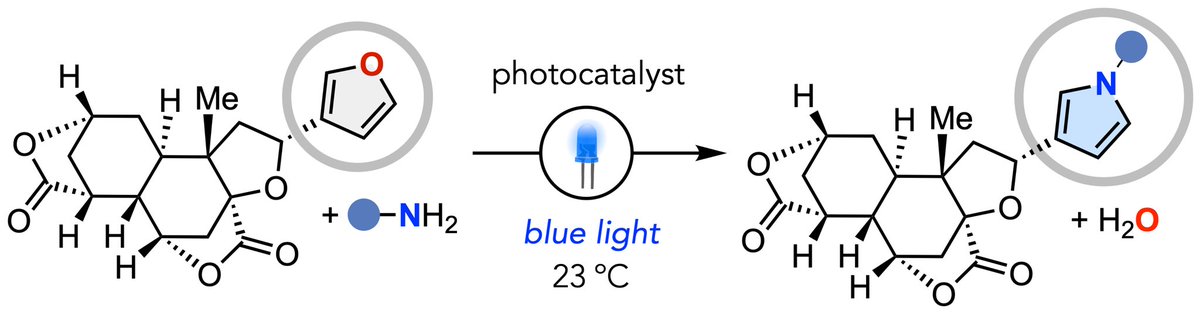
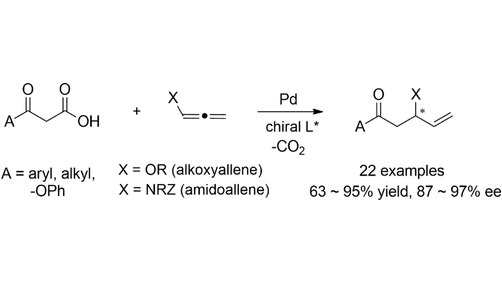
Sukhyun Lee retweetledi

Pd-Catalyzed Counter-Steric Site- and Chemoselective Glycosylation: Total Synthesis of Fridamycin A and Himalomycin B | Journal of the American Chemical Society @postech2020 pubs.acs.org/doi/10.1021/ja…
English
Sukhyun Lee
15 posts