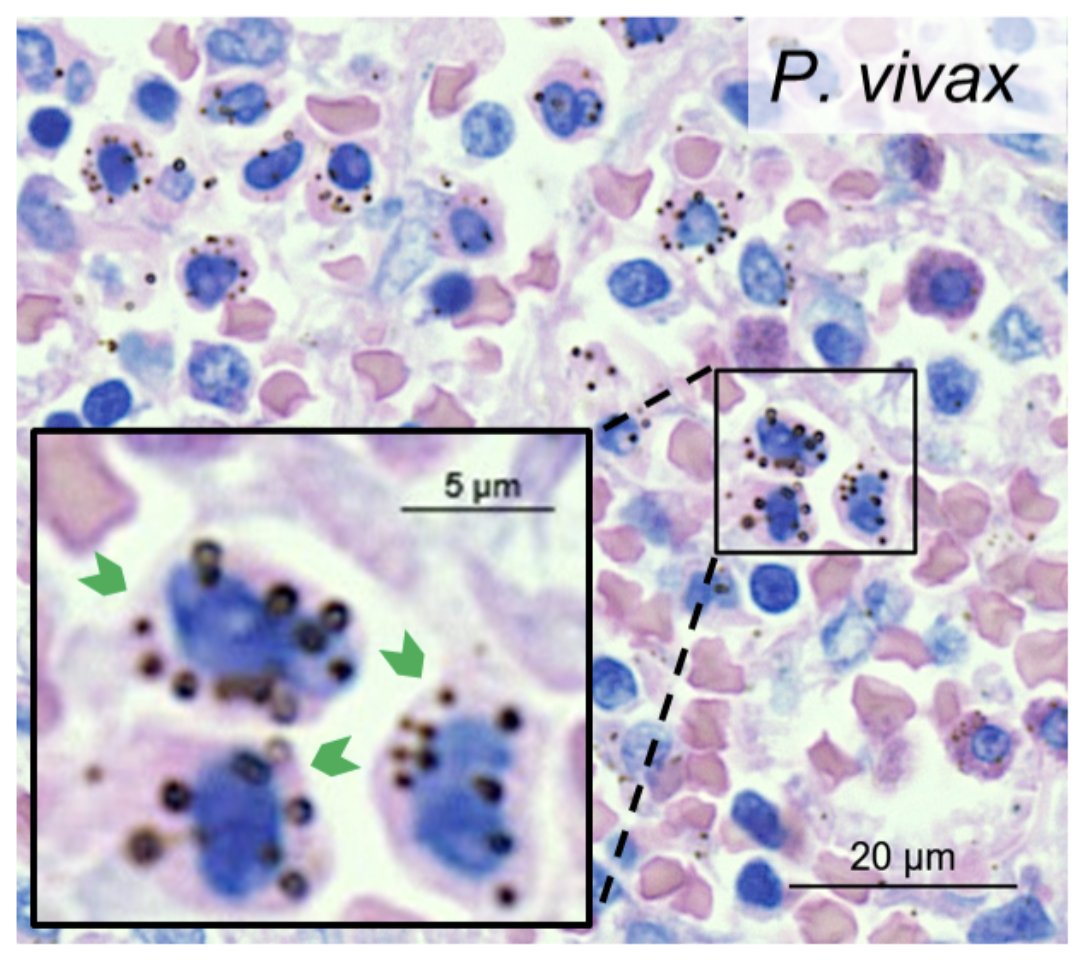
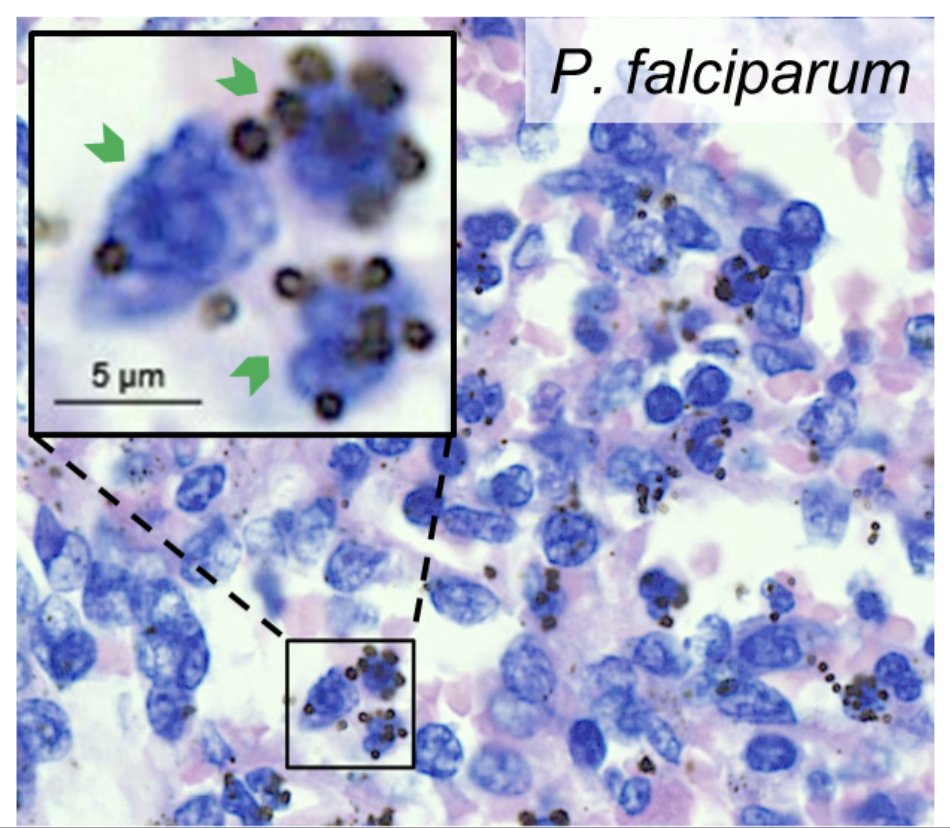
Dennis odera
487 posts




Hearty congratulations are due our newly minted DOCTOR Nancy Mbeyu on the successful defence of your PhD thesis! And to your supervisors led by Prof Caroline Jones @IDeAL_KEMRI_WT @KEMRI_Wellcome @AlunIwanDavies @mbeyuwm








A fascinating work is published today @Nature by Zhang & Zhang et al. on a serendipitous discovery of haemoglobin expression outside the RBC cells--in chondrocytes--and their critical role in keeping our cartilage alive under an oxygen-deprived environment. nature.com/articles/s4158… Our school teachers taught that haemoglobin (Hb) is an oxygen-transporting protein expressed exclusively in the red blood cells. Although there exist occasional sporadic reports of Hb expression outside the RBCs (e.g. neurons, retinal cells etc.), no one would have guessed Hb would play a critical role (required for survival) outside the RBCs. The discovery When studying the cartilage growth plate of neonatal mice, the authors noticed eosin-positive structures in the chondrocytes (cartilage cells) that resembled structures seen in RBCs. Out of curiosity, the researchers went on to stain and examine the chondrocytes of different cartilage tissue types both from mice and humans. They realized that no matter the source or species, the cells always displayed eosin-positive structures under the microscope. The curious researchers were determined to find out what these structures are made of. They carefully dissected these structures out and studied the protein components using mass spectrometry and were surprised to learn the results: the top hits were Hb proteins. Unable to believe the results, they went on to study the proteins using different methods--western blotting, immunohistochemistry--and every time they ended up with the same results: the cytoplasm of cells was loaded with organelles-like bodies made of haemoglobin proteins. Finally, they came to the realization that Hb is abundantly produced in chondrocytes. They named these cytoplasmic Hb bodies as "Hedy". Structure of Hedy The authors studied the structure and formation of Hb bodies floating in the cytoplasm. Are they like an organelle? Do they have a membrane? Through various experiments, the researchers found that the Hedy structures do not have a membrane. The Hb proteins condense together by phase separation to form organelle-like structures in the cytoplasm. This condensation is itself an evolved process, requiring specific sequence structures of the Hb protein. Globin switching We know that there are different forms of Hb each expressed during different developmental stages: embryonic, fetal and adult Hb. There exists a sophisticated molecular machinery (which was believed to be RBC-specific) that switches one Hb type to the other at appropriate times. Using gene silencing experiments, the authors were further awestruck to find that the chondrocytes too switched their Hb types from embryonic to fetal to adult stages, just like RBCs! Regulation of chondrocyte Hb production It is well known that hypoxia induces Hb production via upregulating hypoxia-inducible factors (HIFs), an evolutionarily conserved molecular mechanism (Nobel Prize 2019; nobelprize.org/prizes/medicin…). But it turned out that chondrocytes have evolved to increase their Hb expression not via HIF proteins, but through a different protein, the same protein that is required for fetal to adult Hb switching: KLF1 (nature.com/articles/ng091…). How important is Hb for chondrocytes? Such a high Hb expression in chondrocytes with similar globin switching behaviour as RBCs would mean that this Hb is critical for chondrocytes' survival. By deleting the Hbb gene specifically in the chondrocytes, the authors found that without Hb the chondrocytes die killing the animal a few days after birth. Conclusion Continuous oxygen supply is a prerequisite for the survival of cells in all tissues. The only way the cells can receive oxygen is through RBCs in the blood, which requires the tissue to be highly vascularized. When demand exceeds the supply, the cells evolve to survive an oxygen-depriving environment. Muscles evolved to produce their own globin--myoglobin which has a higher affinity to oxygen than Hb thereby withholding O2 during oxygen excess states and releasing it back during oxygen-deprived state (during exercise). Likewise, the brain has its own globin: neuroglobin (nature.com/articles/35035…). Today, we are learning that cartilage (an avascular tissue), too, has its own globin. But unlike muscle and brain, have evolved to store oxygen not by making a new type of globin but by making just the same type as the ones in RBCs, but with a higher affinity than RBC Hb. When it comes to fundamental biology, we often assume that we have found everything and then one day a discovery like this drops, hitting us on the head to make us realize that there is a whole universe of hidden biological secrets waiting to be discovered. Some recent posts: 1. Gene x sex interaction of PNPLA3 I148M variant (x.com/doctorveera/st…) 2. Effect of consanguineous marriage on the risk of common diseases in offsprings (x.com/doctorveera/st…) 3. Whole genome vs. Whole exome sequencing. Which is more cost-effective for genetic association studies? (x.com/doctorveera/st…)