
Birgit Habenstein
63 posts

Birgit Habenstein
@bihalo
PI @CNRS in #NMR of molecular assemblies, amyloids, membrane proteins, lipids












FR 🌟 La 1ère Conférence du Projet RePo-SUDOE a réuni des experts internationaux pour discuter de drug repurposing, partager des cas de succès et promouvoir de nouvelles opportunités d’innovation pour la région SUDOE💊🌍. Plus d'information: interreg-sudoe.eu/noticia-proyec…


Trois témoignages de jeunes collègues sur le programme Emergence@International de @CNRSchimie présentés dans inc.cnrs.fr/fr/cnrsinfo/em…


#compchem New group paper published in @CommsBio: "Dynamic pre-structuration of lipid nanodomain-segregating remorin proteins" nature.com/articles/s4200… Remorins are multifunctional proteins, regulating immunity, development and symbiosis in plants. When associating to the membrane, remorins sequester specific lipids into functional membrane nanodomains. The multigenic protein family contains six groups, classified upon their protein-domain composition. Membrane targeting of remorins occurs independently from the secretory pathway. Instead, they are directed into different nanodomains depending on their phylogenetic group. All family members contain a C-terminal membrane anchor and a homo-oligomerization domain, flanked by an intrinsically disordered region of variable length at the N-terminal end. We here combined molecular imaging, NMR spectroscopy, protein structure calculations and advanced molecular dynamics simulation to unveil a stable pre-structuration of coiled-coil dimers as nanodomain-targeting units, containing a tunable fuzzy coat and a bar code-like positive surface charge before membrane association. Our data suggest that remorins fold in the cytosol with the N-terminal disordered region as a structural ensemble around a dimeric anti-parallel coiled-coil core containing a symmetric interface motif reminiscent of a hydrophobic Leucine zipper. The domain geometry, the charge distribution in the coiled-coil remorins and the differences in structures and dynamics between C-terminal lipid anchors of the remorin groups provide a selective platform for phospholipid binding when encountering the membrane surface. Great work led by @bihalo. @AdrienSchahl @Matth_Chavent





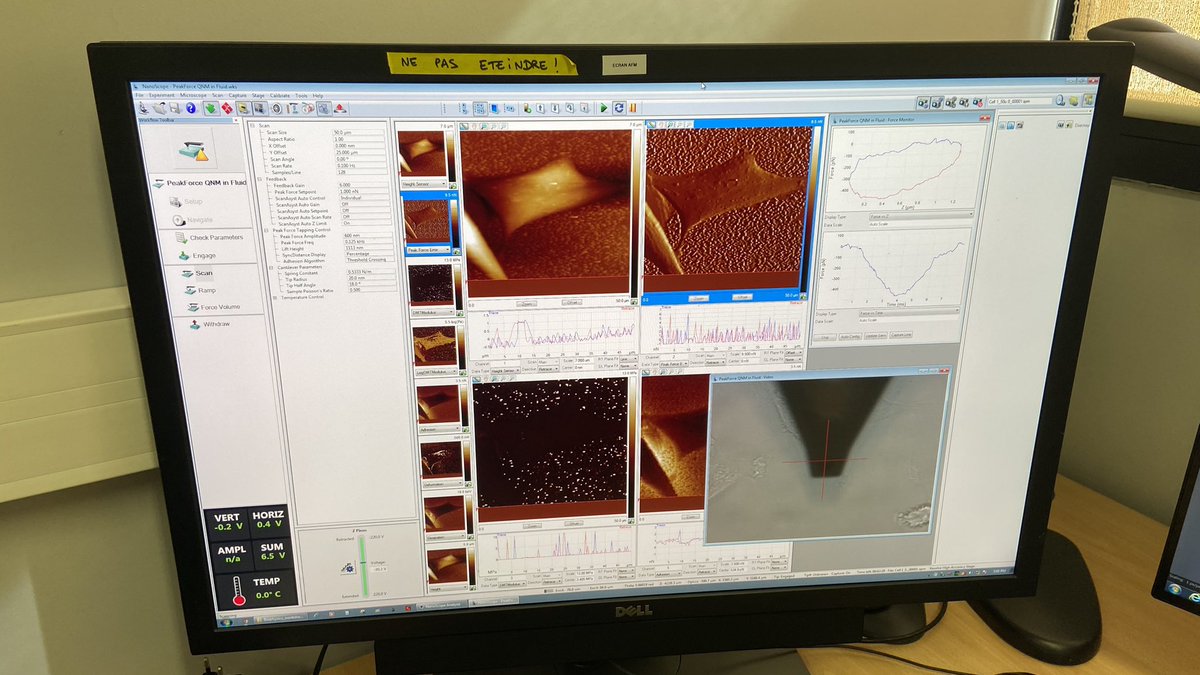


@cerm_cirmmp Final talk of the morning is from Birgit Habenstein @bihalo on solid-state NMR of biological assemblies



That was also good to see the @univbordeaux colleagues @YFichou and B. Habenstein ! @BxFrontiers