Dr Gabriel Parma. Profesor adjunto de cardiologia
5.4K posts


Dr Gabriel Parma. Profesor adjunto de cardiologia
@parma315
muy felizmente casado y padre de la más linda. amante de la imagenologia cardiovascular . aficionado al fobal y bohemio de corazón , riverense de ley
Montevideo , Uruguay Katılım Mart 2016
418 Takip Edilen786 Takipçiler
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

La Unidad de Imagen Cardíaca de la @uacardio_ccvu, @hcmquintela , representada por la Dra. Julia Tabó comparte caso desafiante en #SISIAC2026. Compartir ciencia y experiencia como camino de crecimiento. ❤️
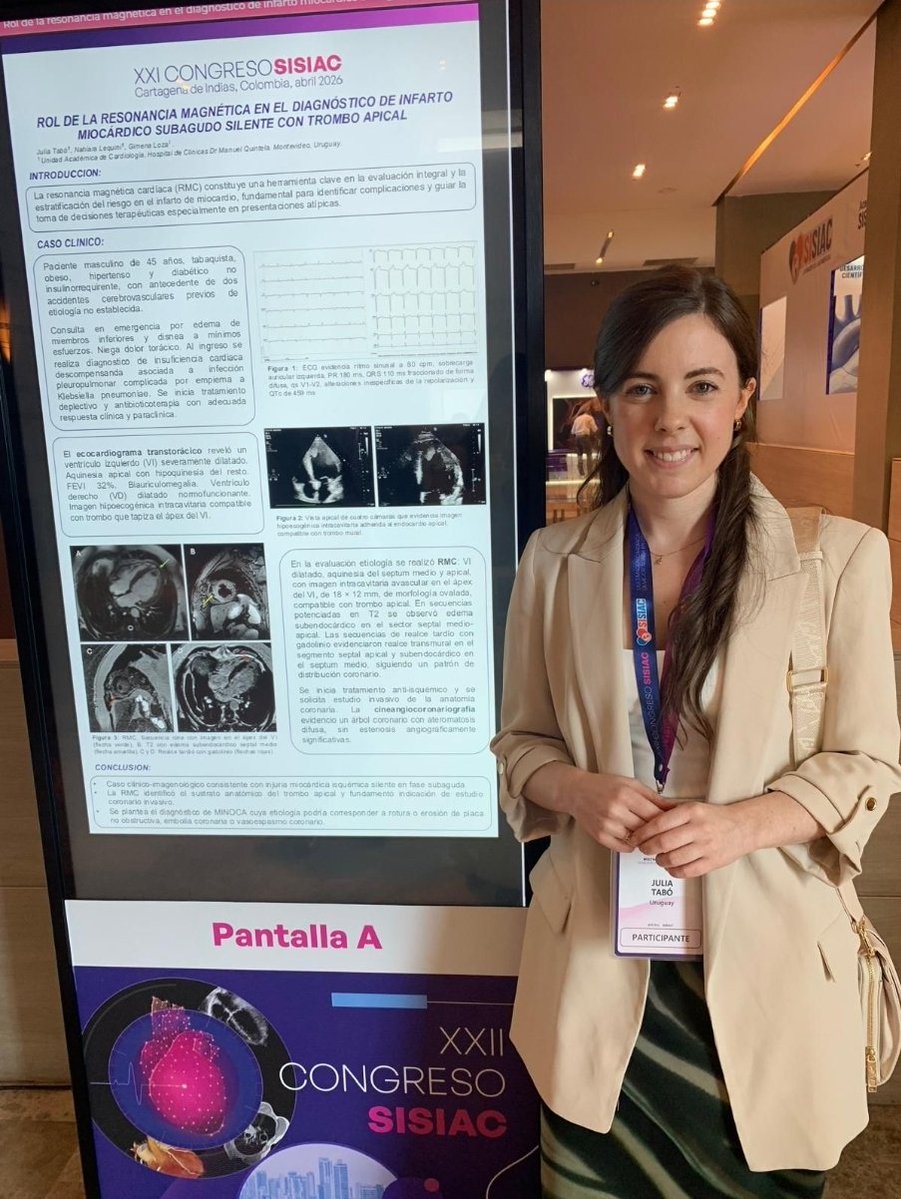
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

❤️CardioSUC 2026 - SIMPOSIO CONJUNTO SUC - American College of Cardilogy Miocardiopatías 2026. Incorporando la genética y la imagen cardiovascular a la práctica diaria 💬 Mensaje del Dr. Federico Acquistapache 👉 Inscribite ahora aquí: buff.ly/oSz4d07
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

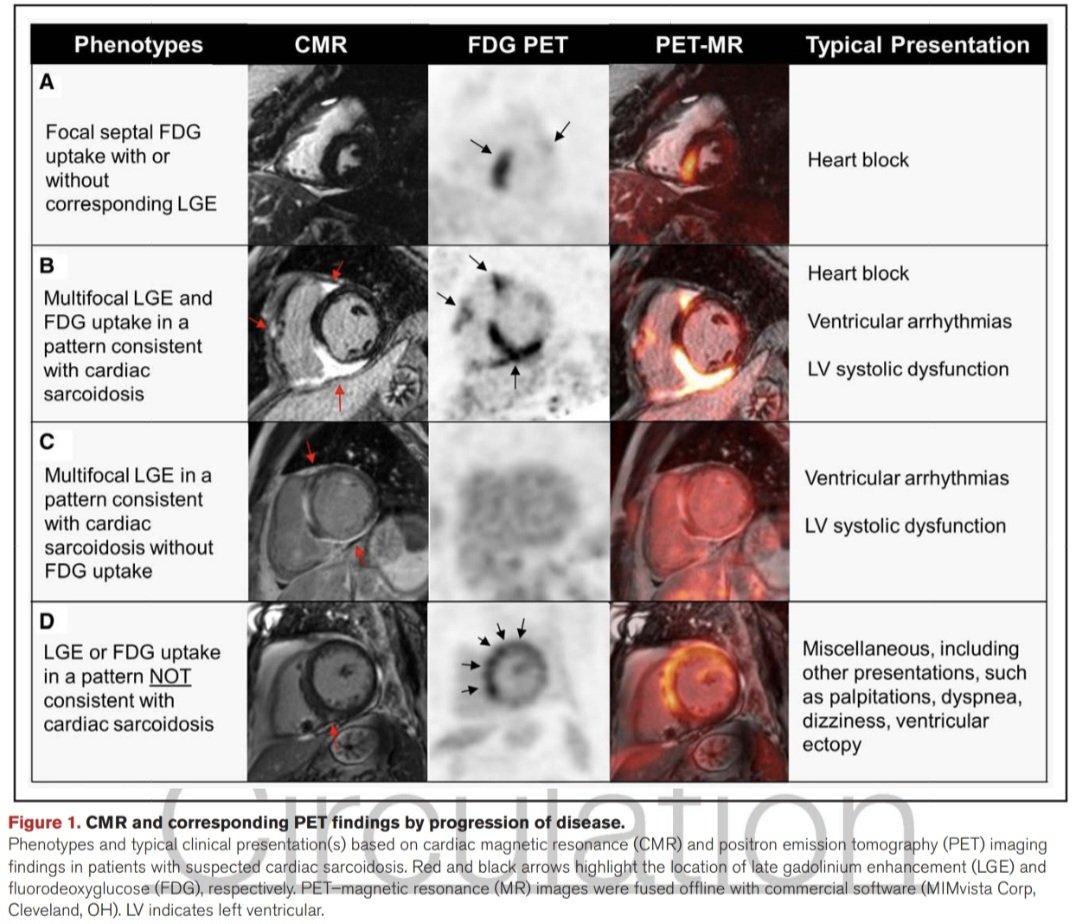
Sarcoidosis Cardíaca. 🫀💥
🛑La sarcoidosis cardíaca es una miocardiopatía infiltrativa secundaria a inflamación granulomatosa no caseificante del miocardio, con manifestaciones clínicas predominantes como trastornos de conducción (+ bloqueo AV), arritmias ventriculares y disfunción ventricular. Presentación altamente variable, hasta 50–65% de los casos puede ser la primera manifestación de la enfermedad sistémica.
🛑Diagnóstico complejo, rara vez se confirma por biopsia endomiocárdica debido a su baja sensibilidad. Se recomienda un enfoque basado en probabilidad diagnóstica (definitiva, probable o posible) integrando datos clínicos, imagen multimodal y exclusión de diagnósticos alternativos.
🛑La RMC y la PET con FDG son pilares diagnósticos: la RMC identifica fibrosis/inflamación mediante LGE, con alto valor pronóstico, mientras que la PET detecta inflamación activa.
🛑Deben sospecharse en escenarios clave como: bloqueo AV de alto grado inexplicable (<60 años), TV sin causa clara, disfunción ventricular o hallazgos estructurales atípicos sin Enf. Coronaria.
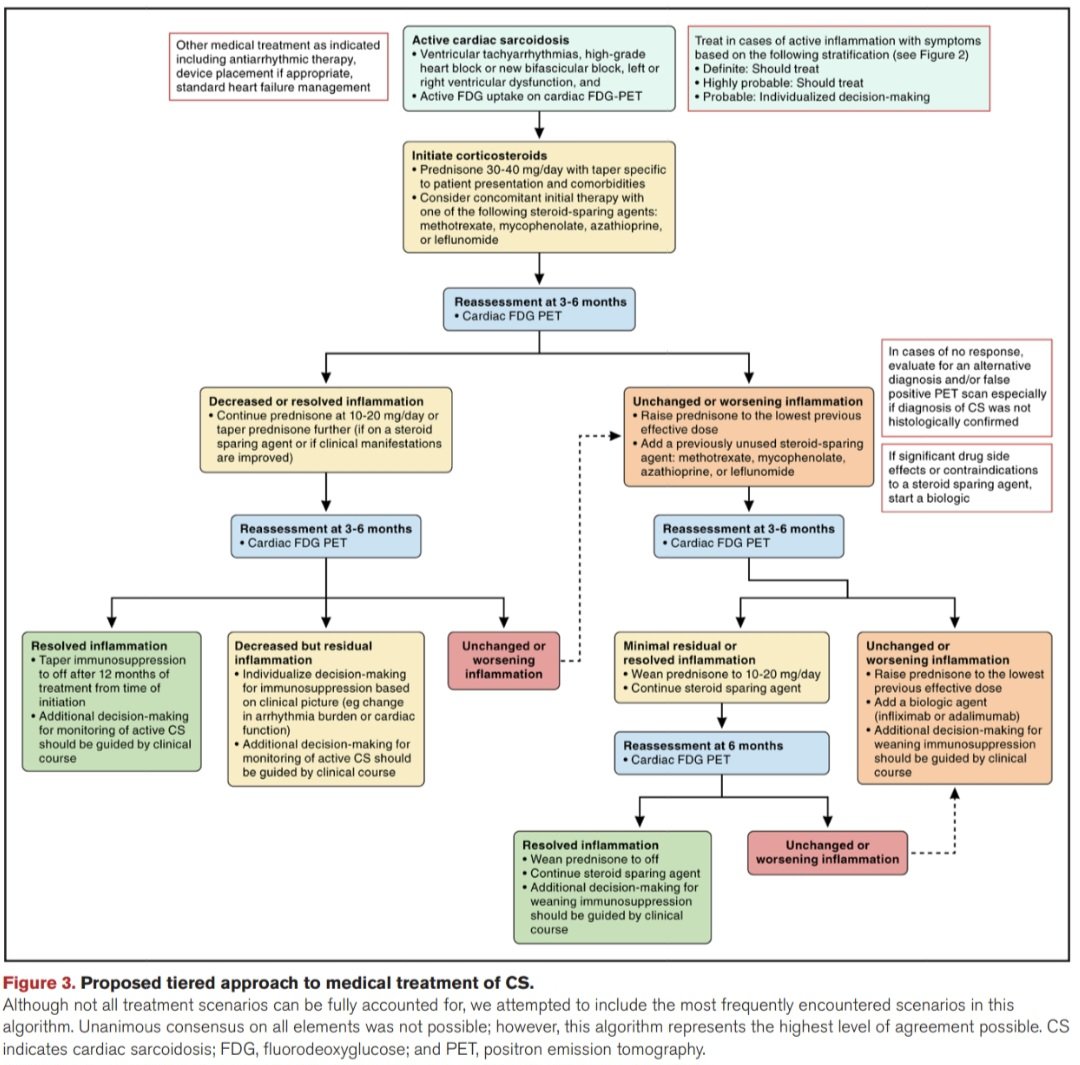
🛑Tratamiento se basa en inmunosupresión: corticosteroides primera línea (prednisona 30-40 mg/día), con esquemas escalonados que incluyen inmunosupresores (metotrexato, azatioprina, micofenolato) y, en casos refractarios, terapias anti-TNF. 💊💉
🔱Scientific Statement @American_Heart 📜🆓️⤵️
DOI: 10.1161/CIR.0000000000001240
t.me/medicinaintern…

Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

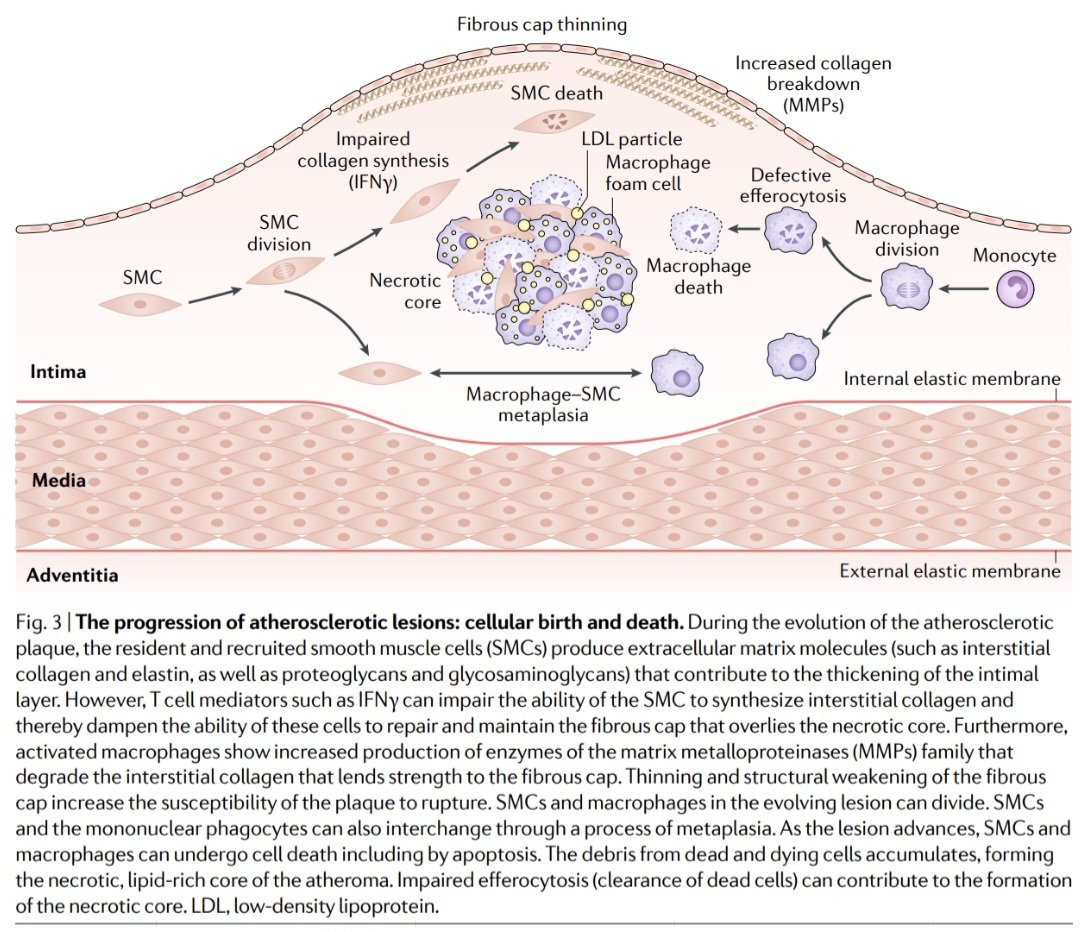
🟩Aterosclerosis: progresión a placa vulnerable. 🫀💥
🟢La progresión de la aterosclerosis es un proceso dinámico de proliferación, transformación y muerte celular dentro de la pared arterial. ♻️❤️🔥
1️⃣ Nacimiento celular (expansión de la placa). La placa crece porque las células no solo se infiltran, también se multiplican localmente: macrófagos proliferan dentro de la lesión, céls musculares lisas (CML) migran desde la media hacia la íntima y se dividen. Además, las CML producen matriz extracelular (colágeno, elastina), formando la estructura de la placa.
2️⃣ Muerte celular (el origen del núcleo necrótico). Con el tiempo, tanto macrófagos como CML sufren apoptosis. En condiciones normales, estos restos deberían eliminarse (eferocitosis), pero en la placa esto falla. Resultado: acumulación de detritos celulares, liberación de lípidos, formación del núcleo necrótico.
3️⃣ Desequilibrio estructural. La estabilidad de la placa depende de su capa fibrosa (colágeno producido por CML). Pero ocurre un doble ataque: ⤵️síntesis de colágeno (inhibida por IFN-γ de linfocitos T), ⤴️degradación de colágeno (por metaloproteinasas de macrófagos). Resultado: capa fibrosa más delgada, menor resistencia mecánica ➡️ placa vulnerable a ruptura! ⚠️💥
📜🆓️⤵️
doi.org/10.1038/s41572…
t.me/medicinaintern…
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🧬 Genetic testing in Dilated Cardiomyopathy: are we using it right?
A new ESC consensus highlights a key shift:
genetics in DCM is no longer just about family screening—it’s now central to clinical decision-making.
🔍 Key messages
🧠 Not just monogenic anymore
DCM is increasingly seen as a continuum:
Rare variants (high impact)
common variants (polygenic background)
environmental triggers
➡️ Disease = interaction, not a single mutation
🧬 Who should be tested? → almost everyone
Genetic testing is recommended when results can impact:
✔️ diagnosis
✔️ prognosis
✔️ treatment (e.g. ICD decisions)
✔️ family screening
➡️ Even “acquired” DCM does NOT exclude genetics
📊 Yield is variable but meaningful
~8–36% overall
up to 55% in familial DCM
➡️ Still clinically impactful despite imperfect yield
⚠️ Gene panels: bigger ≠ better
Large panels increase VUS
Limited gain in actionable variants
➡️ Focus on validated disease genes is key
🔥 Genotype matters clinically
Some genes carry higher arrhythmic risk:
LMNA, FLNC, DSP, RBM20, PLN
➡️ Can influence ICD decision-making beyond EF
🧩 The real paradigm shift: interpretation
Genetic results must be read in context:
phenotype
environment (e.g. myocarditis, chemo, pregnancy)
penetrance
➡️ A variant alone is NOT the diagnosis
👨👩👧 Family impact is huge
Enables cascade screening
Can discharge gene-negative relatives
Opens reproductive options
💡 Take-home message
Genetic testing in DCM is no longer optional or “nice to have”—
it’s becoming a core part of precision cardiology.
👉 But its value depends entirely on how well we interpret it.
#Cardiology #DCM #Genetics #Cardiomyopathy #PrecisionMedicine #HeartFailure #ESC #MedEd 🧬🫀
doi.org/10.1093/eurhea…


English
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🫀 ESTUDIO CADENCE
Sotatercept en hipertensión pulmonar asociada a insuficiencia cardíaca: nuevos datos que generan preguntas clave para la práctica clínica.
📊 Análisis del Dr. Alberto Fernández para @cifacah
siacardio.com/sotatercept-pa…

Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🧬 A novel PPP1R13L variant in paediatric cardiomyopathy: expanding the spectrum of arrhythmogenic disease
A recent case report published in Cardiology in the Young describes a striking and clinically relevant scenario:
👉 a novel homozygous frameshift variant in the PPP1R13L gene associated with syndromic dilated cardiomyopathy and fatal arrhythmias
💡 Why does this matter?
Paediatric dilated cardiomyopathy (DCM), although rare, carries a high burden of:
heart failure
transplantation
sudden cardiac death
Increasingly, genetic causes are central, particularly in early-onset and syndromic presentations.
🧒 Case highlights
4-year-old boy presenting with severe heart failure
Rapid progression with:
biventricular dysfunction
malignant ventricular arrhythmias
Syndromic features:
sparse, dry hair
high anterior hairline
broad nasal bridge
abnormal dentition
Fatal outcome due to ventricular fibrillation
🧬 PPP1R13L and disease mechanisms
PPP1R13L encodes iASPP, a protein involved in:
desmosomal integrity
apoptosis regulation (p53 pathway)
inflammatory signalling (NF-κB pathway)
👉 Loss of function leads to:
structural myocardial instability
maladaptive remodelling
a strong arrhythmogenic substrate
📌 This condition is increasingly recognised as a form of arrhythmogenic cardiomyopathy with a dilated phenotype and extracardiac features.
⚠️ Clinical implications
✔️ Consider PPP1R13L in children with:
early-onset DCM
ectodermal/syndromic features
consanguinity or suspicious family history
✔️ Arrhythmias may be:
👉 early
👉 severe
👉 disproportionate to ventricular dysfunction
✔️ Persistent troponin elevation may reflect:
ongoing myocardial injury
chronic inflammation
disease progression
🔍 Take-home message
This case:
expands the genotype–phenotype spectrum
reinforces the concept of paediatric cardiomyopathies as systemic genetic diseases
highlights the importance of early recognition and multidisciplinary care
👉 Early diagnosis is crucial for:
arrhythmia surveillance
tailored clinical management
family counselling
doi: 10.1017/S1047951126111883

English
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

📢 Call for Papers – Special Issue
I am pleased to invite submissions to our Special Issue on
“Inherited Cardiac Disorders in Pediatrics” in Children (MDPI).
This Special Issue focuses on the full spectrum of paediatric inherited cardiac diseases — from genotype to clinical outcome, including:
🧬 Genetic basis and novel variants
🫀 Cardiomyopathy phenotypes and arrhythmias
🧲 Advanced imaging (CMR, strain, LGE)
⚡ Risk stratification and early detection
👶 Family screening and personalised management
We welcome original research, reviews, and clinical perspectives that advance our understanding and improve care for children with inherited cardiac conditions.
🔗 Submit your work here:
mdpi.com/journal/childr…
Please feel free to share this with colleagues who may be interested.
#Cardiology #Pediatrics #Cardiomyopathy #Genetics #CMR #Arrhythmia

English
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🫀 Sepsis-induced cardiomyopathy is not rare. It is under-recognized.
And more importantly:
👉 It is dynamic, reversible… and frequently misinterpreted
⚠️ The clinical trap
You see a septic patient with:
✔️ Hypotension
✔️ Vasoplegia
✔️ “Normal” or even high LVEF
👉 And you assume the heart is fine
❌ Wrong
🧠 What septic cardiomyopathy really is
Sepsis-induced cardiomyopathy (SICM):
▪️ Acute, reversible myocardial dysfunction
▪️ Can affect LV, RV, systolic and/or diastolic function
▪️ Not related to coronary disease
▪️ Often unmasked after resuscitation
👉 It may appear hours to days after ICU admission
🔥 Phenotype matters!
SICM is NOT one disease.
It is a spectrum of hemodynamic phenotypes:
🟠 LV systolic dysfunction
🔵 Hyperdynamic vasoplegic state
🟣 RV failure
🟡 Persistent hypovolemia
⚪ Apparently “normal” profile
👉 Each requires a completely different treatment strategy
🚨 Why LVEF misleads you
In sepsis:
➡️ LVEF depends on afterload
➡️ Vasoplegia can artificially increase LVEF
➡️ “Normal EF” ≠ normal contractility
👉 Ventriculo-arterial coupling is the real issue
🫀 Pathophysiology
Sepsis affects the heart at every level:
▪️ Cytokine storm → myocardial depression
▪️ β-adrenergic downregulation → poor response to catecholamines
▪️ Calcium handling dysfunction → ↓ contractility
▪️ Mitochondrial failure → ↓ ATP
▪️ Microcirculatory dysfunction → impaired perfusion
👉 Yet: minimal cell death → reversibility is possible
⚡ Clinical reality
SICM can:
✔️ Develop early OR late (up to 72h)
✔️ Mask or mimic other shock states
✔️ Coexist with AMI
✔️ Flip phenotype during resuscitation
👉 This is why single measurements are dangerous
🧩 Management principle
There is no universal treatment
Instead, Treat the phenotype, not the label
Examples:
🟠 LV failure → consider inotropes (carefully)
🔵 Hyperkinetic vasoplegia → vasopressors, avoid overload
🟣 RV failure → ventilatory strategy + afterload reduction
🟡 Hypovolemia → guided fluids
👉 Echocardiography is the cornerstone
🚀 Where the field is going
Future management will rely on:
▪️ Hemodynamic phenotyping
▪️ Biomarker + immune profiling
▪️ AI-guided decision making
▪️ Personalized therapy
👉 Not “one-size-fits-all sepsis bundles” anymore
🎯 Take-home
Septic shock is not only a vascular disease, It is a cardiovascular syndrome with multiple phenotypes
And if you miss the cardiac component You miss the patient ⚠️
📚 Aissaoui N et al. (2025)
European Heart Journal
DOI: 10.1093/eurheartj/ehaf340

English
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🧬 LMNA cardiomyopathy: not all patients progress the same.
Now we can quantify who will.
We’ve become good at predicting:
👉 sudden death
👉 arrhythmic risk
But heart failure?
👉 Still a blind spot in laminopathies
💥 This paper changes that.
For the first time:
👉 a dedicated risk model for HF progression in LMNA patients
📊 The reality (and it’s not reassuring):
• HF-MACE in ~10% at 5 years
• ~45% lifetime risk by age 70
And if LVEF <30%:
👉 50% events within 1 year
ehag104
⚡ Four variables. That’s it.
Independent predictors of HF progression:
• Male sex
• LVEF <50%
• Missense variants (head/rod domains)
• Complete LBBB
🔥 Translation:
👉 Genetics + conduction + early LV dysfunction
= the HF trajectory is already written
📉 What’s really striking (Figure 4):
• 0 risk factors → ~1.5% at 5 years
• 1 factor → ~5%
• ≥2 factors → ~22%
➡️ Not a spectrum
➡️ A stepwise risk escalation
🧠 And here’s the key insight:
👉 Even with preserved EF, risk is not low
Patients with LVEF ≥50% + risk factors:
→ still meaningful HF risk (~11% at 5 years)
ehag104
⚠️ This challenges our mindset:
We wait for EF to drop
👉 But the disease is already progressing
💡 Clinical implication (this is the real shift):
Risk stratification in LMNA should not be:
❌ reactive
❌ EF-driven
But:
👉 proactive + genotype-informed
🚀 What this enables:
• Earlier follow-up intensification
• Earlier HF therapy
• Earlier referral for advanced HF pathways
• Better selection for trials
🎯 Take-home message
LMNA cardiomyopathy is predictable.
👉 Not just arrhythmias
👉 Heart failure too
And if we wait for severe dysfunction…
👉 we are already late
💬 Should we start treating LMNA patients before EF declines?
#Cardiology #LMNA #Cardiomyopathy
#HeartFailure #Genetics
#PrecisionMedicine #EPeeps #CardioTwitter 🧬🫀
doi.org/10.1093/eurhea…

English

@javibalmaseda1 Creo Oscar, Drazen gran jugador de equipo gran asistidor y obvio tiraba como los dioses talvez más completo , pero tirador creo Oscar pero lindo tema para pensar
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

🫀🧬 Rare Cardiomyopathies: Beyond the Usual Suspects
A comprehensive position paper from Italian Societies (SIC & SICP) provides practical guidance on diagnosis and management of rare cardiomyopathies across both paediatric and adult patients 👇
💡 Why this matters:
Rare cardiomyopathies often mimic common phenotypes (HCM, DCM, RCM, ACM)…
👉 but require completely different investigations and treatments
⚠️ The challenge:
🔹 Highly heterogeneous & multisystem diseases
🔹 Often diagnosed late
🔹 Need tailored, multidisciplinary care
🧠 Key concepts from the paper:
👩⚕️ 1. Multidisciplinary team = ESSENTIAL
👉 Cardiologists + geneticists + neurologists + psychologists + surgeons
👉 No single specialist can manage these patients alone
🧬 2. Genetics is central
✔️ Genetic testing recommended in all cardiomyopathies
✔️ Family screening & counselling are crucial
🧪 3. Go beyond imaging
Diagnosis requires:
🔹 ECG + Echo + CMR
🔹 Holter monitoring
🔹 Metabolic & genetic testing
🔹 Detailed family history (3 generations!)
🚨 4. Think “red flags”
Rare causes should be suspected when:
👉 Multisystem involvement
👉 Early onset (especially infancy)
👉 Unusual features (e.g. pre-excitation, metabolic signs)
👶 Paediatric insight:
🔹 Infant HCM is often non-sarcomeric (metabolic, syndromic)
🔹 Prognosis strongly depends on underlying aetiology
👨⚕️ Adult insight:
🔹 Always rule out treatable phenocopies
👉 Cardiac amyloidosis
👉 Fabry disease
🚀 Take-home message:
✔️ Rare cardiomyopathies are not that rare in practice
✔️ Early recognition = life-changing (and sometimes life-saving)
✔️ Precision medicine starts with thinking beyond the obvious
❤️ This paper reinforces a key idea:
👉 “One phenotype ≠ one disease”
📄 Based on: doi.org/10.1016/j.ijca…
#Cardiology #Cardiomyopathy #RareDiseases #Genetics #PrecisionMedicine #PediatricCardiology #CMR #HeartFailure #MedTwitter #ClinicalPractice #InheritedDiseases

English
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

2 Monstruos, el mejor extranjero, Oscar, que ví jugar en Uruguay.. y @4_TatoLopez el uruguayo más grande que he visto en una cancha de básquet !!!
Dr Gabriel Parma. Profesor adjunto de cardiologia@parma315
Se va un pedazo de mi infancia , el mejor de los mejores , un animal , no habrá otro igual , esta foto me recuerda todo lo que amo el Basket . Salud campeón
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

@NBAdabad Una bestia !!! No habrá otro
Español
Dr Gabriel Parma. Profesor adjunto de cardiologia retweetledi

@altamirano45 De los mejores de la historia , un monstruo
Español








