Sabitlenmiş Tweet
Katsuki Arima
2.9K posts


Katsuki Arima
@katsuki_arima
Breast Oncologist, Researcher, Brazil. @ESOncology Fellow Interest: #breastcancer #clinicalresearch Instagram @katsukitiscoski Follow @oncoalert
Porto Alegre, Brasil Katılım Ağustos 2016
1.8K Takip Edilen1.3K Takipçiler
Katsuki Arima retweetledi

Highlights on #ESMOBreast26 from #CommunityOnc:
Her2
1. #DB11 Update: NeoAdj TDXd
2. #PHERGain: De-escalation
3. #SATEEN: Sacituzumab
HR+
4. #PREcoopERA: Role of OFS
5. #CAPItello291 Update: Capivasertib
TNBC
6. #TROPIONBreast02: DatoDXd
#OncTwitter #bcsm @myESMO
1/7

English
Katsuki Arima retweetledi

Interesting data showing that upfront SLNB may be reasonable for select patients presenting with cN1 HR+/HER2- breast cancer - no recurrences in those with at least 12 months of f/u at time of analysis
JAMA Surgery@JAMASurgery
Among patients with cN1 HR+/HER2- #BreastCancer, upfront sentinel lymph node biopsy was feasible, enabling omission of axillary lymph node dissection in nearly 70% with limited nodal disease burden. ja.ma/4narNgi
English
Katsuki Arima retweetledi

What happened this week in #BreastCancer:
1⃣ FDA approval of PROTAC ER degrader in ESR1m
Regulatory clearance of vepdegestrant for ER-positive, HER2-negative, ESR1-mutated advanced or metastatic disease after endocrine therapy based on VERITAC-2 positions it as a new post CDK4/6i endocrine option with improved PFS vs fulvestrant and necessitates ESR1 testing via the simultaneously approved companion diagnostic to detect ESR1 mutations.
fda.gov/drugs/resource…
English
Katsuki Arima retweetledi

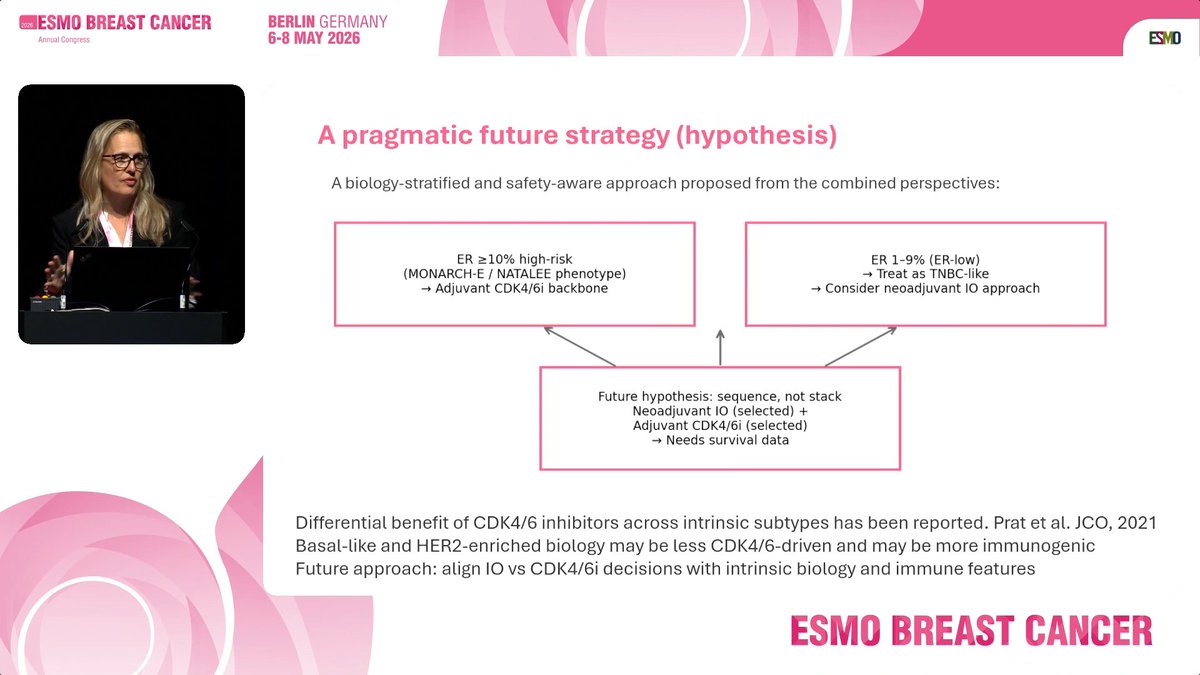
#ESMOBreast26
Neoadjuvant immunotherapy in ER+/HER2− early BC is still an open question.
A pCR signal is interesting, but not enough in this subtype.
We need EFS data.....
From Dr. Rebecca Dent's excellent presentation.
Pragmatic strategy👇

English
Katsuki Arima retweetledi

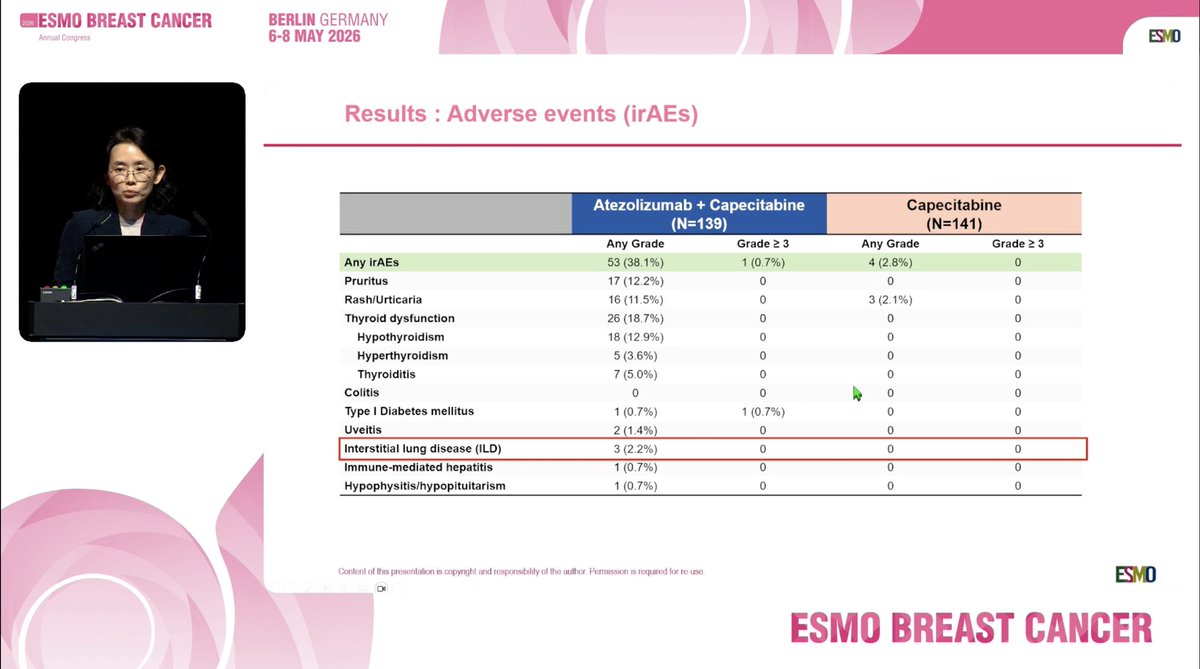
📌 Randomized, phase II trial to evaluate the efficacy and safety of atezolizumab plus capecitabine adjuvant therapy compared to capecitabine for triple-negative breast cancer (TNBC) with residual invasive cancer after neoadjuvant chemotherapy (MIRINAE trial, KCSG-BR18-21) @OncoAlert #OncoAlertAF #ESMOBreast26 ✨Proferred Paper



English
Katsuki Arima retweetledi

📌 A window-of-opportunity (WOO) trial of giredestrant +/- LHRH analogue vs anastrozole + LHRHa in premenopausal patients with ER+/HER2- early breast cancer: PREcoopERA @elisabettasabet ✨
#ESMOBreast26 @OncoAlert #OncoAlertAF ✨ Proferred Paper


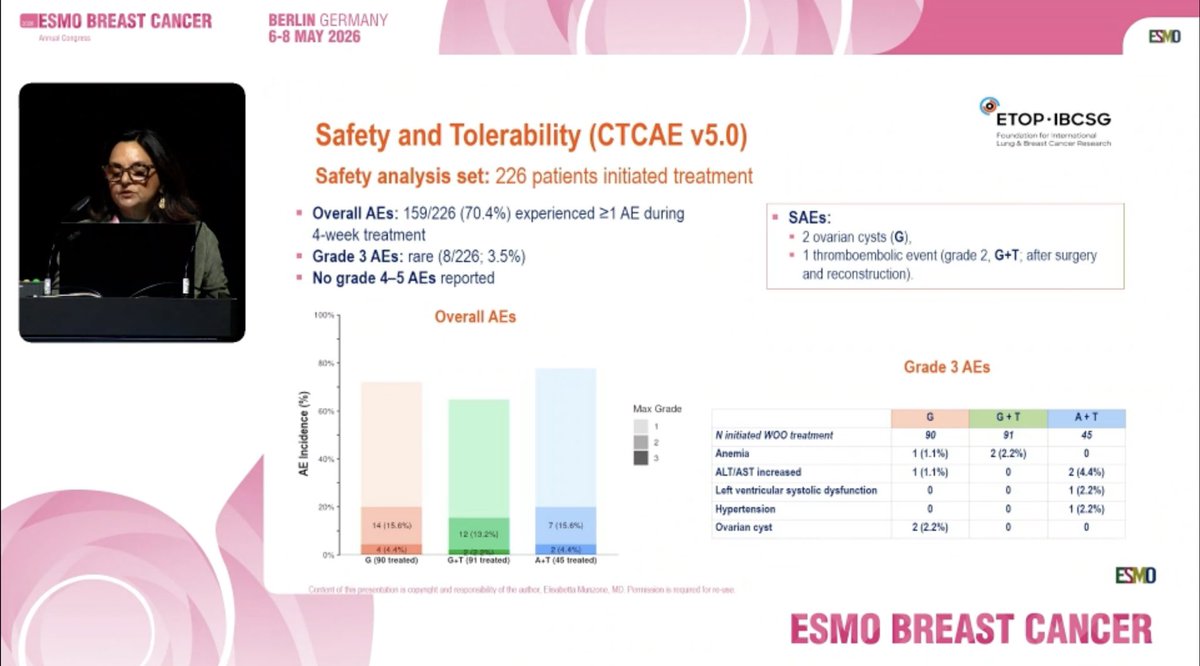

English
Katsuki Arima retweetledi

PREcoopERA Trial: Oral SERD Activity in Premenopausal HR+ Early #BreastCancer, Promise Without Replacement Yet 🧬
Giredestrant with or without ovarian suppression shows robust Ki67 reduction in premenopausal HR+ early breast cancer,
⬇️⬇️
@OncoAlert #ESMOBreast26 #bcsm




English
Katsuki Arima retweetledi

DESTINY-Breast11:
T-DXd–Based Neoadjuvant Therapy Outperforms Anthracycline-Containing Regimens in High-Risk HER2+ EBC, Delivering Higher pCR and RCB 0/1 Rates and Supporting a Shift Toward an Anthracycline-Free Standard
@OncoAlert #ESMOBreast26

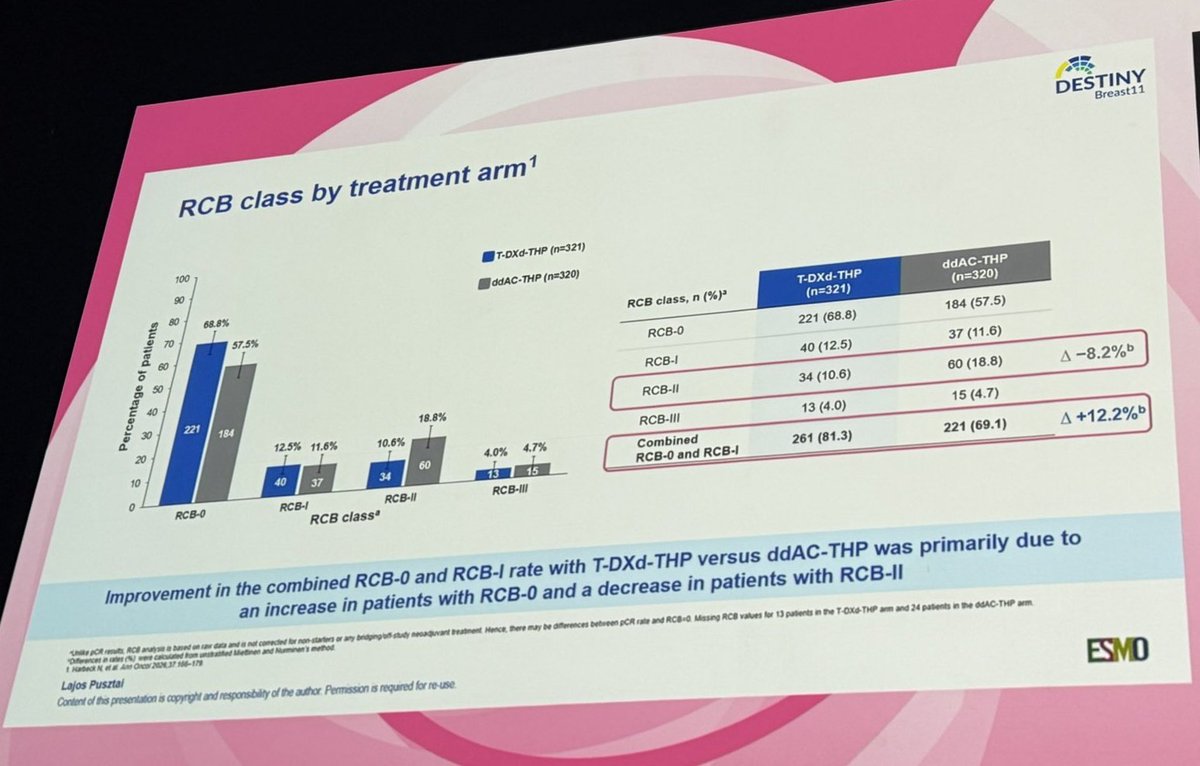
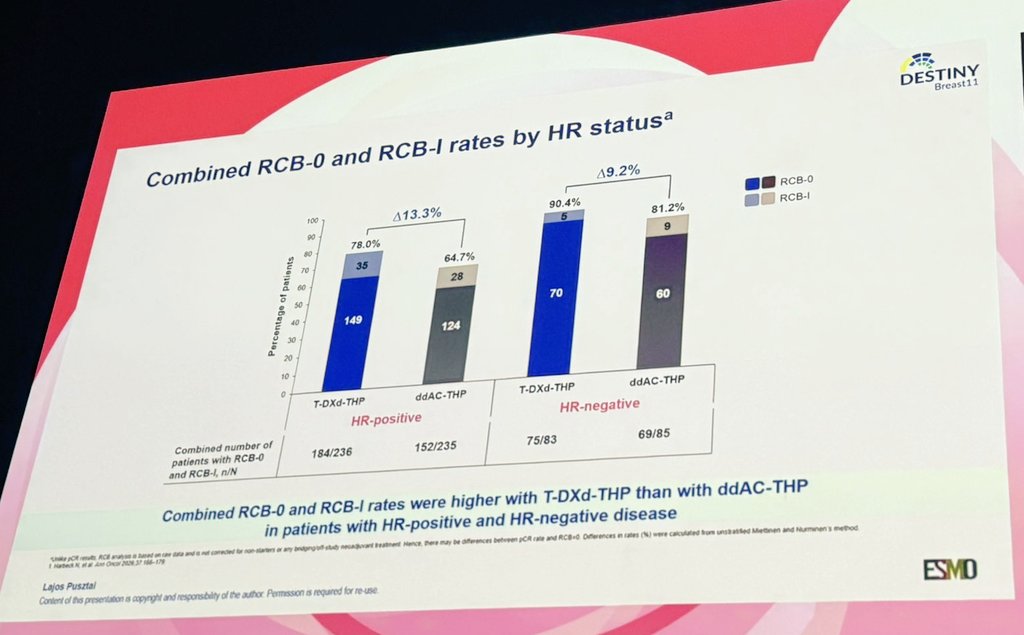
English
Katsuki Arima retweetledi

Simultaneous publication with #ESMOBreast26
An excellent de-escalation study: PHERGain-2 study
A chemotherapy-free, pathological response-adapted strategy using trastuzumab–pertuzumab and T-DM1 in HER2-positive early breast cancer
sciencedirect.com/science?_ob=Ga…

English
Katsuki Arima retweetledi

Esmo Breast 2026 - LBAs are out . What's the new data from Destiny Breast 11 ?
It improves RCB across all the subgroups. Looking forward to full data. @myESMO #esmobreast26



English
Katsuki Arima retweetledi

My favorite study today at #ESMOBreast: PREcoopERA (IBCSG 67-22), a window-of-opportunity trial in premenopausal ER+/HER2- early BC asking whether giredestrant alone is enough, or whether ovarian suppression is still required.
231 patients → giredestrant alone, G + triptorelin, or anastrozole + triptorelin. Primary endpoint: Ki67 at 4 weeks.
Giredestrant alone did NOT meet non-inferiority vs G+OS (-68% vs -80%). And G+OS was not superior to anastrozole+OS (p=0.18).
Bottom line: Even for a next-gen SERD, ovarian suppression adds to anti proliferation effect in premenopausal patients.
#ESMOBreast #BreastCancer #Giredestrant

English
Katsuki Arima retweetledi
Katsuki Arima retweetledi

📌 Chemotherapy-free, pathological complete response (pCR)-guided strategy with trastuzumab-pertuzumab (HP) and T-DM1 in HER2+ early breast cancer (EBC): PHERGain-2
#ESMOBreast26 @OncoAlert #OncoAlertAF




English
Katsuki Arima retweetledi

The activating ERBB2 mutations are significantly enriched in invasive lobular carcinoma (ILC) versus NST metastatic breast cancer (10.7% vs 3.0%).
Findings highlight the biologic heterogeneity of HER2-mutant HR+ metastatic breast cancer and support more refined biomarker-driven treatment strategies. #bcsm #BreastCancer #PrecisionOncology #esmobreast2026 @Larvol @OncoAlert

English
Katsuki Arima retweetledi

HR+/HER2– Early Breast Cancer:
From Endocrine Biology to Immune Precision—Redefining Neoadjuvant Strategy
@OncoAlert #ESMOBreast26 #bcsm #BreastCancer @myESMO
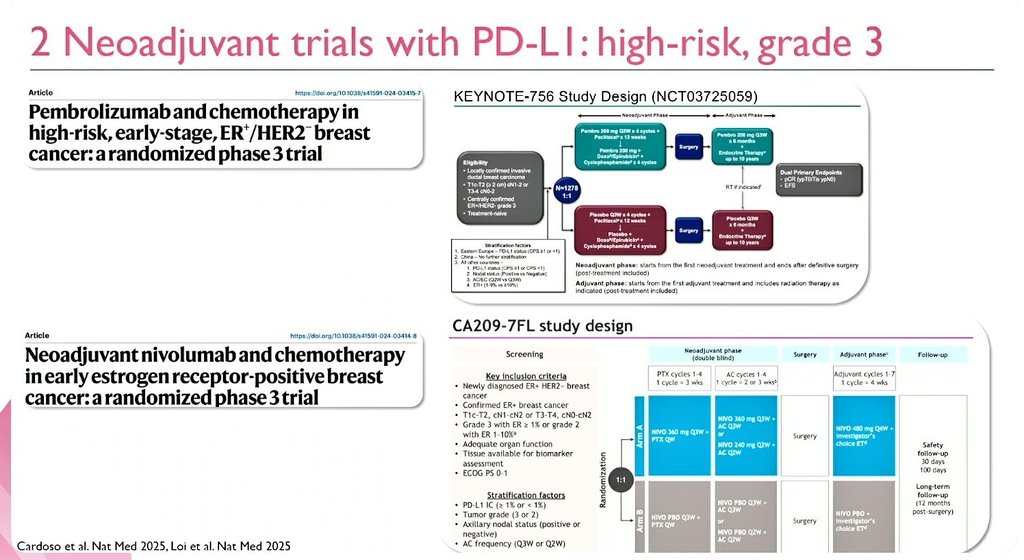


English
Katsuki Arima retweetledi
Katsuki Arima retweetledi

A MUST-READ!
This is probably one of my most important papers where I try to teach how to fish rather than offer fish.
How I Read a Clinical Trial Report?
BG’s primer for Busy Clinicians.
Thank you @JCOOP_ASCO @EthicsdoctorP for the kind invitation. I hope the readers will find this useful.
ascopubs.org/doi/10.1200/OP…

English
Katsuki Arima retweetledi

SONIA trial @JAMAOnc :
In HR+/HER2− advanced #BreastCancer, first-line CDK4/6i did not improve OS vs delayed use (47.9 vs 48.1 mo; HR 0.91), despite modest PFS gain. Higher toxicity without QoL benefit.
jamanetwork.com/journals/jamao…
@OncoAlert #bcsm

English
Katsuki Arima retweetledi

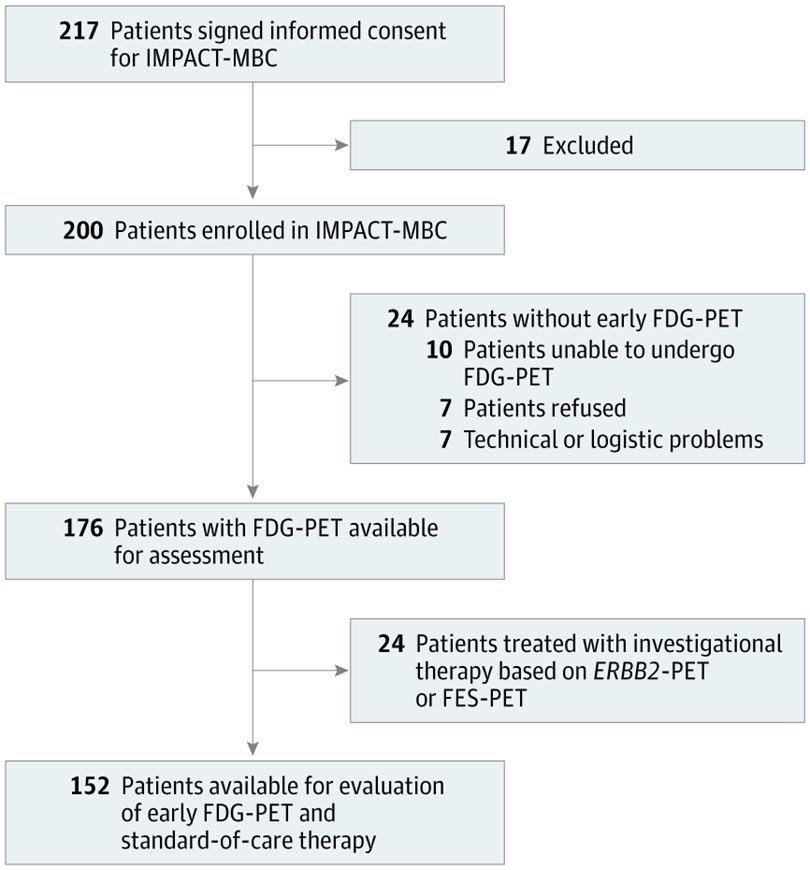
📌 18F-Fluorodeoxyglucose Positron Emission Tomography for Estimating Outcomes After Initial Treatment for Metastatic Breast Cancer
A Nonrandomized Clinical Cohort Trial
🔗 jamanetwork.com/journals/jamao…
@OncoAlert #OncoAlertAF #BreastCancer @JAMANetwork @univgroningen
👉🏻 In this clinical cohort trial of patients with nonrapidly progressive, newly diagnosed MBC before initiation of first-line systemic therapy, early FDG-PET after only 2 weeks of treatment identified patients with MBC with distinct long-term outcomes. Incorporating early FDG-PET can improve outcome estimation of standard CT assessment.


English