Sabitlenmiş Tweet

As far back as 2019, Richard Pazdur, then head of OCE at FDA, recognized the importance of lymphocyte count in progression free survival and overall survival in lung cancer. I just discovered this paper today, just as I recently discovered the 2007 NCI workshop report showing the importance of T cell growth factor.
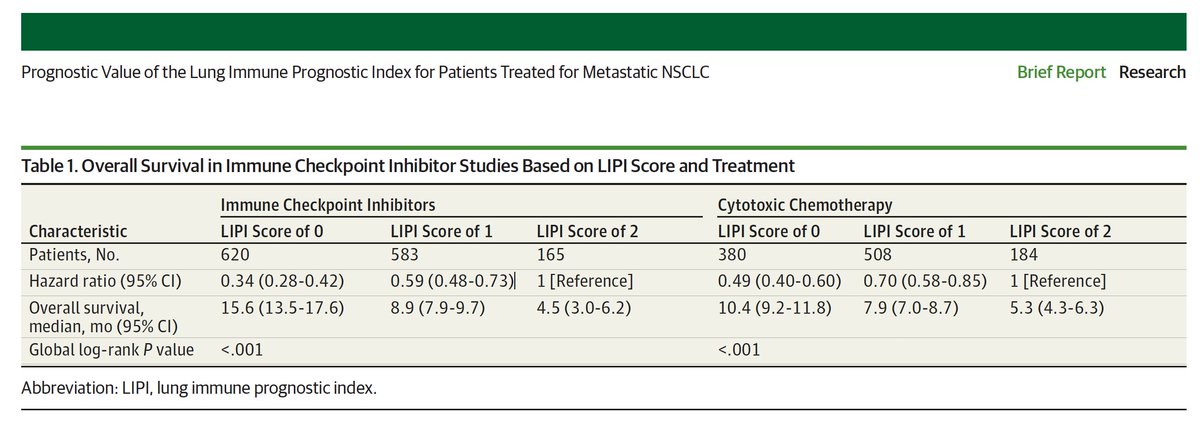
He concludes that "baseline LDH level and dNLR are important prognostic biomarkers irrespective of treatment modality for patients with mNSCLC" meaning that whether the patient receives radiation, chemo, or checkpoint inhibitors, the level of lymphocyte count (ALC) is the critical biomarker for outcomes regardless of treatment modality.
The Saudi FDA recognized this immediately and on that basis approved Anktiva for the treatment of 2nd line lung cancer who relapsed following standard of care.
Pazdur further states "As further prospective clinical trial information is collected, the role of the LIPI score can be better defined." Stay tuned.


English










