Sabitlenmiş Tweet
Nicolò Tampellini
130 posts


Nicolò Tampellini
@ntampellini_
PhD student @MillerGroupYale 🇺🇸 B. Sc. @LabSynthesis 🇮🇹 Asymmetric catalysis with flasks and computers ⚗️💻
New Haven, CT Katılım Ekim 2021
105 Takip Edilen135 Takipçiler

Please check out Rowan, an amazing company taking the technical hassle out of computational chemistry!
The project is of course fully open source. Happy to see this project being adopted!
Code on Github: github.com/ntampellini/pr…
4/4
English

In collaboration with Jonathon from @RowanSci, I packaged my code into a polished standalone library for ease of use and contribution! Working with Jonathon was not only a breeze, but also a great formative experience about modern software development practices (thanks!).
3/4
English

Too many chemically identical rotamers in your CREST ensembles? If you are not interested in them, they can inflate your ensembles greatly!
rowansci.com/blog/screening…
(w/@RowanSci)
🧵1/4
English
Nicolò Tampellini retweetledi

Simple yet Efficient New Method for Direct Site-selective Deoxygenation of Benzylalcohol Derivatives is now out in @ChemRxiv (go.shr.lc/4iLL6JI)

English
Nicolò Tampellini retweetledi

🚨RADICAL RETHINK: STEREORETENTIVE CROSS COUPLING UNLOCKED🚨 Today in @ChemRxiv (chemrxiv.org/engage/chemrxi…) the first method for stereoretentive radical cross-coupling is disclosed. No fancy ligands or redox needed—just a Ni-diazene twist. 120 years after Gomberg, a new chapter begins.
Quick summary:
Since Gomberg discovered free radicals over 120 years ago and Kochi pioneered radical cross-coupling in the 1970s, this field has surged with interest for linking C(sp3) fragments. Unlike traditional polar methods (e.g., Suzuki), radical cross-coupling excels with saturated systems, offering mild conditions and chemoselectivity to build complex molecules from common fragments. Until now, enantiospecific radical cross-coupling was deemed impossible due to rapid racemization, but today that changes with readily available sulfonylhydrazides and a simple Ni-catalyst. This stereoretentive approach, enabled by a unique Ni-diazene transition state and driven by loss of N2, skips chiral ligands and redox steps, opening new possibilities for synthesis.

English
Nicolò Tampellini retweetledi

Cationic, Iodine(III)-Mediated and Directed Diastereoselective Oxidation of Inert C-H Bonds in Cyclic Hydrocarbons (Nuno Maulide and co-workers) @MaulideLab #openaccess 🔓 onlinelibrary.wiley.com/doi/10.1002/an…
English
Nicolò Tampellini retweetledi

Join us today as @ntampellini_ from @MillerGroupYale shares his work on enantiocontrolled cyclization reactions!! Thanks to Sourav @CompoSciTian_SM for hosting!
Link: youtu.be/9e4Hxt5FjH4
@J_A_C_S doi.org/10.1021/jacs.4…

YouTube

English

@ntampellini_ @J_A_C_S Simply amazing, Tampe! Congrats 🎉🎉
English

The final version of this work is now out in @J_A_C_S
pubs.acs.org/doi/10.1021/ja…
Nicolò Tampellini@ntampellini_
It's finally time to share this: my long time obsession with inherently chiral medium-sized rings now has a catalytic, enantioselective method. After a number of attempts, we developed a strategy based on choosing the maximally preorganized disconnection, which... [1/3]
English

@ntampellini_ @J_A_C_S Congrats Nico! Fantastic piece of work
English

@ntampellini_ @J_A_C_S Excellent work, Nick.
Inherently chiral stuff,
challenging, but cool.
English

After long days spent on isosurface visualization, sparse data storage, and the like ... @RowanSci's orbitals workflow is live!
Molecular orbitals, computed with QM, provide a picture of the location of electrons, yielding insights about bonding, excited states, and reactivity.
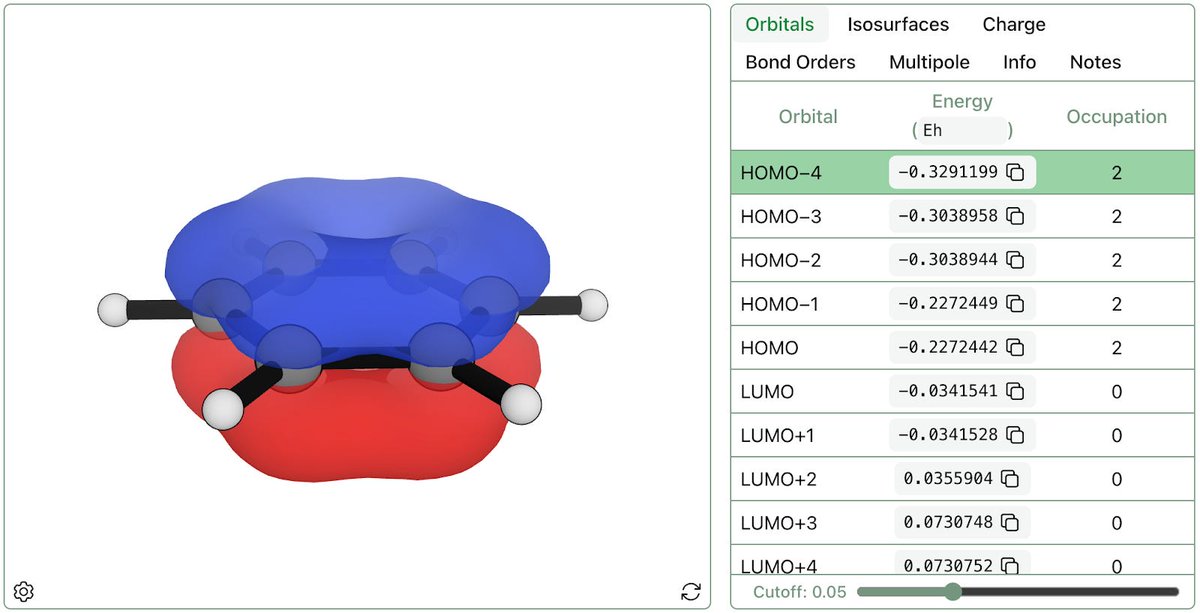
English

@CorinWagen I feel you - I had to write a Python parser for .cube files years ago and I was shocked to learn how they are put together 😅
English

Let's follow each other on Bluesky:
bsky.app/profile/ntampe…
English

@KmorisakiC @ChemRxiv Awesome! Thank you, looking forward to reading about the next steps.
English

@ntampellini_ @ChemRxiv Thank you for your comment! We have not yet tested chiral ligands, but there also would be match-mismatch effects with chiral bidentate ligands. The extent to which transient metal-centered chirality influences enantioinduction should be investigated in due course.
English
Nicolò Tampellini retweetledi

Conceptually new asymmetric transformation—Reflexive Chirality Transfer (RCT)— appeared in @ChemRxiv (go.shr.lc/3Z3f03N).
1,3-Dipolar cycloaddition of amino acid Schiff bases proceeds with "preserving chirality" despite the once loss of the chirality at the α-carbon.

English

@LabSynthesis @ACSCatalysis Congrats @Giovanni_Cent11 @a_pellegrini15 @ChiaraPortolani! Enjoyed reading through the rigorous, comprehensive dissection of the mechanistic aspects. Great job!
English
Don't miss our last work on the Organocatalytic Asymmetric Electrophilic Amination of Allylic Boronates now out in @ACSCatalysis. pubs.acs.org/doi/10.1021/ac…
English


