ทวีตที่ปักหมุด
AscendingBio
1.5K posts


AscendingBio
@AscendingBio
biotechnology stock investor
USA เข้าร่วม Ocak 2025
62 กำลังติดตาม1.1K ผู้ติดตาม

They have stated that they plan to add a QR code with video link. I really expect to see that in the final version. My guess is that during the upcoming human factors studies the FDA would not have permitted persons to access that video.
So, they wait and present this during final labeling discussions.
English

Harry, you said it better than me.
I actually think it would be interesting to have a class centered on this company or at least a class project. There are so many teachable moments from stats to research design to ethical ways to speak about medical results to critical thinking.
There are many good illustrations here of not reporting data in normal ways. Students need to figure out what is missing and the consequences of that data being omitted.
They put emphasis on offering a cure when their strength may actually be a rather modest boost to the efficacy of many cancer treatments. They trash chemo when their drugs might work best when used with chemo. Newer immunotherapy treatments like off-the-shelf CAR-T may not need what they have to offer.
Learning from real life examples is typically more interesting.
English

@AscendingBio @galactiator I am one of the PhD level professors you had.
I’m not sure you get my assertion
1. Post Hoc is good for hypothesis building, but it’s terrible for assertions
2. The person arguing has no clue what they are talking about
English

$IBRX Accelerated Approval for NSCLC? (Part 3 of 3)
I often see posts calling for the FDA to immediately grant accelerated approval to Anktiva for Non Small Cell Lung Cancer (NSCLC). The focus for $IBRX has been 2nd line treatment. (They ended a frontline study.) In a phase 2, Dr. Patrick verbally reported that Anktiva plus a checkpoint inhibitor had a median Overall Survival of 14 months.
Take a look at slides 22 and 23 in this presentation by Iovance. ir.iovance.com/static-files/5… They found that at 24.5 months after treatment the median duration of response has not been met. That means more than half are still alive at 24.5 months and maintaining the response to treatment. So, the Iovance results are likely to be years more of survival than ImmunityBio.
Yes, TIL therapy is more challenging, but it is worth it with results like this. They do not have approval yet from the FDA for this type of cancer. They do not protest or go overseas. They work to fully enroll the study before going to the FDA. With more data like this, the Accelerated Approval path should be open to them. They are also advancing new versions that are even more effective.
A recent study compared Dato-DXd to Docetaxel in the same context (previously treated NSCLC without actionable genomic alterations). They found Median PFS: 4.4 vs 3.7 months (HR 0.75, P=0.004). Median OS: 12.9 vs 11.8 months (HR 0.94, P=0.530 – not statistically significant). They dropped the drug from this indication after getting an OS of 12.9 months. ImmunityBio took their 14 month OS results to Saudi Arabia and sought approval. On the last call, Dr. Patrick proclaimed this result was amazing and they had doubled median OS from 7 months to 14. If you do not study competitors, this sounds good.
There are several reasons the FDA will not accept Anktiva under accelerated approval for NSCLC at the current stage. One is that the phase 2 study used Anktiva and a checkpoint inhibitor. The phase 3 study utilizes Anktiva, a checkpoint inhibitor, and Docetaxel (chemo). That is compared to Docetaxel alone. This resets everything when you make that kind of a change. If you thought the phase 2 combo was good, why did you change it in the phase 3?
Accelerated approval by FDA rules include using one variable that you establish as a predictor for a variable you will not know until the future. That does not apply here. ImmunityBio already has Overall Survival data for the phase 2.
It might be possible to submit the new triple combo data to the FDA a year or so after full enrollment using the phase 3 interim results under Accelerated Approval. Maybe, you have progression free survival data by the 10-month mark. It would take dramatically different progression free survival numbers for treatment versus control. It would need to appear that Overall Survival numbers for the treatment group would not be available for a year or more.
If it looks like the treatment group is likely to have median survival of about 14 months and 7 months for the control, why not wait another 6 months and submit the Overall Survival numbers? That OS data is going to be available during the FDA review process. The FDA will likely pause and delay the approval process in order to consider the new data.
Another factor for the phase 3 study is that it is using a 3 drug combo where the design does not lend itself to determining the contribution made by Anktiva. It is possible that the FDA requires another study to determine this contribution. They may say, "It could be that adding the checkpoint inhibitor to Docetaxel is what prompted treatment versus control differences."
I am not ruling out that Anktiva is eventually approved for 2nd line NSCLC by the FDA. My point is that there is not a short-term path.
I encourage you to go check my work. Go read the regulations on the FDA website. Look up other companies that were recently granted Accelerated Approval. Keep in mind that most companies do not submit under the AA program unless they have informal support from FDA staff. If a company is told they likely need more data to support a surrogate endpoint, they will typically not submit until they have that data in hand.
The basic Accelerated Approval structure is based on law passed by Congress. If you want this to be different you need to get new laws passed by Congress.
English

I will not tear down all post hoc hoc analysis. You might learn something from it. Yet, once you do it, you basically hit a big reset button where you have to start the research from the beginning. It is not a shortcut. It sets you way back.
Personally, I get traumatized thinking about what would happen if you tried to get this by the PhD level stats professors I had. There was one in particular who would open up the gates of hell on you if you tried this. It would have been better to insult his mother.
Guys like him trained the statisticians that work at the FDA. I have watched those folks at ADCOMs. They take things like very seriously.
English

@galactiator @AscendingBio @AscendingBio knows… thee words you never want to hear in biotech are, “post hoc analysis”
All we hear is P-hacking, or data dredging
English

I really do not follow your logic here. Concerning RMAT, that is given when there is an illness for which there is great need for improved treatment. They do consider preliminary data that suggests the new treatment might work. Many times it does not. RMAT does not lower the standard for the data required for approval.
RMAT opens the door to more communication with the FDA.
English

The 'confounding' argument is a red herring. We're measuring a drug's effect, not a patient's starting point. Anktiva's IL-15 mechanism causes ALC to rise. That's the point.
The analysis is exploratory- that's why there's a confirmatory trial. This is how AA works.
FDA already validated the approach with RMAT. The rest is procedural noise.
For the lymphopenia indication, the answer requires only a complete blood count.
English

@Mikeptyg They really need to use a different term than floaters to avoid confusion. These are not seen by patients. Doctors need to use a scope to see these outside the field of vision. They are drug fragments that do no harm.
English

@AscendingBio Not sure the weight the SPA holds going into the decision. On paper , the sol-1 has no re-dose data, and SOC control arm. Safety data is ok . But analyst made a good point in asking whether the re-dose will increase floater rate . In general, the bar is high far wetAMD drugs.
English

$VKTX Enrollment Complete Phase 3 SC
78 weeks from today is: September 22, 2027. So, I am expecting early November data.
ObesityWeek 2027 will be held November 8–11, 2027. All signals point to a big presentation of data there.
ir.vikingtherapeutics.com/2026-03-26-Vik…
English

The first trial like this will move slow. Yet, if this appears to work for this illness, things will move faster for other illnesses. They can build off of relationships with hospitals without having to start from scratch. Doors will open to other hospitals with a record of success.
With the first evidence of success, you can start small trials to study other illnesses.
English

@AscendingBio Overall by 2030 or so (possibly earlier) I don’t see how this isn’t a big player in the sector. Battle with US FDA is a huge hurdle that doesn’t look to end anytime soon. Execution will be very pivotal. Ground work looks to be set but as you know , anything can happen .
English

$IBRX Clinical trial to use Anktiva and AgenT-797 iNKT OFF-the-shelf Cell Therapy for those in ICU with severe community-acquired Pneumonia
There are no study locations listed, so it may be some time before the study starts. Setting up a contract with a hospital may take at least 6 months. The patient informed consent aspect of this is complex when you have persons in this kind of a crisis where they are struggling to breathe.
This is single-arm study of 20 persons with no control group. This looks like a good way to start exploring this treatment. Yet, this study just gets the process started.
One issue with using a combo like this is that there will be no way to determine the impact of each drug. The FDA will want to see some data showing that each drug is needed prior to approval.
It will also take a large well-controlled study prior submittal to the FDA for approval.
It is reasonable to think that this treatment could be helpful. Yet, there is significant work to do to collect the data. Years down the road they might have the data to support FDA approval for this indication.
Pressuring the FDA for approval without collecting this data will not work. This is the right way to take a step forward.
clinicaltrials.gov/study/NCT07492…
English

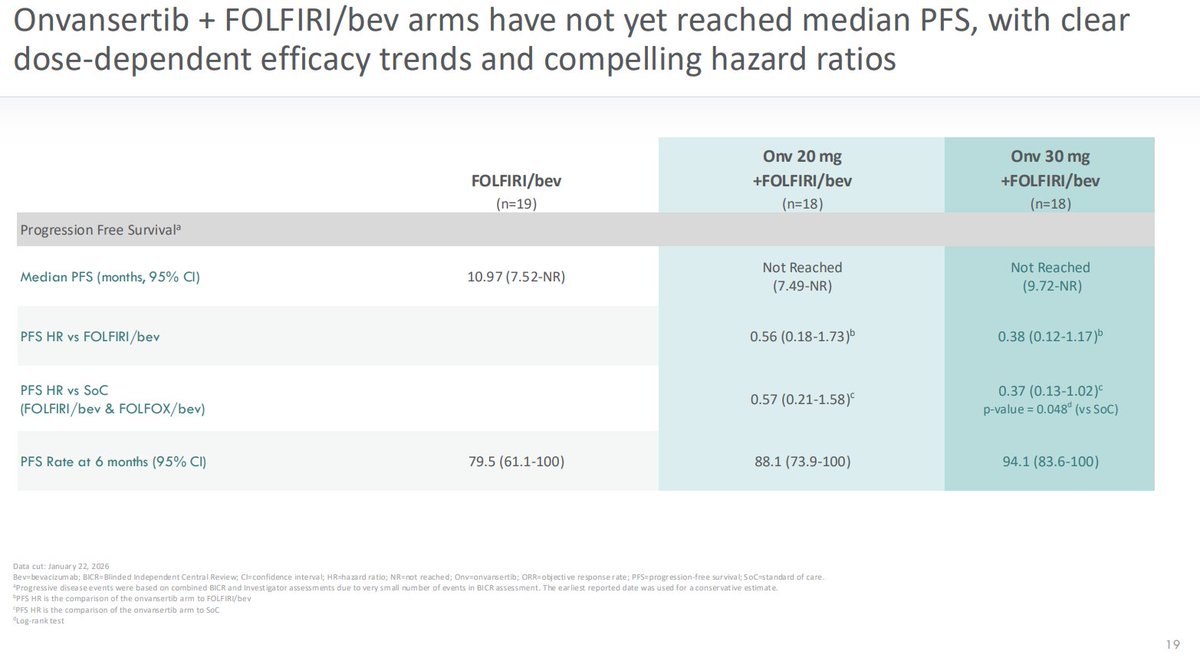
$CRDF Looking Ahead to Late Summer
Today's presentation was a reminder that with a July 22 cutoff of the data in this chart, it will shift from 12 months to 18 months post treatment. The control group had a PFS of 11 months. With a July cutoff, there will be an opportunity to show more contrast with the control. A treatment group PFS value of 17 months or more is what I hope and expect to see.
English

@Mikeptyg I agree that there are unpredictable forces at work at the FDA. V. Prasad leaving should have a big positive impact. Because they have a SPA for SOL-1 with the FDA and this drug is already FDA approved for other applications, I do not expect problems.
English

@AscendingBio Good point and clarification. I’m just not sure if SOL-1 qualifies FDA leadership guidance. Who knows? A lot of gray area to interpret these days with the FDA .
English

@pradracos @InvestorLatin @BIOTECHSCANNER I suggest waiting at least until the next earning.
Yet, to maximize you return, they likely need to turn a profit. That will send the loudest statement to the market. They will probably get close to doing that in the 2Q. The best bet for that happening is the 3Q.
English

@InvestorLatin @BIOTECHSCANNER i have $abeo since $2, was thinking of selling. what is the catalyst now.
English

I appreciate you being very specific in your response.
The key issue is that the design of this study will not answer the question you want it to. The statisticians at the FDA will reject this immediately if it is submitted.
There are many confounding issues here with your approach. One is that persons who are younger and more healthy have higher ALC scores. For example, those with cancer that has impacted the liver likely have lower ALC scores. Some studies have shown that immunotherapy alone tends to not work well with those who have significant liver impact.
(When other companies split into two groups, they report key descriptive stats for each group. We are flying blind here.)
So if you want to break the NSCLC group into two groups, you are going to need to do that prior to treatment. You need to have variables that identify the high-response group in advance. The post-hoc approach is flawed.
Because you are breaking them into two groups in a way that has not been done before, you are going to need a control group to evaluate the results.
If you say, "I don't know how to select the high responders in advance." Then, that takes away your argument for reporting the high responder OS over the pooled OS. I have not heard Dr. Patrick say he knows how to identify high responders - have you?
English

$IBRX
In QUILT-3.055, if the overall ~14.1 month median overall survival (mOS) is the real story, why did the trial protocol (NCT03228667) prospectively list overall survival (OS) prolongation by ALC response as a co-primary endpoint?
Focusing heavily on the pooled~14.1 month mOS from the QUILT-3.055 trial and the early-stopped Lung-MAP S1800D abstract to claim Anktiva “adds nothing” in non-small cell lung cancer (NSCLC) misses the actual study designs and the FDA’s established standards for accelerated approval.
QUILT-3.055: a single-arm Phase 2b trial in heavily pretreated, checkpoint-resistant NSCLC. Every patient received Anktiva plus the same checkpoint inhibitor they had previously failed (true rechallenge setting). The cited overall mOS of ~14.1 months is the average across all participants, including both responders and non-responders.
The trial’s actual co-primary endpoint, defined in the protocol from the start, was OS prolongation by ALC response (achieving/maintaining mean on-treatment ALC ≥1,000 cells/µL). The mature January 2026 data cut showed:
* 77% of patients were ALC responders
* Responders lived a median of 16.2 months (hazard ratio 0.52, p=0.0369)
* Patients reaching even higher counts (≥1,200 cells/µL) lived a median of 21.1 months (hazard ratio 0.33, p=0.0009).
In plain terms, 77% of patients who restored their lymphocyte counts lived substantially longer, with those restoring them to higher levels lived even longer. Responders had roughly half the risk of dying at any given time compared to non-responders, a strong, statistically significant survival benefit. These results meaningfully exceed historical benchmarks of 7-9 months in this refractory setting. The 23% non-responders simply pulled the pooled average down, exactly what one would expect when testing whether Anktiva’s IL-15 superagonist mechanism actually improves outcomes.
The Phase 3 combo change does not “reset everything.” Companies routinely refine regimens during development, especially under RMAT guidance. The surrogate (ALC recovery) remains valid for the broader lymphopenia reversal indication, which is tumor-agnostic.
Lung-MAP S1800D: a randomized Phase II/III trial tested Anktiva + pembrolizumab versus standard chemotherapy in a randomized setting. Stopped early for futility on the primary endpoint, a legitimate data point. However, the full abstract’s conclusion (the portion cropped out in the screenshot in the thread) reads:
"While the study failed to continue accrual past IA1, there is an indication of a subgroup that might benefit from NP (Anktiva + pembrolizumab) with a potential OS difference at 12 months. NP was safe when compared to SoC, and responses were seen in both treatment arms, including partial and complete responses in the NP group. Evaluation of tumor and patient characteristics will be critical to define if there are those who may benefit from N-803 plus pembrolizumab."
Simply put, even though the trial stopped early for futility and did not reach statistical significance (HR 0.73, p=0.32), the authors noted a meaningful numerical improvement in 12-month survival: 44% alive with Anktiva + pembrolizumab versus 25% on standard chemotherapy. They explicitly called for better patient selection- exactly the type of insight the prospective ALC analysis in QUILT-3.055 delivers. Safety was also better on the Anktiva arm (34% vs 53% severe side effects), with tumor responses, including a complete response, observed.
The two trials asked different questions: QUILT-3.055 tested whether immune restoration correlates with survival; Lung-MAP tested a fixed combination against chemotherapy. One delivered a statistically significant, pre-specified surrogate signal; the other did not meet its bar for broad use. Neither erases the other.
The distinction is central to accelerated approval. The FDA allows approval based on a surrogate that is “reasonably likely” to predict clinical benefit in serious conditions with high unmet need (so patients do not have to wait years for large randomized survival studies). Here, the surrogate is reversal of chemotherapy-induced lymphopenia. The prospective ALC-survival correlation in QUILT-3.055, backed by Anktiva’s clear IL-15 mechanism, fits the FDA’s criteria. That is why RMAT designation (Feb 2025) and Expanded Access (June 2025) were granted for lymphopenia reversal across solid tumors.
This is the same Accelerated Approval playbook that let $MRK Merck Keytruda reach melanoma patients in 2014 on Phase 1 response rates, with large confirmatory survival data collected afterward. @ImmunityBio is following that path: BLA discussions for the lymphopenia indication are expected in second half 2026, with confirmatory randomized trials (including ResQ201A) to follow.
Cherry-picking the pooled mOS from one study while omitting the authors’ own subgroup nuance from another does not present a complete picture. Patients suffering chemotherapy-induced lymphopenia deserve the same consistent regulatory standards Merck received with Keytruda: surrogate first, confirmatory data second. The prospective evidence in QUILT-3.055 clearly meets that bar.
So while a lung-cancer-specific accelerated approval in the next 6-12 months is unlikely without stronger randomized data, a broader lymphopenia approval that includes lung cancer patients as part of the label is still very much on the table for 2H 2026 / early 2027.
English

Maybe you are not used to following companies closely that are working on cancer treatments, but ImmunityBio has very limited clinical results. I follow many other companies, they typically present results at research conferences two to three times per year. They place the posters from these presentations on their website. They submit major data milestones to peer-reviewed journals. They have a file called corporate presentation on their website. It is an extended PowerPoint file that includes the most relevant data to date.
ImmunityBio issues many press releases, but they do not include robust information on clinical results. They often prefer to talk about individual patients.
English

@AscendingBio I don’t think he needs to mis-present the data. All he has to do is present the details exactly as stated by the clinical results.
English

$IBRX FDA Letter Impact
What is clear from this letter is that $IBRX is going to need to use much more caution when appearing on TV shows and podcasts. They must avoid claims that are not supported yet with data.
Before receiving this public letter, there were other letters sent by the FDA. They state, "OPDP is concerned that, despite receiving these previous Untitled Letters, ImmunityBio continues to promote Anktiva in a similarly misleading manner."
Dr, Patrick and the company can face serious legal action if they do not make big changes.
My guess is that this is the final warning. The FDA is likely to refer this to the Justice Department if similar statements are made in the future.
fda.gov/inspections-co…
English

@cabriseno1 Ok, then take one of the FDA concerns in the letter and provide the data that supports a different view than that taken by the FDA.
English

There really is no need for a conspiracy theory here. The FDA has sent many letters to companies over the last few months identifying misrepresentation in their communications. Most of these are somewhat subtle concerns about advertising. (You can read these on the FDA website.)
The specific concerns cited in the ImmunityBio are not subtle. These are major infractions that are well documented. The only more alarming letter was the one the FDA sent to Hims and Hers.
Match the ImmunityBio statements to the available data and there is a big problem here.
If ImmunityBio does a reset, changes how they communicate, and quickly complies with the demands of this letter, that will likely be the end of it as far as the FDA is concerned.
I presume there would still be some risk of shareholder litigation. Usually that if futile and only helps the lawyers. Here, Dr. Patrick has independent assets that may change things.
English

@AscendingBio Big Pharma is really lean hard on their paid for puppets at the FDA. Pressure is mounting for them. They see Anktiva's efficacy and know their time is coming to an end.
English

@Neuroscope_mp @bfbuster1 I had one person report to me that they were rejected by multiple companies due to dual diagnosis. Yet, I agree that all they can say is no, so you ask.
English

You are correct on both.
Dual diagnosis is common, and therefore, it is hard to find patients for a very specific criterion.
This is why it's worthwhile to apply and see whether they would or could. Certain dual diagnoses or co-occurring pathologies are accepted.
I do not know which specifically.
English