YPrime
1K posts


YPrime
@Y_Prime
At YPrime, we streamline the clinical trial journey with a configurable platform designed for speed, quality, and certainty: https://t.co/7llG0lAEBl
Malvern, PA Katılım Şubat 2011
209 Takip Edilen369 Takipçiler

Manual PI sign-off processes slow down site workflows and create compliance risk. That’s why YPrime developed our Advanced eCOA Oversight—a guided digital workflow that enables investigators to review and sign off on participant data in real time: hubs.ly/Q03SmFSJ0
#eCOA

English

Oncology trials demand more from your eCOA platform—flexibility, high-quality data, and a better experience for patients. If you're struggling with your existing solution, find out what to look for in an oncology-ready eCOA platform: hubs.ly/Q03Sg6Dq0
#eCOA #OncologyTrials

English

With fewer than 200,000 patients affected per condition—and often much smaller populations—rare disease trials must optimize every data point. Learn how modern eCOA platforms help sponsors reach dispersed populations in our new playbook: hubs.ly/Q03RPVrC0
#RareDisease #eCOA

English

Global regulators are clear: investigator oversight must be documented, attributable, and electronic. YPrime’s Advanced eCOA Oversight meets these evolving EMA, FDA, and ICH E6(R3) expectations. Find out more: hubs.ly/Q03QJQ7r0
#ClinicalTrials #eCOA #DigitalHealth

English

We’re heading to CNS Summit 2025, where AI and automation are shaping what’s next in clinical research. YPrime’s Jonathan Norman will share how these technologies are reducing human error in eCOA localization. If you’ll be there, let's connect.
#CNSSummit #AI #eCOA #ClinicalTrial
English

From Boston to Barcelona, our team is on the move this week!
📍IRT Conference (Boston, booth 20)
📍SCOPE Summit Europe (Barcelona, booth 47)
Don’t miss our session with Amgen and Takeda tomorrow.
#IRTConference #SCOPESummit #SCOPEEurope #SCOPEEU #ClinicalTrials #ClinicalResearch


English

In honor of Breast Cancer Awareness Month, we stand with survivors, honor those we’ve lost, and support the ongoing fight against breast cancer. Together, we can accelerate progress toward a future where breast cancer is preventable, treatable, and curable.
#BreastCancerAwareness

English

Today, we’re proud to release our ESG Report, detailing how we’re embedding environmental stewardship, social responsibility, and ethical governance into every part of our business. Read the full story in our press release: hubs.ly/Q03KCBln0
#ESG #ClinicalResearch

English

This September, YPrime joins the global community in recognizing World Alzheimer’s Month. Through innovation in clinical trial technology, we’re proud to support the studies that bring new hope to patients and families worldwide.
#WorldAlzMonth #AlzheimersAwareness #ClinicalTrial

English

We’re counting down the days until Innovation Network! Don’t miss YPrime's CHRO, Lindsay Daniel, as she dives into the future of safe workplaces—covering how organizations can foster trust, well-being, and lasting impact for their people. We hope to see you there!
#ING2025

English

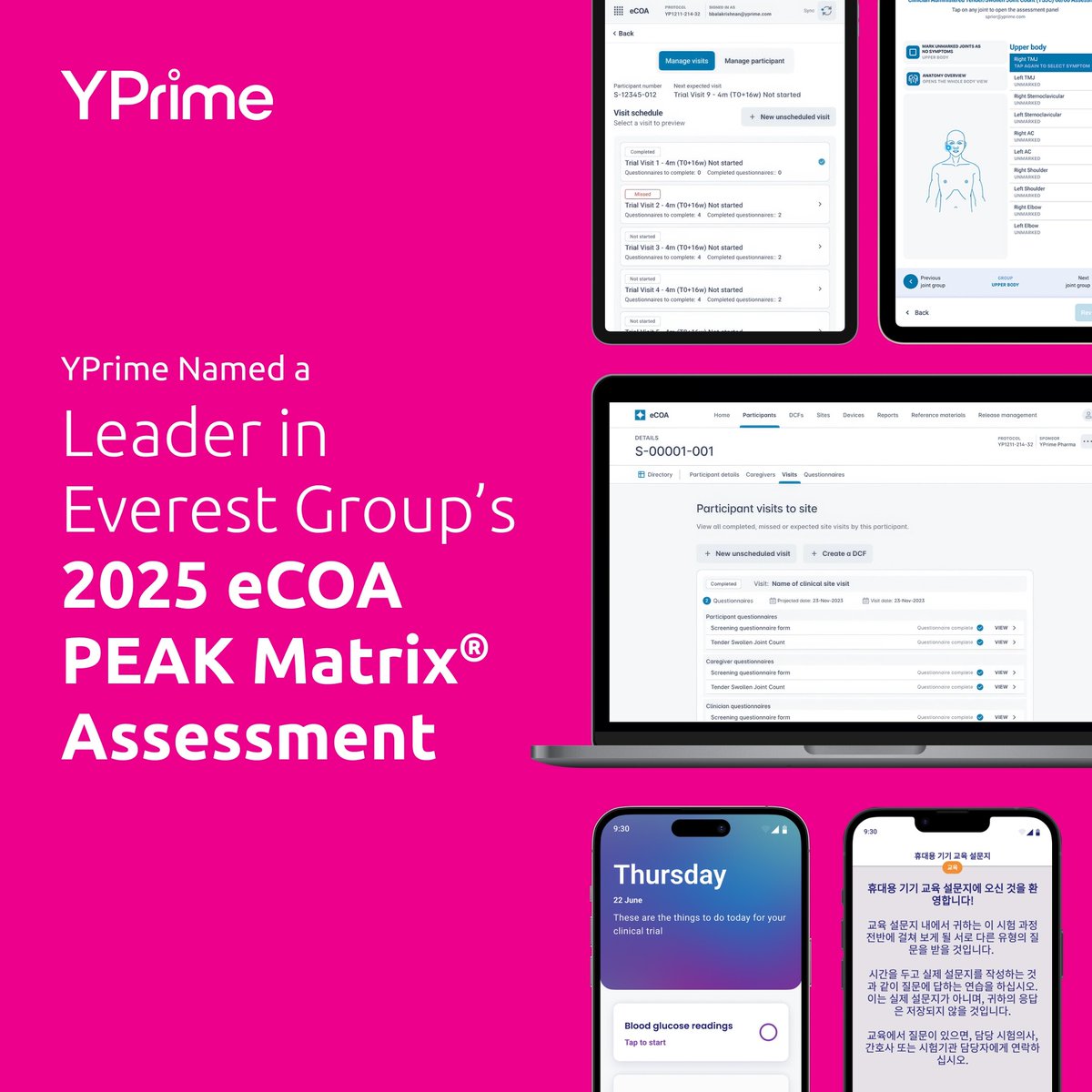
Recognition from Everest Group as a Leader in the 2025 eCOA PEAK Matrix® Assessment reinforces our commitment to speed, flexibility, and quality. Find out why our eCOA is best in class: hubs.ly/Q03H778R0
#EverestGroup #eCOA #ClinicalTrials #ClinicalResearch #LifeSciences

English

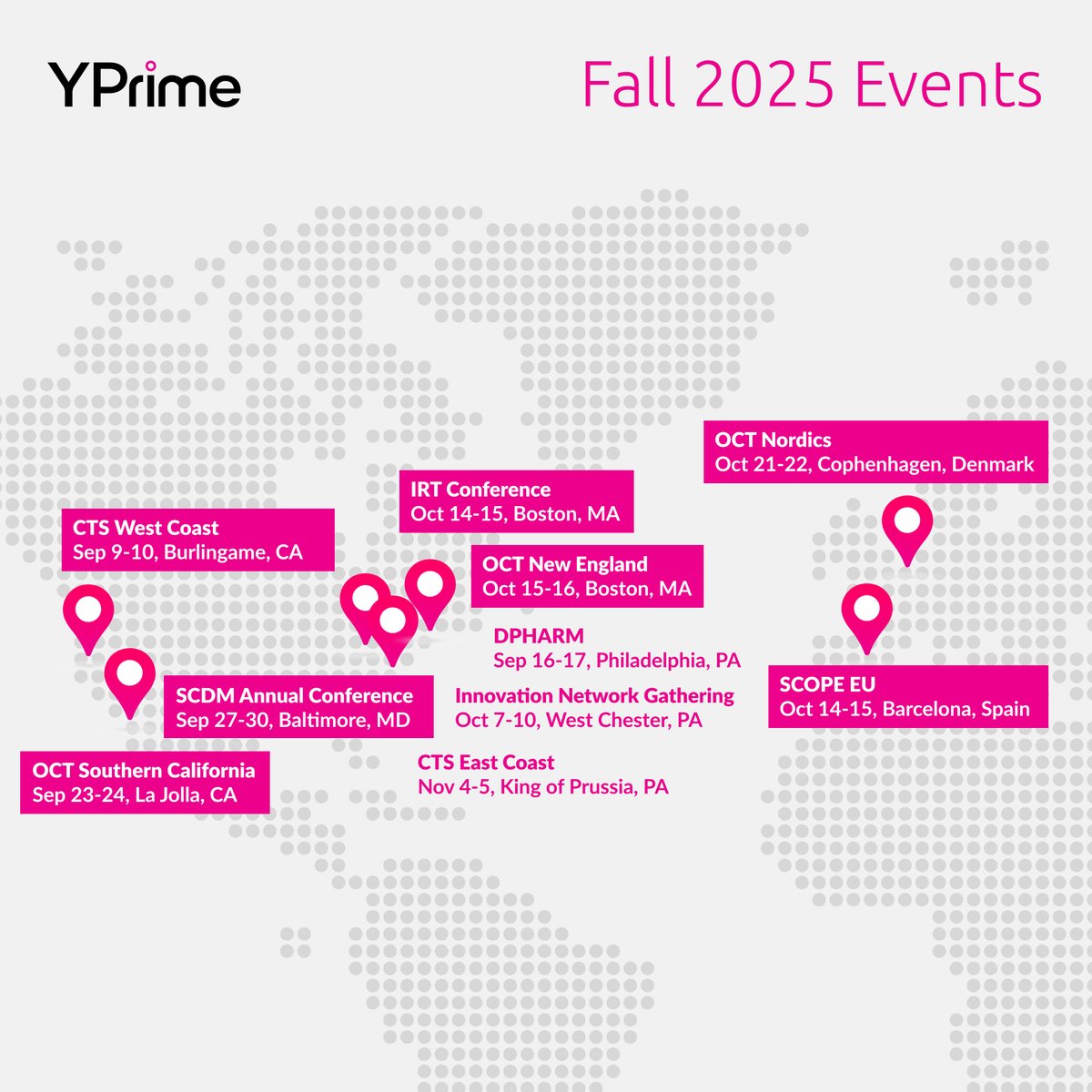
YPrime is hitting the road this fall! From California to Barcelona, we’re bringing faster and more flexible clinical trial tech to the world stage. See where we’re headed and book a meeting: hubs.ly/Q03H3br-0
#ClinicalTrials #ClinicalResearch #eCOA #IRT #eConsent
English

We’re proud to share that YPrime has been recognized as a Leader in Everest Group’s 2025 eCOA PEAK Matrix® Assessment. Read the full announcement: hubs.ly/Q03GspyD0
#ClinicalTrials #eCOA #LifeSciences #EverestGroup #TrialTechnology
English

Oncology trials have requirements that other clinical trials do not—and your eCOA platform should support this. YPrime’s Oncology eCOA Playbook breaks down what sponsors should demand from their platforms. Download it here: hubs.ly/Q03FCxDJ0
#OncologyTrials #eCOA

English

Q4 is right around the corner—are your clinical trial systems where they need to be? If your trial is being slowed down by protocol amendments, device integration issues, or ICH E6(R3) requirements, let's talk. Let’s build your Q4 ramp-up plan together: hubs.ly/Q03CWVtQ0

English

We’re proud to share that YPrime's Ryan Ridge has been named a finalist for Informa's IRT Young Professional Award. This recognition celebrates rising leaders who are shaping the future of clinical trial operations—and Ryan is doing just that. Vote today: hubs.ly/Q03CKYXw0

English

Pain studies require advanced eCOA to support episodic symptoms, psychological impacts, and more—with patient-first technology. Discover how the right eCOA makes the difference in our new playbook: hubs.ly/Q03CrqM80
#eCOA #PainManagement #ClinicalTrials #ClinicalResearch
GIF
English

GLP-1 trials are accelerating, and so is the complexity. In his latest blog, YPrime's Karl McEvoy shares how eCOA innovation can help sponsors manage large-scale, multi-endpoint studies with greater efficiency: hubs.ly/Q03Bq2mZ0
#GLP1 #ClinicalTrials #eCOA #DigitalHealth
English

