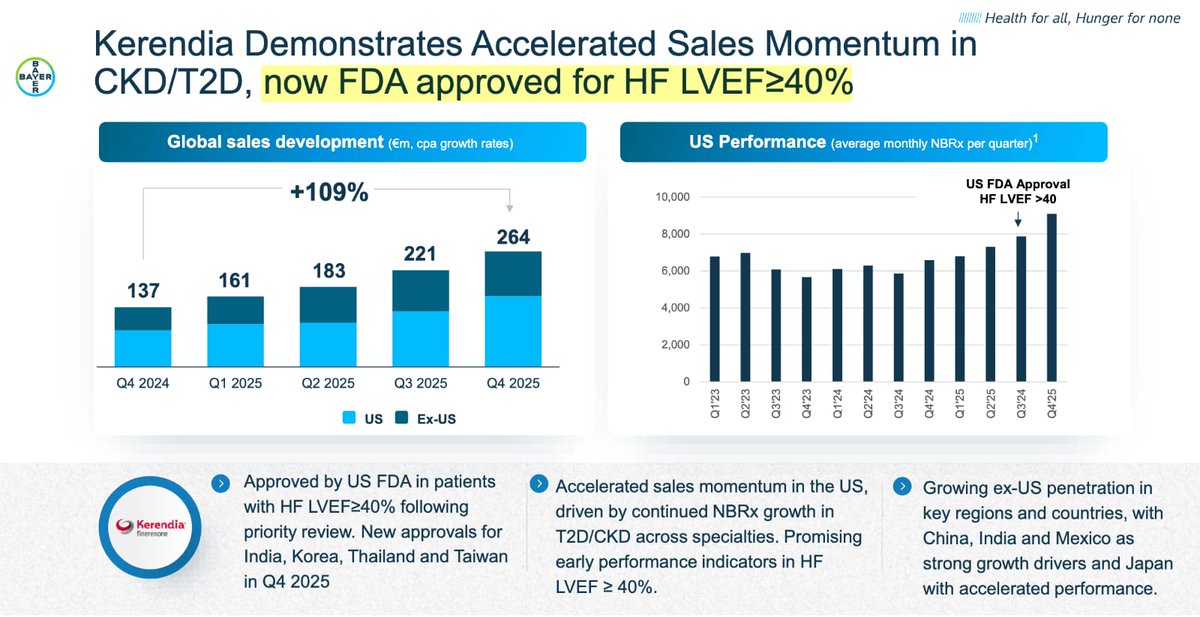
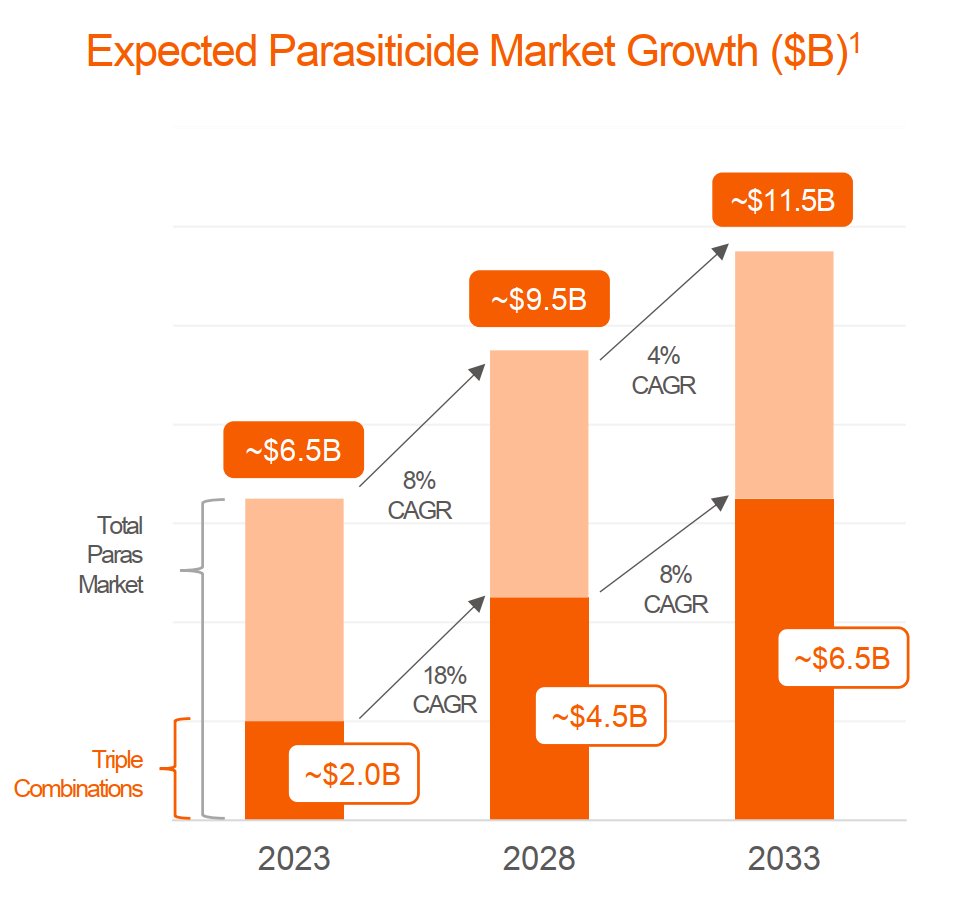
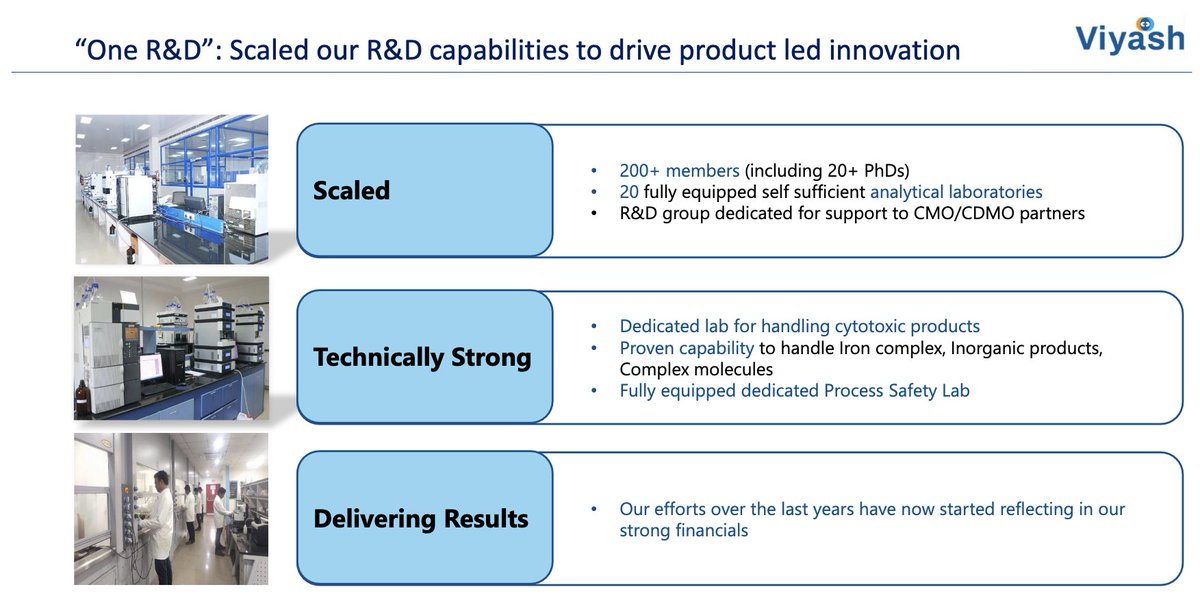
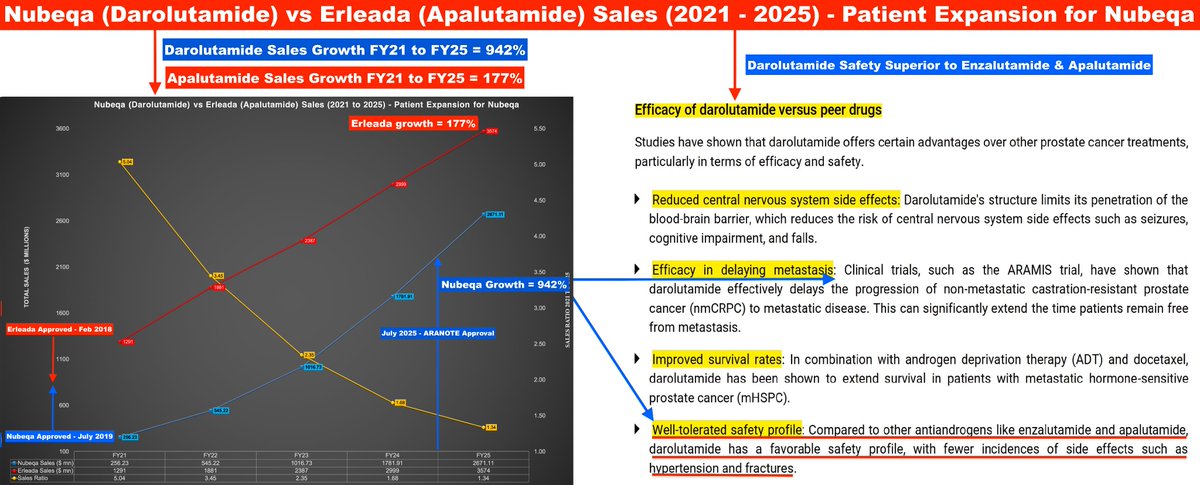
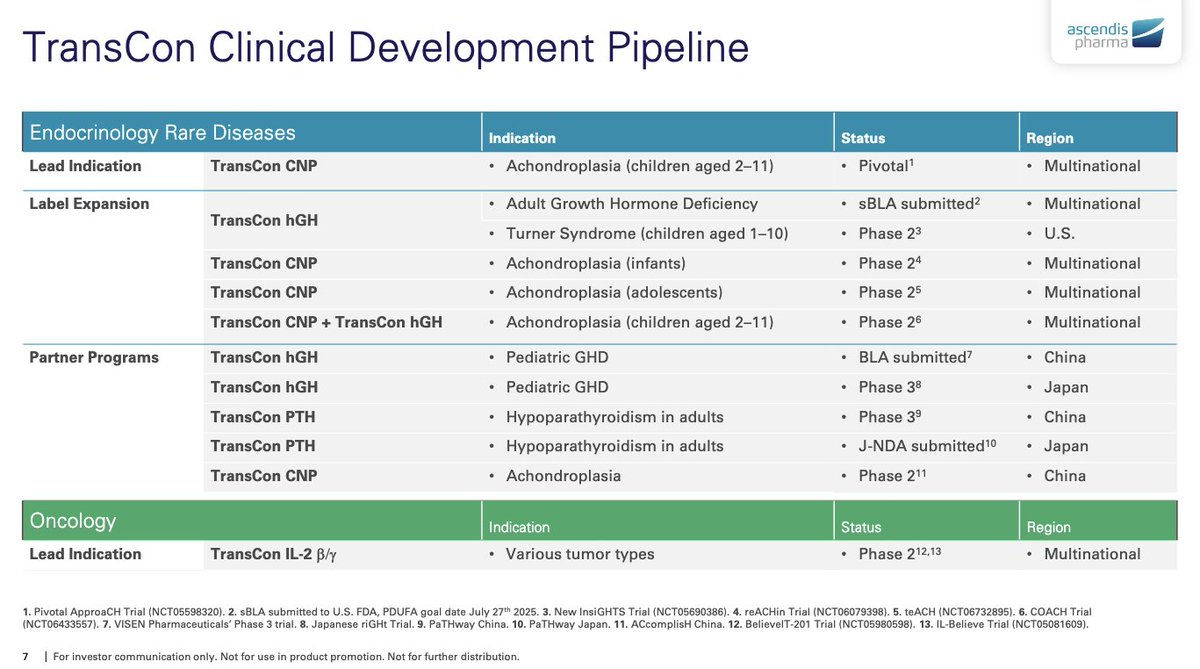
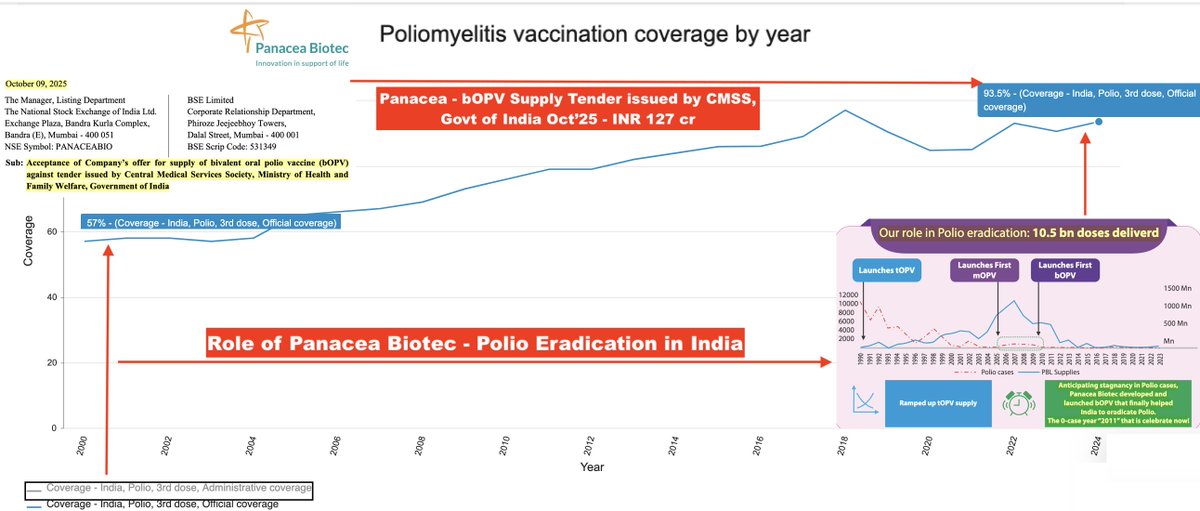
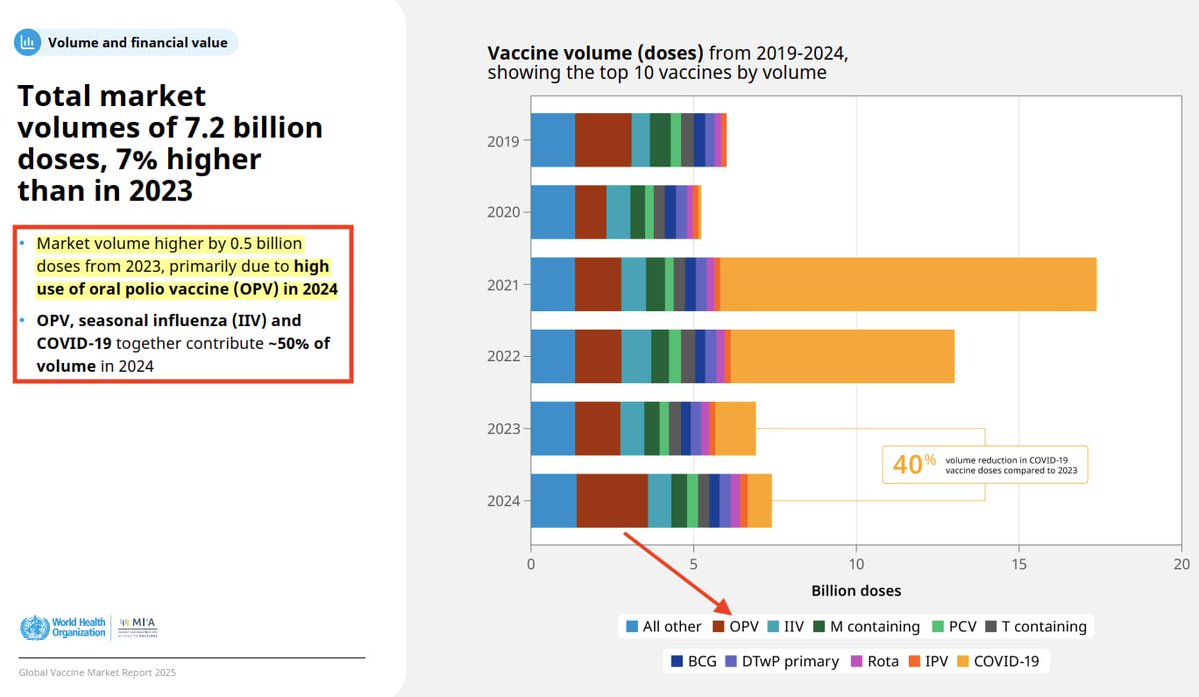
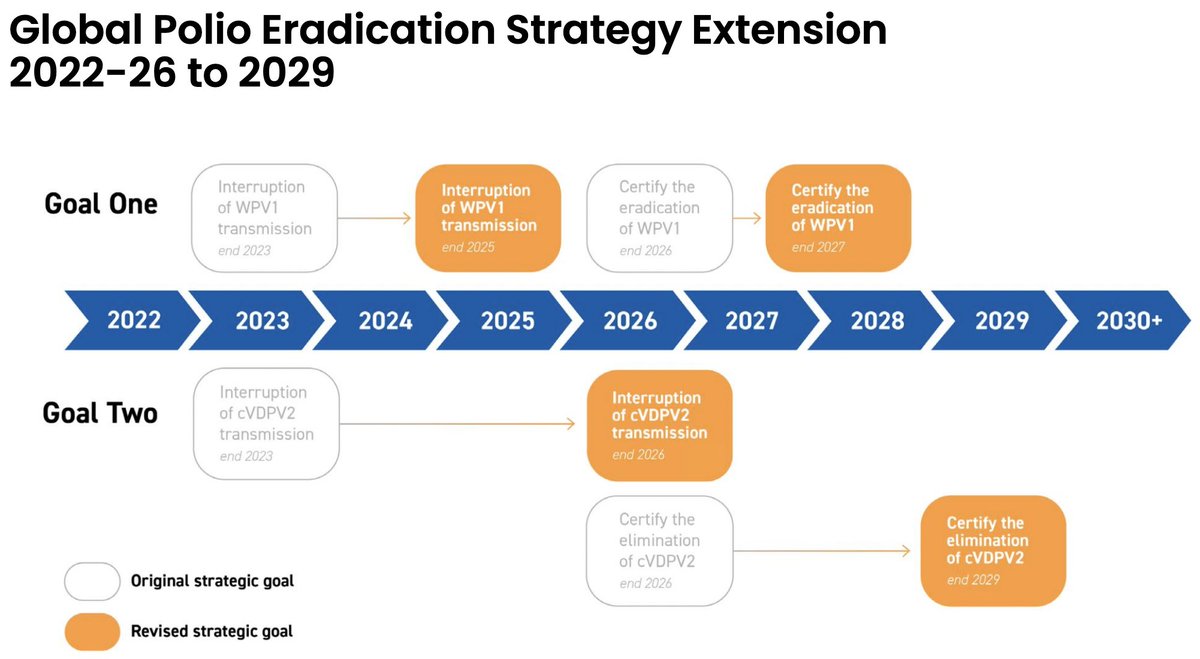
Vaccine Characteristics | Dengue Serotypes | Efficacy & Side-Effects
The Global Dengue Vaccine Landscape & Characteristics
📸1: DengiALL (Preclinical Study) - Successful In the Rhesus Monkey challenge study - conducted by NIH, USA against all 4 Dengue serotypes. (Ref: Panacea Biotec)
📸2: DengiALL (Clinical Study) - Early clinical data from Phase I/ll trials in healthy Indian adults showed that a single dose generated strong immunogenicity against all 4 serotypes, while maintaining a favorable safety profile.
Qdenga Limitations (Efficacy & Side-Effects)
📸3: Dengue is caused by 4 serotypes (DENV-1 to DENV-4). While TAK-003 (Qdenga) is highly effective against DENV-2 (its genetic backbone), its efficacy is notably lower against DENV-3 and DENV-4
📸4: Other Dengue Vaccines & Qdenga Age group related side-effects
Dengvaxia®: Production is being discontinued in 2026.
Qdenga®: In clinical studies, the most frequently reported reactions in subjects aged 6 to 45 years of age were injection site pain (54%), headache (36%), myalgia (34%), injection site erythema (29%), malaise (24%), asthenia (21%) and fever (10%)




English