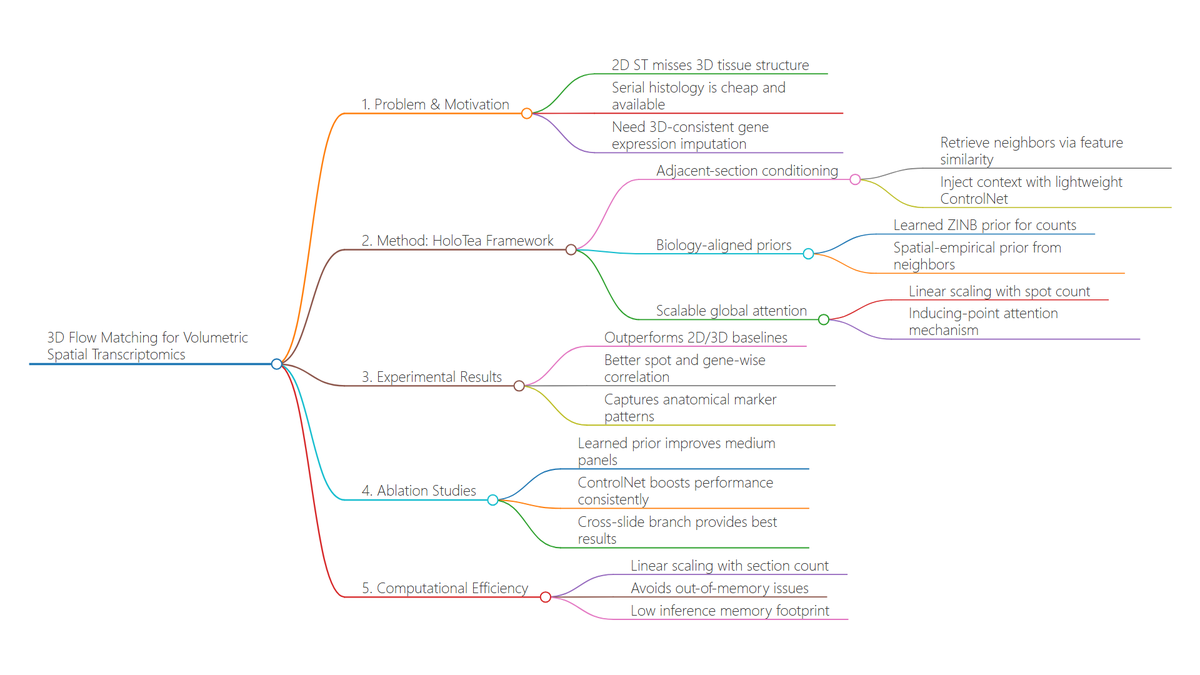
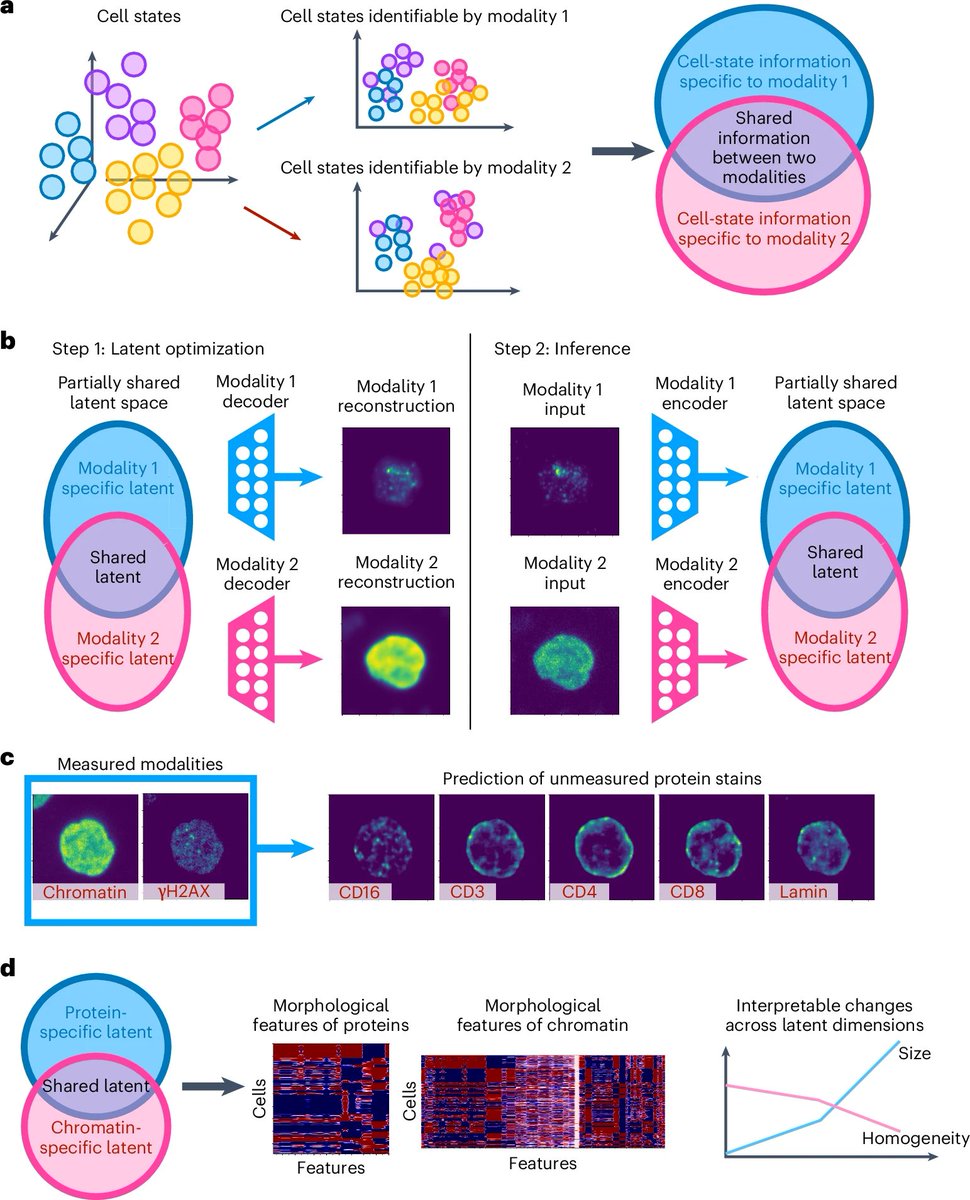
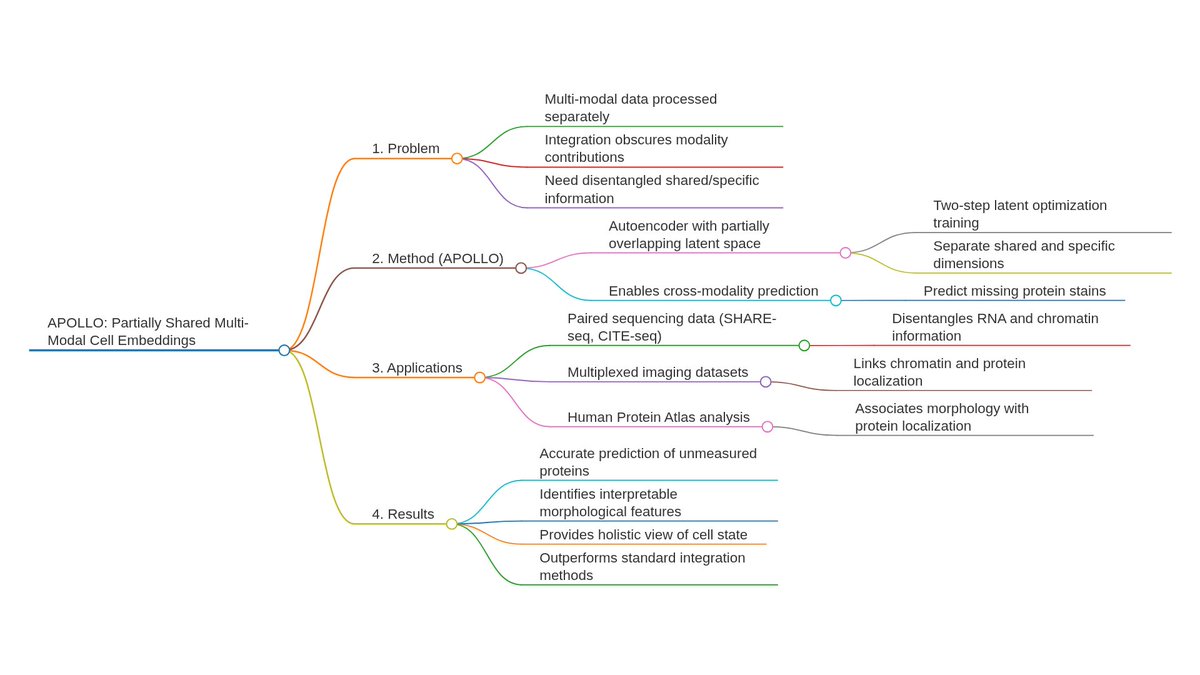
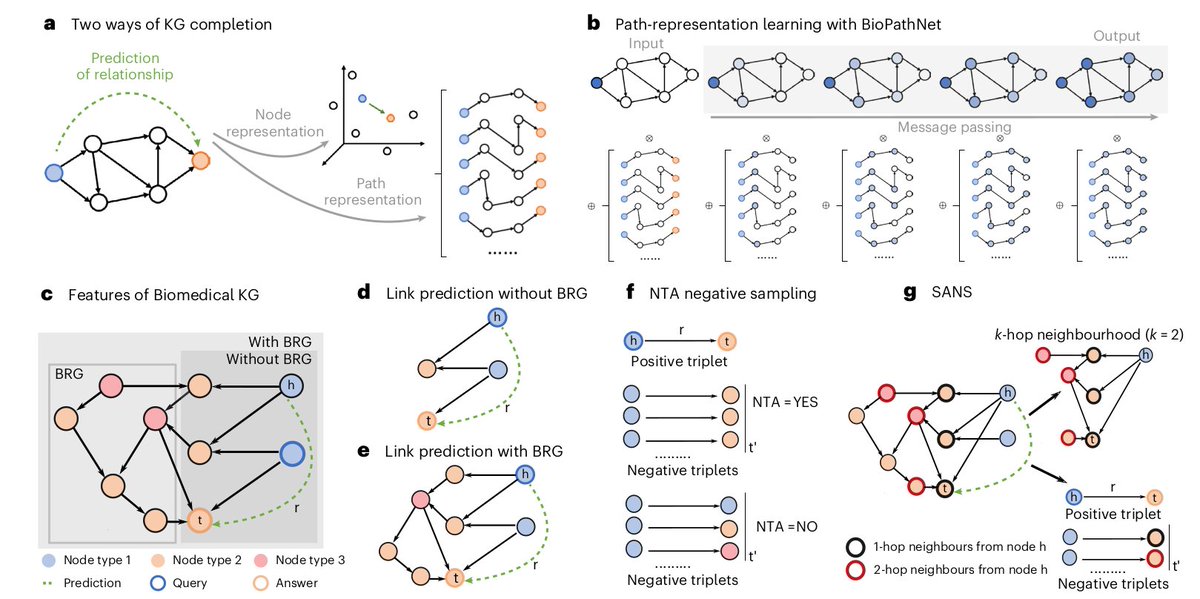
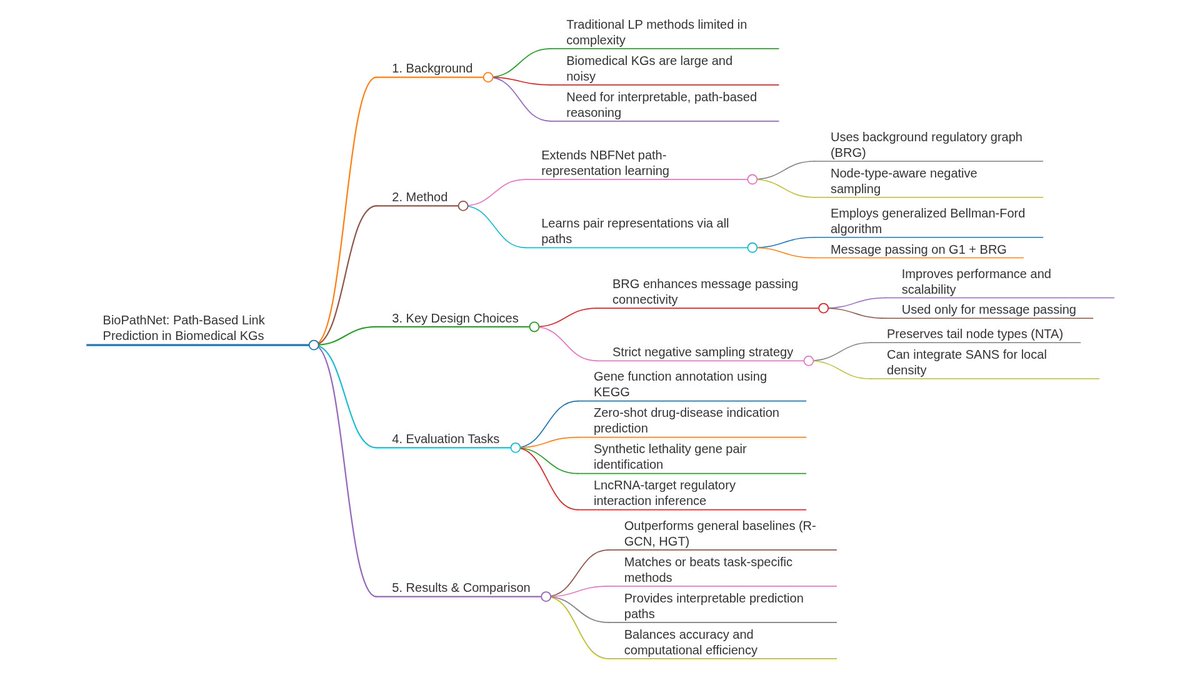
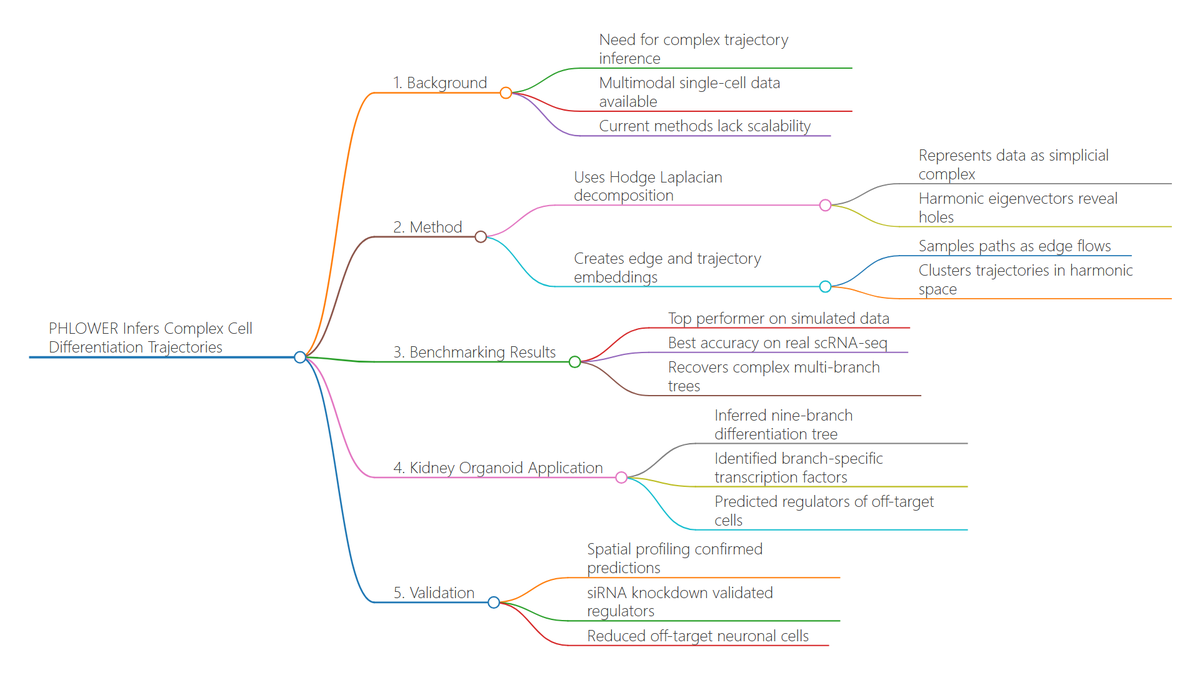
Ever tried to watch a movie from a few random, blurry stills? That's the challenge of modeling cell development with single-cell data. 🧬 We get sparse, noisy snapshots, not continuous timelines.
New review in Comms Biology breaks down how Neural ODEs are solving this. They learn the continuous vector field that drives cells from one state to another, turning static snapshots into a dynamic movie of development. 🎬
The big leap? **Flow Matching**. It's a simulation-free way to train these models, efficiently aligning cell population distributions across time points. It's like finding the smoothest path connecting all the dots.
Could this framework finally let us *simulate* cellular development and disease progression in silico? 🤔 What would you model first?
#AI #Science #SingleCell #GenerativeModels #NeuralODE #ComputationalBiology #Bioinformatics
📄 Paper: doi.org/10.1038/s42003…


English