.
2.2K posts


A year ago, Dr. Prasad came to the FDA to implement 4 major long-lasting reforms: 2-to-1 pivotal trial requirement, national priority reviews, a risk-stratified covid vaccine framework, & the new plausible mechanism framework for ultra rare diseases which we launched last week. Also, under his leadership, his center hit a record number of approvals in Dec. He got a tremendous amount accomplished within his one-year sabbatical from UCSF and will be returning back to his academic home later next month. We will name a successor before his departure.
I want to thank him for his service and personal sacrifice to take time away from his family.
English

It’s not just public posturing.
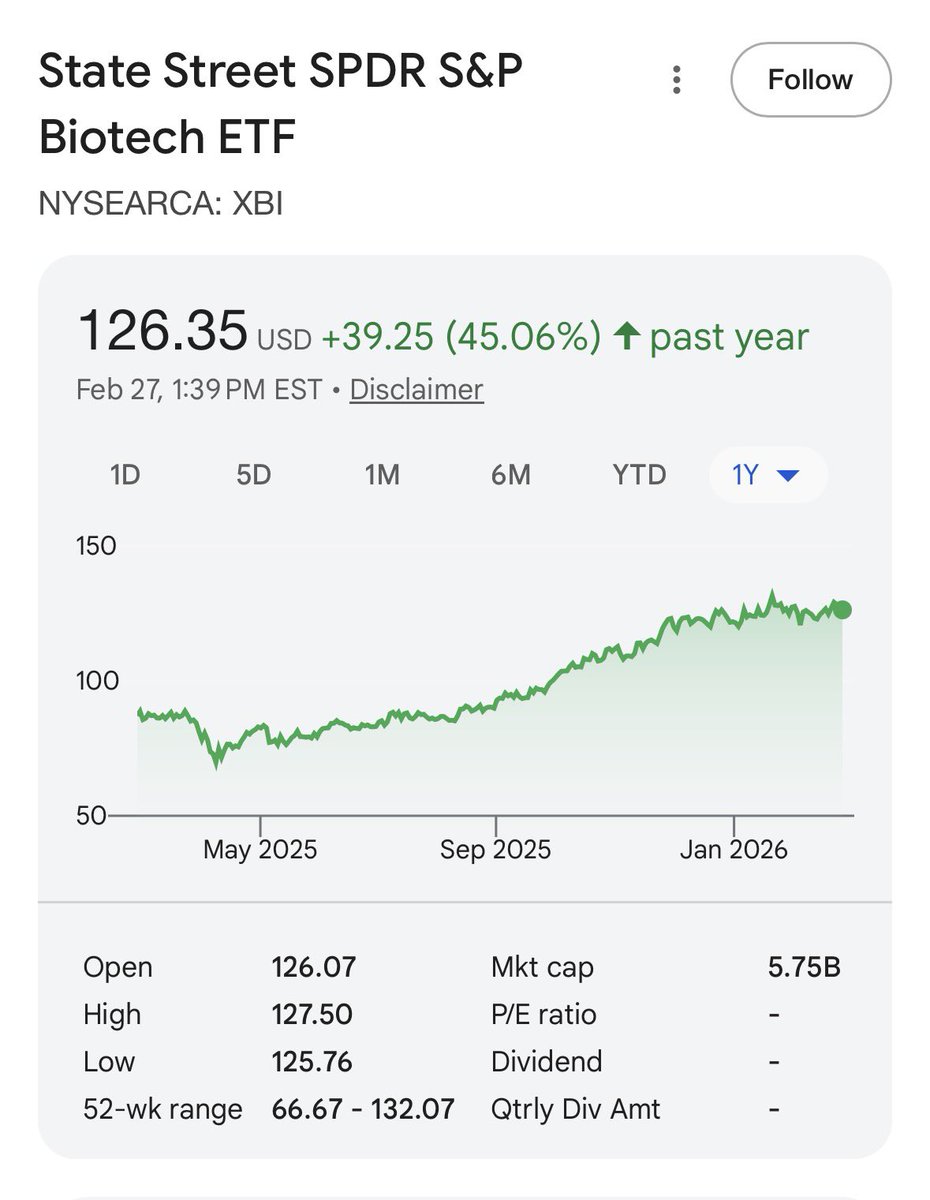
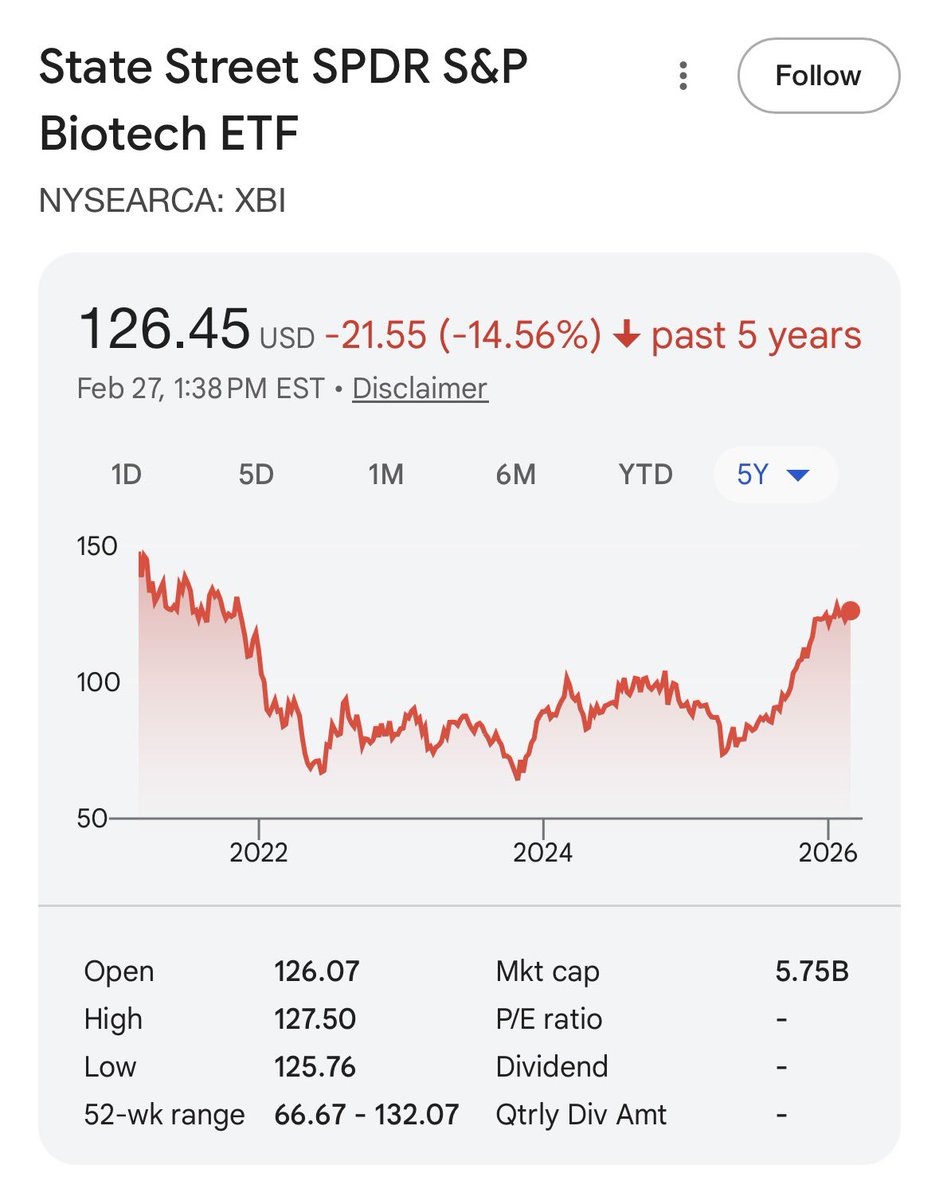
XBI - which is a reasonable measure of investor confidence in drug approvals- is up over the last year.
Down big 2021-2024. No daily sky falling replace the FDA headlines I can recall from that time.
I’m sorry Moderna’s platform appears to have some real problems - wld be even higher if not for that.
Brian Skorney@BrianSkorney
Vinay is an agent of restrictive regulatory policy. Maybe the strictest in my career observing the agency. But what the administration advertises to the public is flexibility unlike anything ever seen. Regulatory policy should be determined by society. We can choose to have no bar or only allow things that are certain to market or anything in between. But having Makary talking about “plausible mechanism” approvals while VP hands out RTFs. The crux of the issue is that they are telling the stakeholders they are tacking right when they are really tacking left. And now they are starting to receive the blowback from those stakeholders who realize this.
English

Have to give @VivekGRamaswamy a lot of credit for putting himself out there. I don’t agree with everything he says or often his style, but he has shown how a key to success is going for it. Interested to see if his biotech experience impacts things either way the next four years.
English

I call BS on $CRDF clinical data and its plan to jump headlong into front-line settings with onvansertib. Via @ApexOnco -> oncologypipeline.com/apexonco/anoth…
English

$RLAY Updated dose escalation results for ReFocus, a first-in-human study of highly selective FGFR2 inhibitor RLY-4008 in cholangiocarcinoma and other solid tumors. #ASCO23
PR: ir.relaytx.com/news-releases/…

English