
$RCKT better get FDA approval and respond with a huge move or a lot of money is going to be zeroed on these super heavily leaning call positions. 260% IV
singhlosophy
426 posts


$RCKT better get FDA approval and respond with a huge move or a lot of money is going to be zeroed on these super heavily leaning call positions. 260% IV

$wve i (virtually) participated in the study just to show you what a 3% smaller waist looks like in real life. I am wearing here the same belt btw.

$WVE The claim that fat loss was balanced out with lean mass gain is the most ayurvedic level nonsense I've ever read. Put it in the Louvre for bullshit pseudoscience claims. This is absolutely a short, not immediately, but in short order. It will never be approved. Hilarious.

I didn't realize what a recovery $RPRX has gone through for the last 15 months. 2023 and 2024 were non stop selling and 2025 was a massive turnaround. April 5 Tividenofusp BLA for Hunter $DNLI is probably the better way to play it but they've been on quite a run as well.

There are some real trash stocks being pumped right now. $AVXL is definitely one of them. Up 30% in the last month. Puts are cheap. $1 is the first target.

$PASG with a hell of a wipeout year so far -62% wiping out the entire pump and chasers. This was a very popular biotwit position last year.

$JHG for $46? That's an awful deal. No way any shareholders will accept that. No premium and losing their dividends, no thanks. $60 is the minimum based on comparative market players.

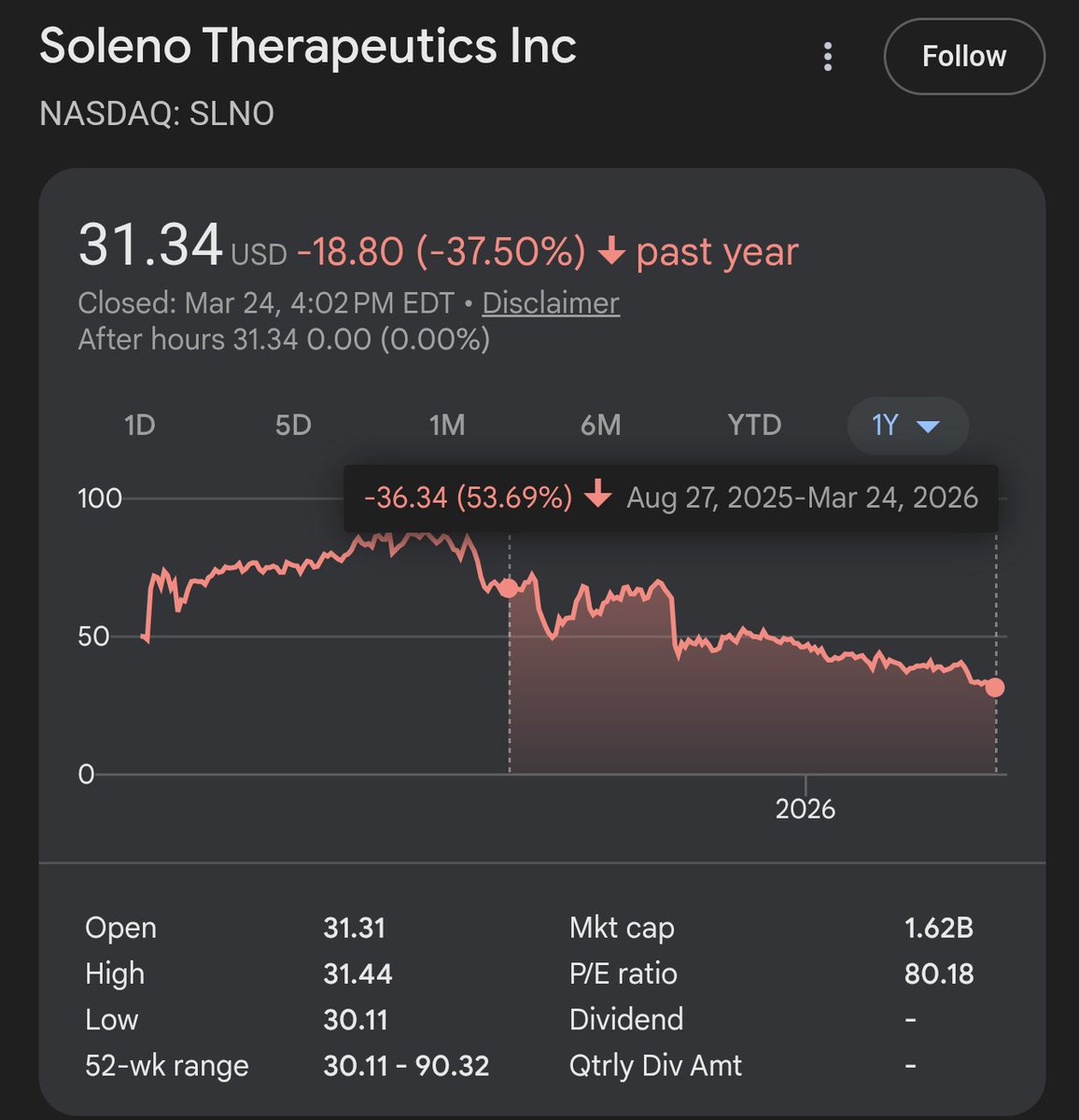
1/2 $SLNO Soleno Therapeutics - Mgmt stock sales exhibit serious red flags. The speed and magnitude of stock dumping within days of FDA approval strikes us as rare and unusual, like many things w this company. Insiders appear a lot less bullish about their drug’s prospects than investors, based on our analysis of their trading behavior. Several elements signal, to us, an extreme haste to sell upon approval vs. waiting around for drug launch. We suspect that investors who bought into the equity offering in July at $85/share were unaware of the nature of these sales, and were thus played like fools. 1. Over two consecutive trading days starting on 03/27/25 (FDA approval was 3/26) we calculate that seven insiders collectively sold 859,163 shares for total net proceeds of $47.9 million. Please see disclosures at end. 2. After initially placing 300,000 shares into his 10b5-1 plan last September, CEO Bhatnagar *increased* his sell-down to 523,809 shares which reduced his holdings by 40%. $29mm proceeds. The regulatory and commercial heads reduced their holdings by ~65%. The commercial head had been at SLNO for only ~14 mos, so she strikes us as downright nervous about the near-term. 3. The lead independent director (Pauls) sold 90% of his holdings. He’s CEO of a rare disease company focused on respiratory diseases, so we presume he recognizes the gravity of pulmonary edema as an adverse effect. 4. The stock awards smack of self-serving behavior as the officers were loaded up with equity right before the NDA submission date and then cashed in $58 million worth of stock (gross) as soon as the process was completed just eight months later. The awards suggest to us a board/investor group fixed on incentivizing FDA approval vs. launch. In July 2024, the board awarded the six executive officers with a one-time performance stock award. The officers were issued a total of 1,195,000 shares with a combined market value of $58.1 million, with the CEO receiving 850,000 of them. For most officers, these shares would vest 25% *just two weeks later* on 08/01/24, which was weeks before the FDA accepted the Company’s NDA and granted priority review. Another 25% were to vest on the acceptance date, and 50% on the date the FDA approves the drug. It is remarkable that these awards were granted so late in the submission process, and that a quarter of these so-called performance shares vested almost immediately without any performance requirement. 5. The trading appears to have played fast and loose with the company’s own insider trading policy. The Company announced the FDA granted priority review of DCCR in late August 2024, and over the next two weeks Rule 10b5-1 plans were set up by CEO Bhatnagar, CFO Mackaness, SVP of Regulatory Hirano, and SVP of Clinical Operations Yen. These four plans covered a total of 579,115 shares which was almost 50% of the entire 12-persons insider group’s combined ownership at the time. Given that 10b5- 1 plans are required to “cool” for 90 days before they can start selling, the SLNO officers’ plans were timed to become effective just in advance of the 12/27/24 PDUFA date. But then the FDA announced an extension of the review period, and it set a new PDUFA date of 03/27/25. The CEO and CFO responded by terminating their Rule 10b5-1 plans in 1Q25 and they did not initiate new ones. They essentially removed the 10b5-1 shackles after presumably having further discussions with the FDA, and they appeared to have been ready and eager to sell as soon as VYKAT XR received the FDA’s blessing.


BTIG must be the most cocaine driven fund out there for biotechs. Every one of their price targets is some ridiculously high number 5x, 8x, 10x the current price. Or in $NRXP case: 12.5x

$IBRX having a market cap equivalent to $ARWR $AXSM $JAZZ $ABVX is laughable. Not only that. It's bigger than $CYTK $RYTM $BIO $BLCO $GPCR $MIRM just to name a few. The Saudis are always behind some of the shadiest nonsense.