Paul D. Rennert
20.5K posts

Paul D. Rennert
@PDRennert
Co-founder, President & CSO, Aleta Biotherapeutics, https://t.co/vyLXDleOdp - Founder & Principal, SugarCone Biotech, commentary at https://t.co/nE18E8qx8B





no one is focused on allo

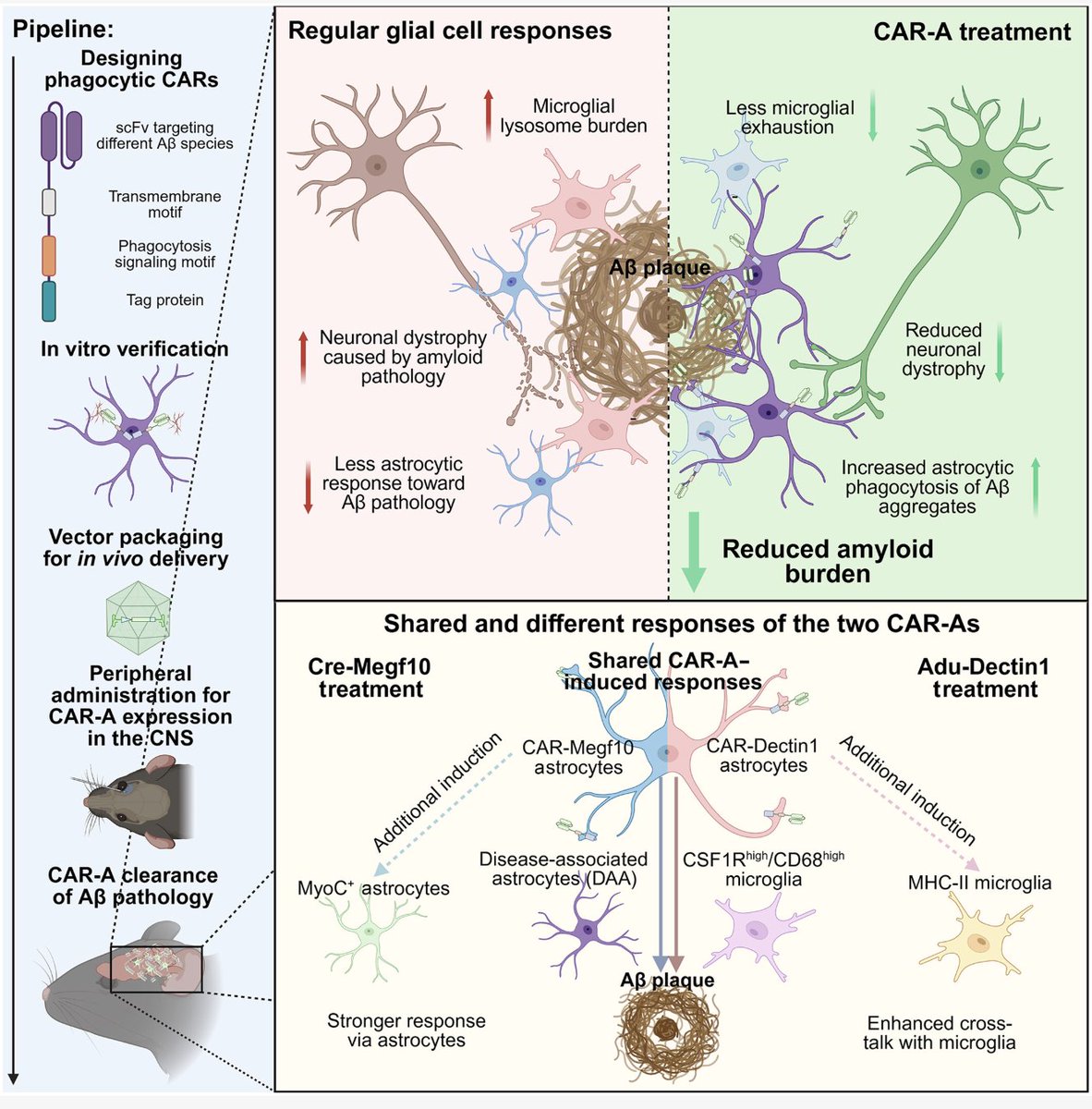
Everyone is focused on allogeneic as the fix for CAR-T access. But a Nature paper this month made a stronger case for something else entirely: skip the manufacturing step altogether. In vivo CAR-T is coming. And it could make the autologous vs. allogeneic debate irrelevant. nature.com/articles/s4158…







More exceptional heat pulses from West to East are expected next week with widespread 100s again in the desert areas and parts of the Central States Not even the Dust Bowl heat waves or anything else ever seen so far can compare to this: All climatologists are baffled.


an interesting follow-on question is what level of sustained efficacy do CAR-Ts need to show in refractory autoimmune disorders like gMG, CIDP, versus anti-FcRn or anti-C1s antibodies, especially if long-term rituximab maintenance is needed to maintain the CAR-T-induced response

interesting and different efficacy outcomes in ASyS and SSc autoimmune patients treated with blinatumomab (CD3/CD19) and teclistamab (CD3/BCMA), respectively, + Rituximab maintenance. Potential for targeting both BCMA and CD19, especially in ASys. nature.com/articles/s4159…