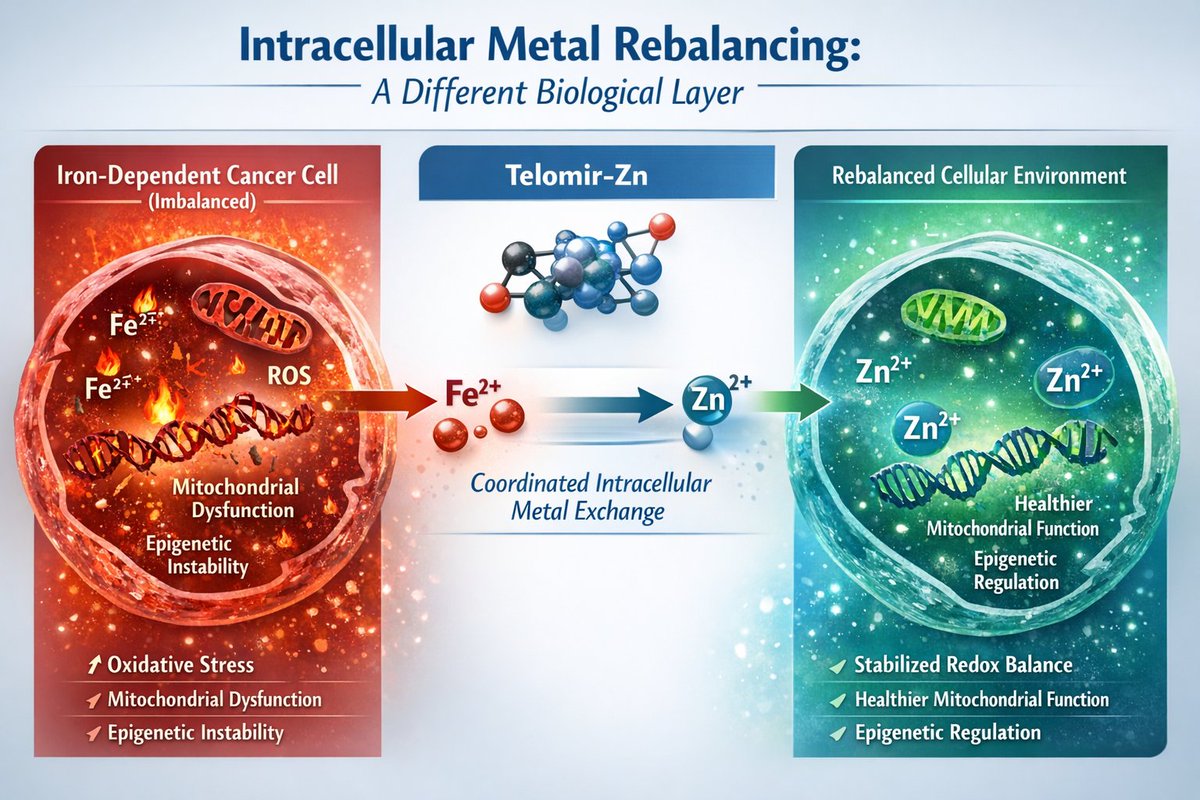
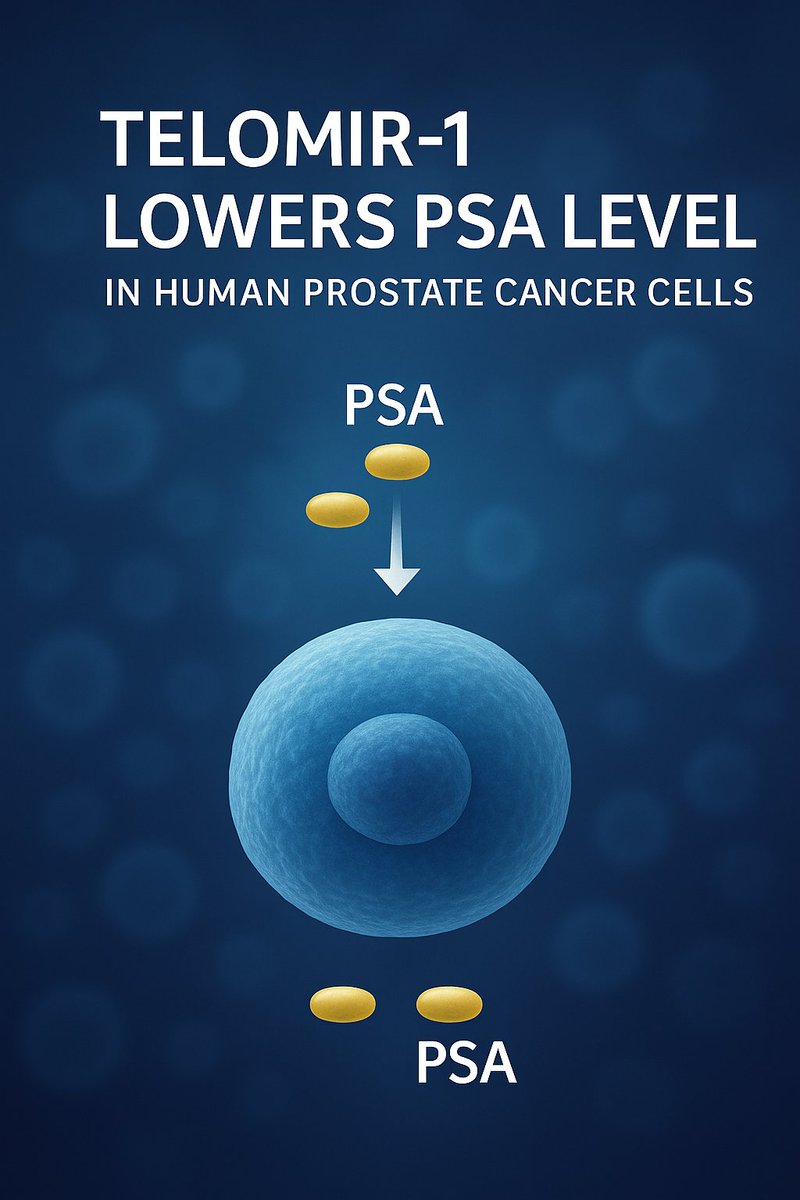
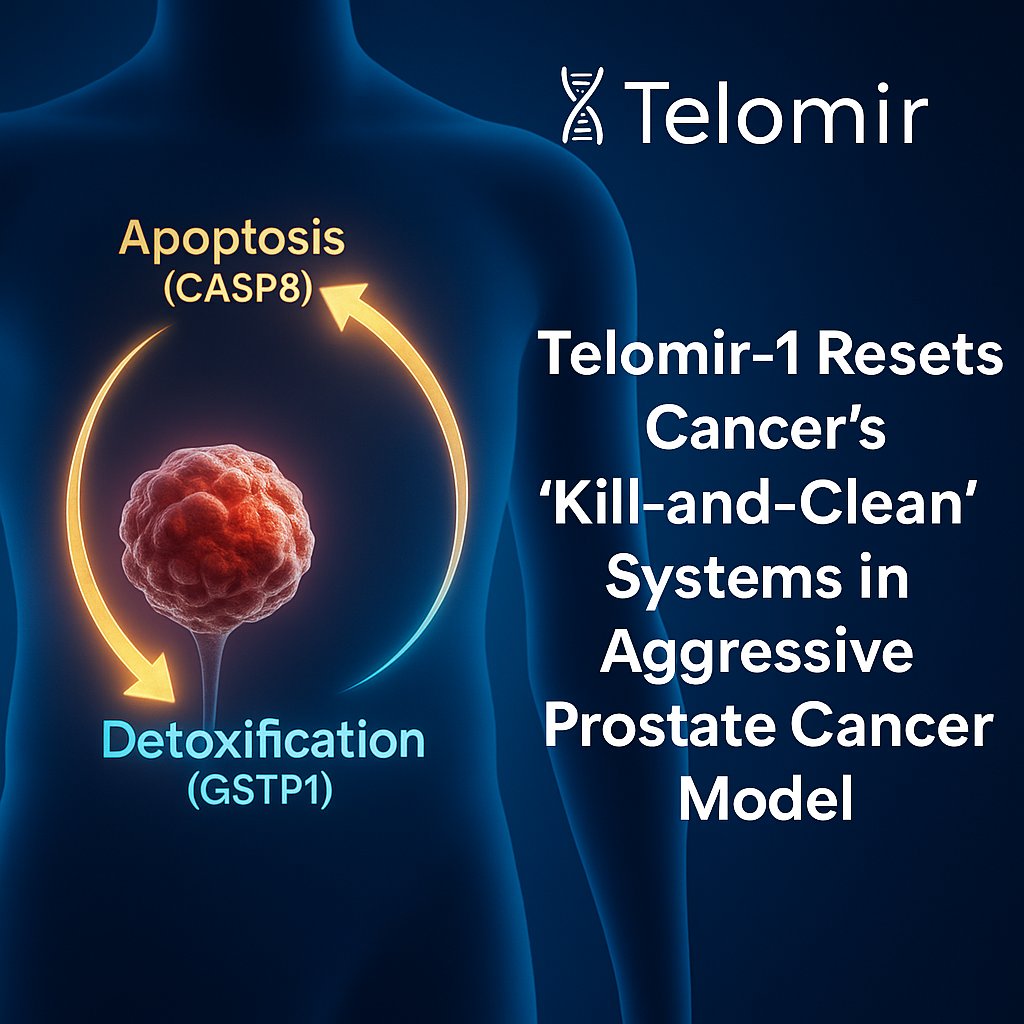
Across IND-enabling GLP safety studies conducted to date, Telomir-1 (Telomir-Zn) has been well tolerated, with no treatment-related adverse or dose-limiting toxicities observed.
The GLP safety package includes cardiovascular telemetry, respiratory assessment, repeat-dose toxicology in rats and dogs, and phototoxicity evaluation. Across studies, systemic exposure was consistent following oral administration, supporting predictable pharmacokinetic behavior.
These data supported IND clearance by the FDA and advancement into first-in-human clinical development.
Follow along for continued clinical and regulatory updates.
#GLPSafety #TelomirZn #IND #ClinicalDevelopment #PreclinicalData #Biotech
English