ทวีตที่ปักหมุด
Biotech Autist
630 posts


Biotech Autist
@BiotechAutist
Regulatory Arbitrage. I short certainty.
เข้าร่วม Ağustos 2017
168 กำลังติดตาม1.9K ผู้ติดตาม

@Quantnova01 A PMR is not a regulatory get out of jail free card for a broken primary endpoint. Statutorily, the FDA uses PMRs to track the long-term durability or safety of an efficacy signal that already cleared the substantial evidence hurdle.
English

@BiotechAutist Considering LAD-1 is ultra - rare lethal disease with currently no approved treatment beyond high risk BM transplant , FDA might tolerate weaker control structure considering RCKT has adequately addressed CMC issue. Might go for post marketing requirement.
English

@RKamonere I think both will be approved. CMC looks clean on both
English

@BiotechAutist What’s your take on CING and GRCE sir? Are they worth a shout ahead of their PDUFA dates?
English

Again, thanks for the pushback. Very helpful for testing if my thesis actually survives scrutiny.
Accelerated approval does not mean “skip the usual proof if the data looks promising.” FDCA §506(c) only lets FDA use a surrogate that is “reasonably likely” to predict clinical benefit. It does not waive the need for substantial evidence from adequate and well-controlled investigations. And 21 CFR 314.126 is pretty clear that uncontrolled or partially controlled studies are not acceptable as the sole basis for approval.
That is the problem here. Open-label. Nonrandomized. No external/concurrent control. Heavy reliance on within-patient biomarker change. No multiplicity control for the biomarker package. A bunch of secondary/exploratory endpoints do not solve the inferential problem FDA statisticians actually have to assess.
And the Takeda RCT is not irrelevant just because it was imperfect. It is still the only public randomized human trial in this disease setting, and it gave FDA a concrete reason to doubt that substrate lowering necessarily translates into cognitive benefit. That does not prove Denali fails. It does prove this is nowhere close to the “no-brainer approval."
English

$DNLI $SPRB There is nothing wrong with using an open-label design for accelerated approval. The whole point of accelerated approval is to allow companies to skip typical steps when the data is promising. And yes safety was the primary objective but this is irrelevant because the secondary endpoints still assessed efficacy using a multitude of variables.
The rct you are talking about is not comparable or relevant because CSF GAG reduction is known to be a bad biomarker and we use mass spectometry now. They also waited too long to treat those patients and even if there would have been benefit it would've been hard to determine because they used older patients.
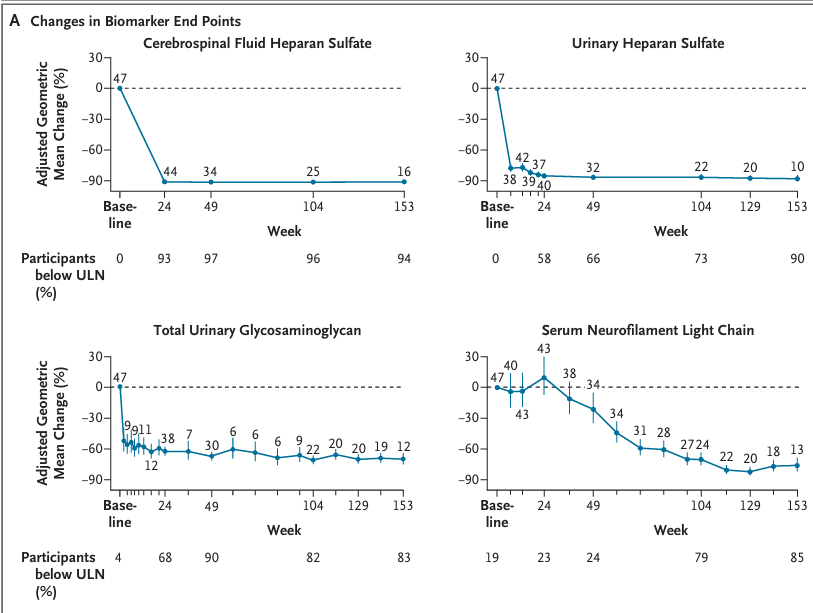
I'm not even invested in Denali but this is a no-brainer approval, I'm not sure why one would short here. They used much more objective biomarkers to measure sulfate decline. Furthermore, they already illustrated cognitive improvement from baseline through multiple endpoints and the NfL reduction is too dramatic to be explained by confounding.
In contrast Regenix data used a biomarker for sulfate decline that lacked academic consensus (although tbh I actually think the D2S6 biomarker was good along with the reasoning behind it but it was stupid not to include total HS reduction) and the length of treatment reflected in the data was only one year which was way too short. Would I have still approved it, probably but I'm not surprised the FDA declined to. But Denali is a different story altogether.
English

Appreciate the thoughtful pushback.
CDER vs CBER is not the point. The point is the file. DNLI still submitted an open-label, nonrandomized, no-control-arm phase 1/2 with safety as the primary objective. Mechanistic plausibility is not substantial evidence, and the only randomized controlled nMPS II trial with public results still showed meaningful CSF substrate lowering without a significant primary cognitive benefit. Different from RGNX, yes. Clearly approvable, I don't see it.
English

No rgnx used a different surrogate with less empirical backing, they also had poor data with very weak complementary variables. Denali’s drug will likely and in fact should be approved. Very clear mechanistic plausibility and they paired it with very convincing end point data over longer periods of time. Furthermore, it’s getting reviewed by CDER not CBER.
English

Appreciate the effort, but this is a deeply unserious read of the file.
1/ A Type C meeting is not approval.
It means FDA was willing to review the package on that theory. RGNX already showed that can still blow up at action.
2/ “Different from RGNX” does not solve the core problem.
DNLI still filed an open-label, nonrandomized, no-control-arm phase 1/2 with safety as the primary objective.
3/ The Takeda trial absolutely matters.
It is the only randomized controlled trial in neuronopathic MPS II we have results of, and it gave FDA a concrete reason to doubt that lowering CSF substrate reliably translates into cognitive benefit.
4/ BBB penetration is mechanism, not evidence.
A better mechanistic story is not the same thing as substantial evidence.
5/ The clinical signals are supportive.
They are not a substitute for control.
6/ Process chatter is not evidence.
No AdCom, labeling, PDUFA extension, launch prep, Royalty, Jefferies, none of that answers approvability.
7/ COMPASS being enrolled helps confirmatory posture.
It does not make the filed package is enough.
Regardless, this is still an uncontrolled biomarker package, and the approval case is basically “maybe FDA stretches.” That can happen. But dressing that up as some obvious base-case approval call is just cope.
Ps: Stop having grok write your messages, it's embarrassing.
English

@BiotechAutist I appreciate the regulatory arbitrage lens, but this one misses the DNLI-specific facts that differentiate it from RGNX.
1/ The Phase 1/2 (N=47, open-label, safety-primary) is exactly what the FDA agreed could support accelerated approval on the pre-specified surrogate. Denali announced post-Sept 2024 Type C CDER meeting:
“Agreement was reached that cerebrospinal fluid heparan sulfate (CSF HS) is reasonably likely to predict clinical benefit and can be used as a surrogate endpoint.”
Public press release, not slide-deck font size. That’s on the record since 2024 and never walked back.
2/ RGNX’s CRL (Feb 7, 2026) did hit the surrogate, but on CSF HS D2S6 (a specific disaccharide fragment), with eligibility/natural-history-control issues in a small AAV gene-therapy dataset (N≈13-20 range).
DNLI is using total CSF HS (the full substrate), ERT modality (FDA has decades of GAG/HS precedent), larger N=47 + up to 201+ weeks follow-up, plus clinical signals: Vineland adaptive behavior gains/stabilization, cognition, hearing thresholds, liver normalization, and serum NfL ↓76%. NEJM publication (Jan 2026 issue) backs it. Different package.
3/ The PMC10826424 RCT you cite? Intrathecal idursulfase (non-brain-penetrant delivery). No comparison to a TfR-enabled systemic ERT that actually crosses the BBB like DNL310. And Denali’s data does show downstream clinical correlates the literature trial missed.
4/ Process signals are the opposite of “safety study trying to talk its way in”:
- Oct 2025 PDUFA extension = purely clinical pharmacology update (FDA explicitly: “not related to efficacy, safety, or biomarkers”). No new data requested.
- Late-cycle meeting done, labeling discussions underway, commercial readiness “fully established,” COMPASS neuronopathic cohort fully enrolled Dec 2025 for confirmatory data.
- No AdCom scheduled or hinted. Company still saying “on the cusp of launching” (Jan 2026 update).
Royalty Pharma wired $275 tied to US accelerated approval. Analysts (Jefferies post-RGNX note, consensus) still see 70%+ odds.
FDA doesn’t need to “act like adults” by issuing a CRL here; they just need to honor their own documented alignment on a differentiated surrogate in a clean review. This isn’t RGNX 2.0.
Happy to be proven wrong on April 6, but the facts say approval base case still >65%
English

Appreciate the thoughtful thread and the useful pushback. I agree FDA has left the door open in principle here. But that is not the real question. The real question is whether this specific application, on this specific record, clears substantial evidence of effectiveness. And I still think that is a much harder case than your thread suggests. DNLI is asking FDA to move from open-label, safety-primary, biomarker-heavy data to a conclusion on effectiveness. Showing that approval is legally possible is not the same as showing this file is sufficient.
English

couple thoughts on this $dnli thread. overall vastly overplays the negative hand imho. but shorts gotta go full throttle.
1/ yes a p1/2 open label w/ primary obj of safety; biomarker, adaptive behavior, & liver-vol secondary. this has been known from the beginning.
Biotech Autist@BiotechAutist
1/ I’m SHORT $DNLI. They filed a safety study as their pivotal trial in the BLA. This application relies entirely on the clinical assumption that the FDA employs morons who treat uncontrolled substrate shifts like stone tablets from God.
English

@BiotechAutist Thanks, but i was meaning if you had the full paper since its paywalled haha
English

@BiotechAutist Hey man, congrats on the ALDX news! I wanted to ask if you could share the Kresaldi NEJM paper with me. I used to use Sci-Hub, but it’s not working anymore and I’m not sure what the best way to get it is these days
English

@travfourr Probably like 40. Bought 15p for .55. Should be 450% roi if it hits 12
English

12/ $DNLI is a safety study trying to win on a surrogate that just got publicly bloodied in the same disease
And if FDA decides to act like adults, this ends exactly how it should:
CRL
Follow @BiotechAutist, or keep trading based on the font size of corporate slide decks.
English



