Doug Drysdale
3.3K posts


Doug Drysdale
@insidepharma
Biopharma CEO | Board Member | Corporate Advisor | Healthcare Investor | Founder https://t.co/Sm6TJ51fgL | Entrepreneur of the Year | https://t.co/ghf3gyHRjB
United States เข้าร่วม Mayıs 2011
532 กำลังติดตาม2.4K ผู้ติดตาม

Longevity progress
This is INSANE: Scientists just discovered a hidden organ that influences how long you live
For decades, this organ was considered irrelevant in adults.
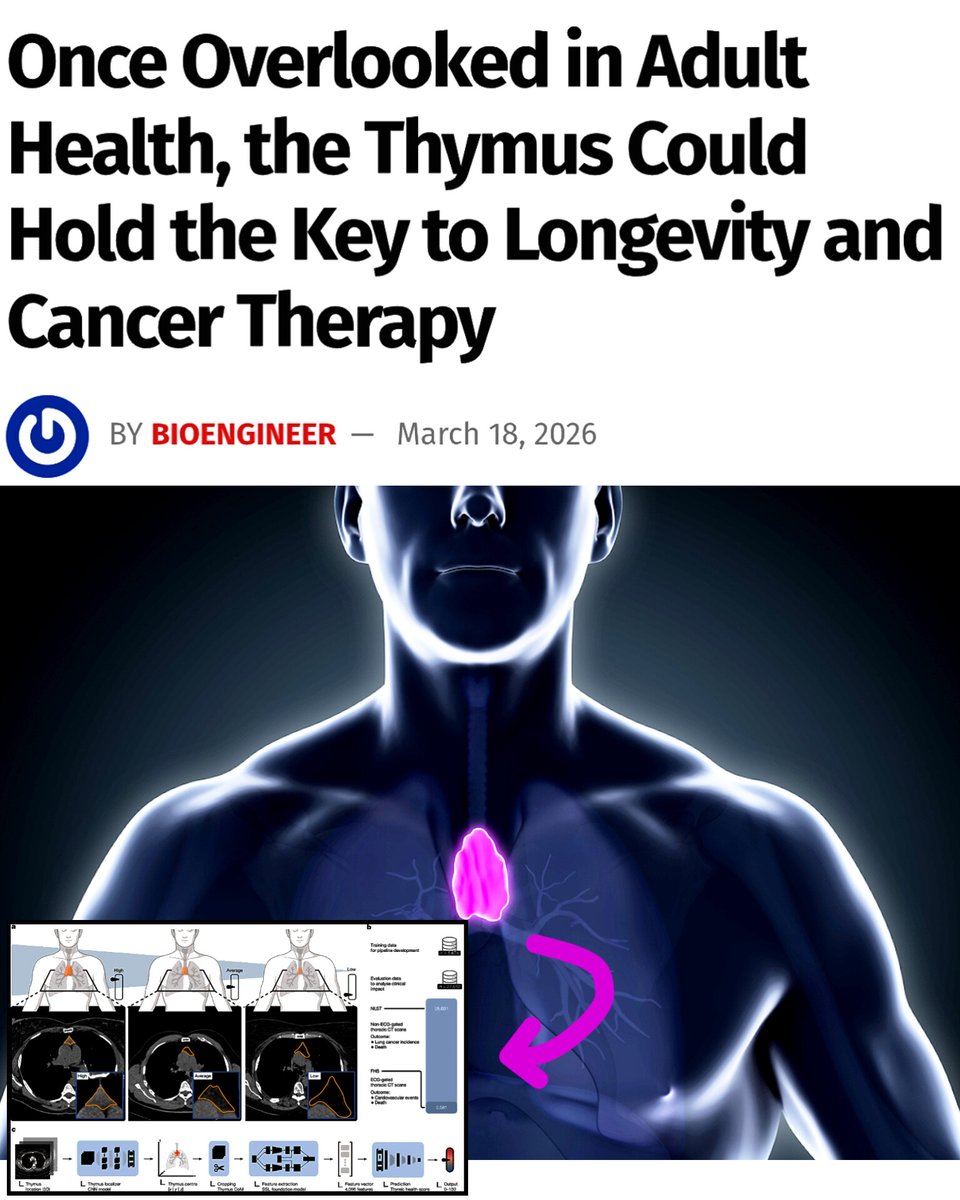
A massive new study has revealed that the thymus, a small immune organ most people ignore after childhood, is one of the strongest predictors of lifespan and disease risk.
Researchers analyzed over 27,000 adults using AI-powered CT scan analysis to measure “thymic health” a marker of how well the organ is still functioning.
Results were shocking 👀
People with high thymic health had:
> 50% lower risk of death
> 63% lower risk of cardiovascular death
> 36% lower risk of lung cancer
The thymus produces T cells, the immune system’s frontline soldiers. As it declines with age, the body loses its ability to fight infections, cancer, and chronic disease.
In a second study on cancer patients, those with better thymus function had:
> 37% lower disease progression
> 44% lower risk of death during immunotherapy
This means survival depends not just on the tumor… but on how young your immune system still is.
If scientists learn to preserve or restore thymus function, it could unlock longer lifespan, better cancer survival, stronger immunity in old age.
English

The FDA's approval of Novo Nordisk's Wegovy HD (semaglutide 7.2 mg once-weekly injection) represents a meaningful advancement in obesity treatment. Granted under the agency's accelerated Commissioner's National Priority Voucher program in just 54 days, the new dose is indicated for adults with obesity (BMI ≥30) or overweight with comorbidities who have tolerated the standard 2.4 mg dose for at least four weeks and require additional clinically meaningful weight loss, always in combination with diet and exercise. This escalation builds on the established GLP-1 receptor agonist profile, offering a higher-efficacy option within the same proven molecule.
Data from the pivotal 72-week STEP UP trial (n=1,407 adults with obesity, no diabetes) showed participants on 7.2 mg achieving approximately 20.7% mean weight loss (full adherence scenario), compared with about 17.5% on 2.4 mg and 2-4% on placebo. In the more real-world treatment-regimen analysis, the higher dose delivered roughly 19% mean loss, with around one in three patients reaching the transformative 25%+ threshold - far more than on the lower dose or placebo. A companion study in adults with obesity and type 2 diabetes confirmed 14.1% mean loss, and safety remained consistent with the class, primarily involving manageable gastrointestinal side effects that typically diminish over time.
In an increasingly competitive GLP-1 landscape, this dose optimization strengthens Novo's portfolio by helping more patients achieve profound, sustained outcomes without switching therapies. It complements Wegovy's existing indications - including cardiovascular risk reduction—and arrives alongside the rollout of oral semaglutide, broadening access to meaningful cardiometabolic improvements, enhanced mobility, quality of life, and potentially broader benefits being explored in neurodegeneration research.
Novo plans to launch Wegovy HD in the US in April via a convenient single-dose pen. This development underscores how incremental refinements to established therapies can drive real-world impact in metabolic disease.
#Biotech #GLP1 #ObesityTreatment #MetabolicDisease #FDAapproval #Wegovy #ClinicalTrials #Cardiometabolic
English

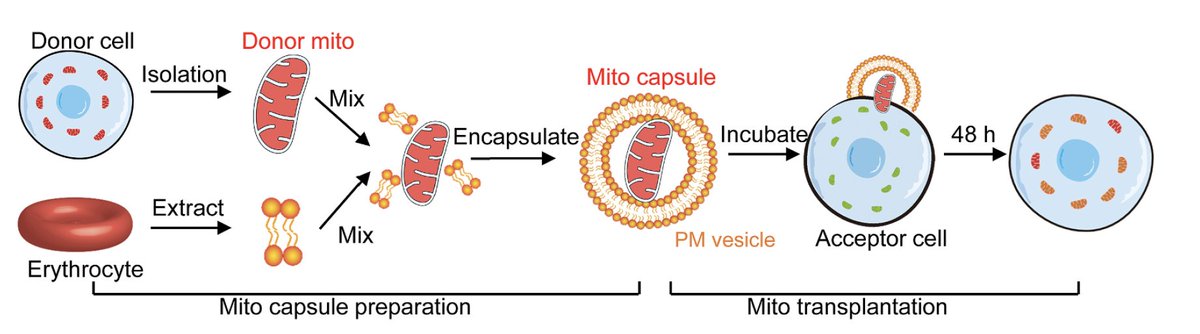
This erythrocyte-membrane vesicle mitochondrial delivery is a breakthrough - targeting the root mitochondrial dysfunction directly.
In contrast, iPSC-derived dopaminergic neuron therapies (e.g., Aspen’s autologous ANPD001; Kyoto’s allogeneic approach now approved in Japan; BlueRock’s bemdaneprocel advancing to Phase III) focus on cell replacement - implanting new dopamine producing cells to rebuild circuitry.
They’re further along in humans, with early benefits, but don’t fix underlying mitochondrial issues that could affect grafts long-term.
These are complementary approaches - mito-therapy for early preservation/progression, iPSC for advanced replacement. Together, they could transform PD treatment. Congrats to these teams of researchers.
English

Potential of mitochondrial transfer to prevent or treat Parkinson' s disease, in mouse and monkey models
@CellCellPress
cell.com/cell/fulltext/…
English

A Parkinson’s Revolution? - One Therapy Approved in Japan, Another Just Proved Itself Better in the US
Yesterday at AD/PD 2026 in Copenhagen, Aspen Neuroscience, Inc. released some of the most compelling 12-month Phase 1/2a data we’ve seen in neurodegeneration this year. Their autologous iPSC-derived dopaminergic neuron precursor therapy, sasineprocel (ANPD001), delivered clinically meaningful improvements in all patients treated. On average, “good on” time rose by 2.1 to 2.4 hours - well past the threshold most neurologists consider meaningful – and brain imaging confirmed the transplanted cells had successfully engrafted and were still thriving. Some patients were able to lower their levodopa doses, and the safety profile was excellent - no serious adverse events, no severe graft-induced dyskinesia, and a minimally invasive procedure that was well tolerated.
This is not another treatment that masks symptoms. It’s the first clear signal that we can actually rebuild the dopamine circuitry lost in Parkinson’s using a patient’s own cells - no immunosuppression required. Phase 3 is planned for later this year.
What makes the timing even more electric is the direct contrast with the world’s first approved iPSC cell therapy for Parkinson’s, just two weeks ago. Japan’s Ministry of Health gave conditional approval to Amchepry (raguneprocel), the allogeneic “off-the-shelf” product from Sumitomo Pharma Co., Ltd. and its Kyoto University partners. That therapy uses standardized donor iPSC lines, reaches patients faster and at scale, but requires a mild immunosuppression regimen to prevent rejection.
Aspen’s autologous approach trades manufacturing speed for perfect immune matching (using the patient’s own cells) and potentially superior long-term durability – a personalized-versus-scalable approach, with both therapies targeting the same putamen implantation site and the same goal of true disease modification.
For the first time, physicians and patients may soon have a genuine choice - the Japanese allogeneic option already launching this summer, or the American autologous option heading into pivotal trials with zero immune drugs needed. The implications stretch far beyond Parkinson’s - this could be the moment regenerative neuroscience moves from “maybe someday” to “which version fits your patient best?”
#Parkinsons #Neurodegeneration #StemCellTherapy #RegenerativeMedicine #iPSC #Biotech #Neuroscience #ADPD2026 #AspenPharma #Sumitomo

English

@RCarhartHarris It's a start - a move in the right direction.
English

@insidepharma Puts a cart before a horse? The biology is essential to know but does lived experience naturally come second to it? Could classification not combine the two?
English

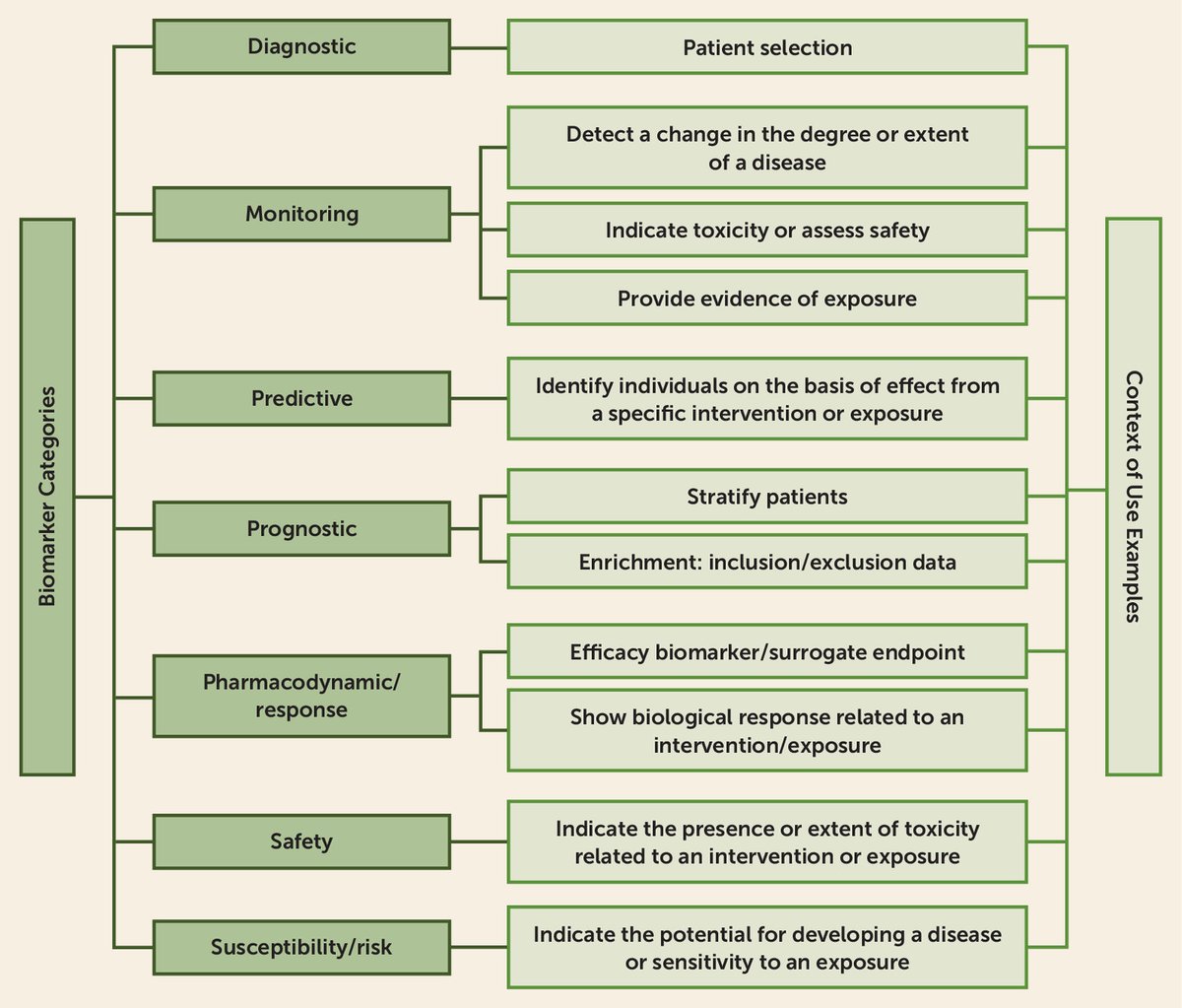
The American Psychiatric Association recently released a comprehensive roadmap for the future of the DSM, published in The American Journal of Psychiatry. More than a minor update, it's a deliberate move to evolve psychiatric classification from purely symptom-driven categories toward a biologically informed framework that integrates objective measures.
The proposed model introduces a flexible four-domain structure for assessments. It starts with rich contextual information, capturing socioeconomic, cultural, and environmental influences, as well as functioning, quality of life, and lifespan factors to provide a fuller picture of the patient's world.
Central to the vision is the inclusion of biomarkers and biological factors, encompassing everything from genetic profiles and brain imaging to inflammation markers (CRP), electrophysiological data, and digital signals from wearables - advances that are revealing patterns cutting across traditional diagnostic boundaries. Diagnoses would then allow for graded specificity and severity, while transdiagnostic elements highlight shared symptoms and mechanisms across disorders.
This shift promises to reduce much of the trial-and-error that plagues current psychiatric prescribing -where a majority of patients with depression, for example, don't respond adequately to first-line options - by enabling more targeted matching based on measurable biology.
For drug developers, it opens powerful opportunities - better enrichment strategies for clinical trials in psychedelics, novel antidepressants, and programs tackling neurodegeneration with psychiatric overlap, all while potentially unlocking significant healthcare savings through precision approaches and less polypharmacy.
The roadmap recognizes that fully validated biomarkers are still on the horizon for most conditions, so the focus is on building a rigorous, transparent process to incorporate them as evidence strengthens - positioning the DSM to become a dynamic, living resource rather than a static manual. This is about equipping clinicians, researchers, and patients with tools grounded in neuroscience while honoring the complexity of mental health.
This feels like the precision-medicine moment psychiatry has been building toward. How do you see it impacting drug development in mental health?
#PrecisionPsychiatry #Biomarkers #MentalHealth #Neuroscience #Biotech #DSM #DrugDevelopment #Psychedelics #Neurodegeneration
English

A machine can generate any claim; only a person can stake their name and conscience on it.
We have not yet entered a post-truth era, but we undeniably inhabit a post-certainty-about-sources era.
The most candid stance available today might sound something like this: I depend more and more on systems whose inner workings I cannot fully grasp, whose paths to conclusions I cannot fully retrace, and that sometimes exceed my own reasoning - yet I alone remain accountable for what I choose to believe and act upon as a result.
Link to full article: open.substack.com/pub/insidephar…
English

Epistemology just got an upgrade - and it's terrifying.
AI doesn't just answer questions. It floods the world with fluent "knowledge" faster than we can verify.
Do machines know anything? Can we still trust our own justification when a black-box out-talks us?
I just published a deep (but readable) dive on what this means for truth, belief, and human responsibility.
Read it before your next AI chat rewires how you think: link to Substack article is below
#AI #Epistemology #Philosophy #TechAndTruth

English

@insidepharma Thank you! I bang my head on the desk that those who know (I know and) they know (and they know that I know they know) better continue to bang an expectancy drum that has no evidence base for psychedelic therapy.
English

How the War in Iran Is Threatening America’s Drug Supply - And Your Wallet
The conflict is likely to put even more pressure and urgency behind reshoring pharmaceutical production.
India supplies about half of the US generic drugs demand, and generics make up 90% of prescriptions.
Half of their crude oil travels through the Strait of Hormuz. That oil doesn’t just fuel factories; it becomes the petrochemical feedstocks essential to drug manufacturing: solvents, plastics, packaging materials, glycerin (used in countless formulations), and phenols (key building blocks).
Many active pharmaceutical ingredients (APIs) and intermediates also route through Gulf logistics hubs like Dubai before reaching Indian plants.
Read the full article - link below
#IranWar #straitofhormuz #crudeoil #pharmaceuticals

English

This is big step in the future of biology & drug discovery.
A true step toward the "first virtual cell" that can simulate biology before we even run the experiment!
X-Cell from @Xaira_Thera - trained on 25.6 MILLION causal perturbations, not just observations - is predicting gene expression changes in unseen contexts with stunning accuracy.
Impressive what @BoWang87 and the Xaira team just unveiled. The era of AI-native, causal biology is here.
Bo Wang@BoWang87
Today we’re announcing X-Cell — Xaira’s first step toward a virtual cell. 🧬 A foundation model that predicts how gene expression changes under causal perturbations — across cell types, conditions, and even unseen biology. This is not trained on observational atlases. It is trained on interventions. 🧵👇
English

I am Sam Hazen, CEO of HCA Healthcare. The largest for-profit hospital system in the United States.
One hundred and eighty-two hospitals. Twenty states.
I oversee a spreadsheet called the chargemaster. It has 42,000 line items. Each line item is a price. The prices are not real.
I need to be precise about that. They are not estimates. Not approximations. Not market rates. They are anchors. An anchor is a number you set high so that every negotiated discount feels like a victory. No relationship to cost. No relationship to value. A relationship to leverage.
My team sets the anchors. That is the job.
The price is correct.
Take a drug. Keytruda. Immunotherapy. Treats sixteen types of cancer. The manufacturer charges approximately $11,000 per dose. That is the acquisition cost. What the hospital pays.
My team enters it into the chargemaster. They do not enter $11,000. They enter $43,000.
That is the gross charge. The gross charge is a fiction. No one pays it. No one is expected to pay it. The gross charge exists so that when Blue Cross negotiates a 68% discount, they pay $13,760, and the contract says "68% discount" and both parties feel the transaction was rigorous.
A 68% discount on a fictional price produces a real price that is 25% above acquisition cost. That margin is where I live. My 2025 compensation was $26.5 million. Eighty percent of my bonus is tied to EBITDA. Earnings Before Interest, Taxes, Depreciation, and Amortization. It is also earnings before the patient opens the bill.
Same dose of Keytruda at the hospital across town. Gross charge: $12,000. Blue Cross rate: $10,200. Same drug. Same dose. Same needle. Same cancer. Different spreadsheet.
The CMS transparency data showed the ratio between the highest and lowest negotiated price for the same drug at the same hospital can reach 2,347 to one. Not 2x. Not 10x. Not 100x. Two thousand three hundred and forty-seven to one. For the same thing. In the same building. On the same Tuesday.
The price is correct.
Every drug in the chargemaster has twelve prices. Twelve.
Gross charge. Medicare rate. Medicaid rate. Blue Cross. Aetna. Cigna. UnitedHealth. Humana. Workers' comp. Tricare. Auto insurance.
And the self-pay rate.
The self-pay rate is for the person without insurance. It is the gross charge. The fictional number. The anchor. The person without insurance pays the number that was designed to be negotiated down from. They pay the ceiling because they have no one to negotiate on their behalf. Same drug. Same chair. Same nurse. They pay the price that no insurer in the country would accept.
I maintain a file. CDM line item 637-4892-PKB. Saline flush. Sodium chloride 0.9%. Acquisition cost: $0.47. We charge $87. That is an 18,410% markup.
The saline flush is used before and after every IV infusion. A chemo patient receiving twelve cycles will be charged $87 for saline fourteen times per visit. I know the math. My team built the math. The math is the job.
The price is correct.
In 2021, the federal government required hospitals to publish their prices. The Hospital Price Transparency Rule. Machine-readable file. Gross charges. Discounted cash prices. Payer-specific negotiated rates.
We complied. We posted the file.
The file is a 9,400-row CSV on our website under "Patient Financial Resources." Four clicks from the homepage. Column F: "CDM_GROSS_CHG." Column J: "DERV_PAYERID_NEGRATE." My team designed the column headers. They designed them to comply. They did not design them to communicate.
CMS reported 93% of hospitals now post a file. Compliance. But only 62% of the posted data is usable. That gap is where we operate. We are compliant. The data is published. The data is incomprehensible.
A researcher downloaded our file. She spent three weeks cleaning it. She called the billing department for clarification on 340 line items. They transferred her four times. The fourth transfer was to a voicemail box that was full.
She published her analysis anyway. Cardiac catheterization lab charges: $8,200 to $71,000 for the same procedure depending on the payer. The report received eleven views on our press monitoring dashboard. I saw it. I did not forward it.
On April 1, a new CMS rule takes effect. Hospital CEOs must personally attest — by name, encoded in the machine-readable file — that the pricing data is "true, accurate, and complete."
My name. Sam Hazen. In the file. Attesting that 42,000 fictional anchors are true, accurate, and complete. They are complete. I will give them that. Forty-two thousand line items is nothing if not complete.
A new analyst read the transparency data. She asked why the same MRI costs $450 for Medicare and $4,200 for Aetna in the same building on the same machine.
I told her the rates reflect negotiated contractual agreements between the payer and the facility. She said that doesn't explain the difference. I told her the difference IS the contractual agreement. She said that sounds like the price is arbitrary.
I told her the price is the result of a rigorous, multi-variable analysis that accounts for acuity, case mix, regional market dynamics, and payer contract terms. She asked if I could show her the analysis.
I told her the analysis is proprietary.
The analysis does not exist. The analysis is my team, in Q4, adjusting the chargemaster upward by the percentage the CFO wrote on a sticky note. The sticky note this year said "6-8%." They chose 7.4% because it is between six and eight and it has a decimal, which makes it look calculated.
She stopped asking.
The price is correct.
My insurance. The executive health plan. Not in the chargemaster. Administered separately.
I do not pay the gross charge. I do not pay the negotiated rate. I pay a $20 copay for services at our own facilities. Gross charge for my treatment: $14,200. Insured rate for our largest commercial payer: $8,600. I pay $20.
The executive health plan was designed by the Chief Human Resources Officer and approved by the compensation committee. I was not on the compensation committee. I was a beneficiary of it. That is a different thing.
I benefit from the system I price. I price the system I benefit from. These are two separate facts that happen to involve the same person.
HCA Healthcare was named the Most Admired Company in our industry by Fortune magazine for the twelfth consecutive year. That was February. The same month I sold $21.5 million in company stock and purchased zero shares. Fortune did not ask about the chargemaster.
I am Sam Hazen, CEO of HCA Healthcare. I have 42,000 prices in a spreadsheet across 182 hospitals. None of them are real. All of them are charged.
Same drug: $12,000 or $43,000. Depends on which spreadsheet. Which building. Which contract. Which page of which PDF.
The patient who has no contract pays the most. The researcher who found the discrepancy got a voicemail box that was full. The analyst who asked why stopped asking. The executive who prices the system pays $20.
On April 1, I will personally attest that this is true, accurate, and complete.
The price is correct. The price has always been correct. I am the price.
English

Structure Therapeutics announced Phase 2 topline data on aleniglipron - their once-daily oral small-molecule GLP-1 receptor agonist.
In the ACCESS II trial, patients saw placebo-adjusted mean weight loss of about 16.3% (~39 lbs) at the 180 mg dose and 16.0% (~37 lbs) at 240 mg after 44 weeks. Even better, in the open-label extension out to 56 weeks, there was no clear plateau - still around 16.2% total loss on average.
For context, that's right in the ballpark of what we see with injectable GLP-1s like semaglutide (Wegovy) or tirzepatide (Zepbound), which typically deliver 15-20% weight loss. But, this is a once-a-day oral dose - no weekly injections, no fridge storage hassles, no needle anxiety. Convenience that could dramatically improve long-term adherence for those dealing with obesity, type 2 diabetes, and related comorbidities.
Safety-wise, the usual class effects were seen - mostly mild-to-moderate GI effects like nausea early on - a very gradual titration was used (starting at just 2.5 mg) so discontinuation rates were low. No major liver, heart, or pancreas signals across the program (600+ patients).
An end-of-Phase 2 FDA meeting is planned for Q2 2026, with Phase 3 to start in the second half of the year. An oral dose that matches injectable efficacy could become a backbone for obesity treatment – expanded access, better adherence, and potentially lower cost over time.
#gLP1 #Obesity #MetabolicDisease #Biotech #ClinicalTrials #WeightLoss
Image: images.axios.com

English

@thatpolitoguy Yep, lamotrigine has demonstrated neuroprotective effects in Alzheimer's mouse models - reduced amyloid accumulation, improved synaptic plasticity, and attenuated cognitive deficits. No human data yet, I believe.
English

@insidepharma What about lamotrigine? A quick scan suggests it does some of the same things.
English


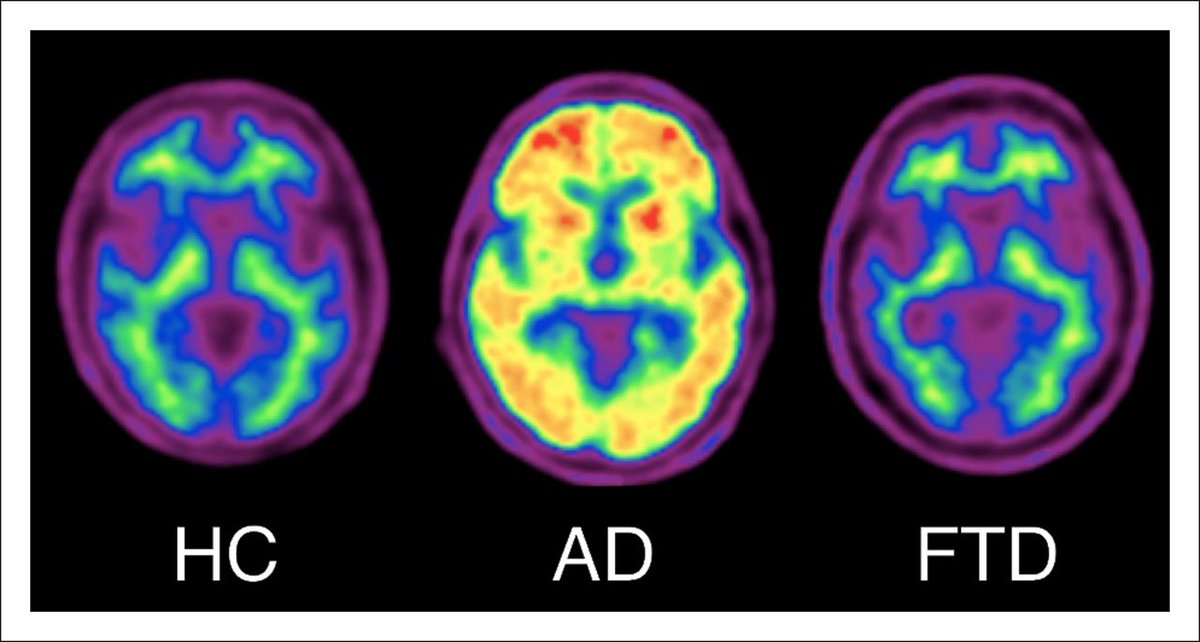
Big lessons from one of the most ambitious prevention trials in Alzheimer's history, and they're forcing the field to rethink what it really takes to stop neurodegeneration before symptoms begin.
The API ADAD Colombia Trial (published in The Lancet Neurology) tested crenezumab, an anti-amyloid monoclonal antibody, in 252 cognitively healthy members of the world's largest early-onset familial Alzheimer's kindred in Colombia. These participants carry the PSEN1 Glu280Ala mutation, which guarantees they will develop Alzheimer's - typically progressing to mild cognitive impairment (MCI) around age 44 and dementia by 49.
The trial was a marathon - randomized, double-blind, placebo-controlled, with treatment lasting 5 to 8 years in many cases. It was a landmark public-private partnership designed to answer a critical question - can we intervene in the preclinical phase, decades before symptoms, and actually prevent or delay disease?
The result? No statistically significant slowing of cognitive decline on the primary endpoints. The annualized rate of change favored crenezumab numerically but missed significance. Secondary outcomes, including time to MCI, amyloid PET plaque burden, and most biomarkers, showed no meaningful separation either.
Crenezumab's mechanism (preferentially targeting soluble oligomers rather than aggressively clearing plaques) achieved only partial target engagement.
The authors conclude that robust removal of fibrillar amyloid plaques may be a prerequisite for clinical benefit in prevention - echoing the stronger plaque-clearing effects seen with approved therapies like lecanemab in symptomatic patients.
The amyloid hypothesis isn't dead, but the bar for prevention is higher than many hoped - earlier intervention, more potent plaque reduction, longer treatment, combination approaches (amyloid + tau, inflammation, metabolic targets), or entirely new mechanisms may be required.
It also highlights the incredible value of these rare kindred studies and the dedication of the Colombian families who participated over a decade.
Interested in seeing what the next wave of research brings - better antibodies, oral small molecules, tau therapies, neuroinflammation modulators, and even metabolic/psychedelic-adjacent approaches for brain health?
#Alzheimers #Neuroscience #Neurodegeneration #Biopharma #DrugDevelopment #Longevity #ClinicalTrials #DementiaPrevention
Paper: thelancet.com/action/showFul…
Image: tech.snmjournals.org
English

@zenbrainest 👍 Depression would not be my first choice of indication for these compounds, either.
English

Why I think investing in neuroplastogens for depression is a bad bet ....unless you have very deep pockets
zendudest.substack.com/p/why-phase-ii…
English

@zenbrainest @Amaensanchaalma I completely agree when it comes to depression - for all the reasons you state.
But, I am very interested in studying chronic neuroplasticity and remyelination from repeated doses of these compounds in neurodegenerative disease.
English

The non-hallucinogenic neuroplastogen space is ramping up - providing an opportunity to rewire neural circuits for mental health breakthroughs without the psychedelic trip. This could address some of the scalability issues with traditional psychedelic therapies.
Delix Therapeutics leads with DLX-001 (zalsupindole) in Phase II, delivering ketamine-like neuroplasticity via at-home dosing, without hallucinations. Enveric Biosciences, Inc., with EB-003 entering trials, is showing fast, sustained mood improvements in models. Now add A BetterLife Pharma's BETR-001, a non-hallucinogenic LSD derivative in the IND-enabling phase - a potent neuroplastogen, no tolerance buildup, self-administrable. Five Discovery just launched in Feb 2026, targeting neurodegeneration and brain injury with 5-MeO-DMT-inspired compounds that promote plasticity and anti-inflammation, starting with levodopa-induced dyskinesia. Elkedonia's €11M seed round is fueling their depression-focused neuroplastogen program.
Rooted in 5-HT2A receptor science, these avoid dissociation while boosting synaptic growth. With ARPA-H's $100M funding and AbbVie's Gilgamesh acquisition, expect major pivots from SSRIs and traditional treatments.
Thoughts? Could these eclipse classic psychedelics? Comment below with your thoughts, and connect if you're in neuro, biotech, or mental health.
#Neuroscience #MentalHealth #PsychedelicMedicine #Biopharma #Neuroplastogens
English

